Abstract
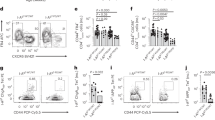
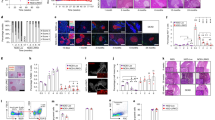
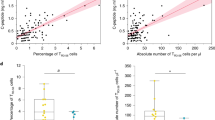
For unknown reasons, autoimmune diseases such as type 1 diabetes develop after prolonged periods of inflammation of mononuclear cells in target tissues1. Here we show that progression of pancreatic islet inflammation to overt diabetes in nonobese diabetic (NOD) mice is driven by the ‘avidity maturation’ of a prevailing, pancreatic beta-cell-specific T-lymphocyte population carrying the CD8 antigen. This T-lymphocyte population recognizes two related peptides (NRP and NRP-A7)2 in the context of H-2Kd class I molecules of the major histocompatibility complex (MHC). As pre-diabetic NOD mice age, their islet-associated CD8+ T lymphocytes contain increasing numbers of NRP-A7-reactive cells, and these cells bind NRP-A7/H-2Kd tetramers with increased specificity, increased avidity and longer half-lives. Repeated treatment of pre-diabetic NOD mice with soluble NRP-A7 peptide blunts the avidity maturation of the NRP-A7-reactive CD8+ T-cell population by selectively deleting those clonotypes expressing T-cell receptors with the highest affinity and lowest dissociation rates for peptide–MHC binding. This inhibits the local production of T cells that are cytotoxic to beta cells, and halts the progression from severe insulitis to diabetes. We conclude that avidity maturation of pathogenic T-cell populations may be the key event in the progression of benign inflammation to overt disease in autoimmunity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Delovitch, T. & Singh, B. The nonobese diabetic mouse as a model of autoimmune diabetes: immune disregulation gets the NOD. Immunity 7, 727–738 ( 1997).
Anderson, B., Park, B. J., Verdaguer, J., Amrani, A. & Santamaria, P. Prevalent CD8+ T cell response against one peptide/MHC complex in autoimmune diabetes. Proc. Natl Acad. Sci. USA 96, 9311– 9316 (1999).
Nagata, M., Santamaria, P., Kawamura, T., Utsugi, T. & Yoon, J.-W. Evidence for the role of CD8+ cytotoxic T cells in the destruction of pancreatic beta cells in NOD mice. J. Immunol. 152, 2042–2050 (1994).
Katz, J., Benoist, C. & Mathis, D. Major histocompatibility complex class I molecules are required for the generation of insulitis in non-obese diabetic mice. Eur. J. Immunol. 23, 3358–3360 (1993).
Wicker, L. et al. β2-microglobulin-deficient NOD mice do not develop insulitis or diabetes. Diabetes 43, 500– 504 (1994).
Serreze, D., Leiter, E., Christianson, G., Greiner, D. & Roopenian, D. Major histocompatibility complex class I-deficient NOD.β1mnull mice are diabetes and insulitis resistant. Diabetes 43, 505– 508 (1994).
Verdaguer, J. et al. Acceleration of spontaneous diabetes in TCRβ-transgenic nonobese diabetic mice by beta cell-cytotoxic CD8+ T cells expressing identical endogenous TCRα chains. J. Immunol. 157, 4726–4735 (1996).
Verdaguer, J. et al. Spontaneous autoimmune diabetes in monoclonal T cell nonobese diabetic mice. J. Exp. Med. 186, 1663– 1676 (1997).
DiLorenzo, T. et al. Major histocompatibility complex class I-restricted T cells are required for all but the end stages of diabetes development in nonobese diabetic mice and use prevalent T cell receptor α chain gene rearrangement. Proc. Natl Acad. Sci. USA 95, 12538– 12543 (1998).
Santamaria, P. et al. Beta cell cytotoxic CD8+ T cells from non-obese diabetic mice use highly homologous T cell receptor alpha chain CDR3 sequences. J. Immunol. 154, 2494–2503 (1995).
Rammensee, H. G., Friede, T. & Stevanoviic, S. MHC ligands and peptide motifs: first listing. Immunogenetics 41, 178–228 (1995).
Wong, S. et al. Identification of an MHC class I-restricted autoantigen in type 1 diabetes by screening an organ-specific cDNA library. Nature Med. 5, 1026–1031 ( 1999).
Savage, P. A., Boniface, J. J. & Davis, M. M. A kinetic basis for T cell receptor repertoire selection during an immune response. Immunity 10, 485-492 (1999).
Abiru, N. & Eisenbarth, G. Autoantibodies and autoantigens in type 1 diabetes: role in pathogenesis, prediction and prevention. Can. J. Diab. Care 23, 59–65 (1999).
Kaufman, D. et al. Spontaneous loss of T-cell tolerance to glutamic acid decarboxylase in murine insulin-dependent diabetes. Nature 366, 69–72 (1993).
Tisch, R. et al. Immune response to glutamic acid decarboxylase correlates with insulitis in non-obese diabetic mice. Nature 366, 72–75 (1993).
Wegmann, D. The immune response to islet in experimental diabetes and insulin-dependent diabetes mellitus. Curr. Opin. Immunol. 8, 860–864 (1996).
Alam, S. et al. T-cell receptor affinity and thymocyte positive selection. Nature 381, 616–620 ( 1996).
Kersh, G., Kersh, E., Fremont, D. & Allen, P. High and low potency ligands with similar affinities for the TCR: the importance of kinetics in TCR signaling. Immunity 9, 817– 826 (1998).
Lyons, D. et al. A TCR binds to antagonist ligands with lower affinities and faster dissociation rates than to agonists. Immunity 5, 53–61 (1996).
Matsui, K., Boniface, J., Steffner, P., Reay, P. & Davis, M. Kinetics of T-cell receptor binding to peptide/I-Ak complexes:correlation of the dissociation rate with T-cell responsiveness. Proc. Natl Acad. Sci. USA 91, 12862–12866 (1994).
Monks, C., Freiberg, B., Kupfer, H., Sciaky, N. & Kupfer, A. Three dimensional segregation of supramolecular activation clusters in T-cells. Nature 395, 82– 86 (1998).
Reich, Z. et al. Ligand-specific oligomerization of T-cell receptor molecules. Nature 387, 617–620 (1997).
Altman, J. et al. Direct visualization and phenotypic analysis of virus-specific T lymphocytes in HIV-infected individuals. Science 274, 94–96 (1996).
Acknowledgements
The authors thank S. Bou, K. Rouleau, M. Deumas and S. Culp for animal care; S. Thiessen for scoring insulitis; M Bevan, K. Kane and T. Utsugi for reagents; S. Wong for testing the INS tetramer; Y. Yang for reading the manuscript; and H. Kominek for editorial assistance. This work was supported by grants from the Medical Research Council of Canada, the MRC/Juvenile Diabetes Foundation International (JDFI) Program II and the Canadian Diabetes Association (to P.S.). A.A. was supported by fellowships from the Alberta Heritage Foundation for Medical Research (AHFMR) and the JDFI. P.S. is a Senior Scholar of the AHFMR. R.T. and P.S. are members of the Beta Cell Apoptosis Network (BetaCAN).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Amrani, A., Verdaguer, J., Serra, P. et al. Progression of autoimmune diabetes driven by avidity maturation of a T-cell population. Nature 406, 739–742 (2000). https://doi.org/10.1038/35021081
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/35021081
This article is cited by
-
Re-programming mouse liver-resident invariant natural killer T cells for suppressing hepatic and diabetogenic autoimmunity
Nature Communications (2022)
-
Not-so-opposite ends of the spectrum: CD8+ T cell dysfunction across chronic infection, cancer and autoimmunity
Nature Immunology (2021)
-
Increased yields and biological potency of knob-into-hole-based soluble MHC class II molecules
Nature Communications (2019)
-
Antigen-specific therapeutic approaches for autoimmunity
Nature Biotechnology (2019)
-
Gut microbiota metabolites for sweetening type I diabetes
Cellular & Molecular Immunology (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.