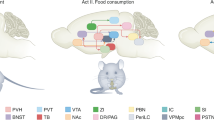
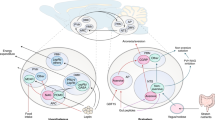
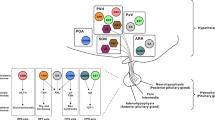
Abstract
New information regarding neuronal circuits that control food intake and their hormonal regulation has extended our understanding of energy homeostasis, the process whereby energy intake is matched to energy expenditure over time. The profound obesity that results in rodents (and in the rare human case as well) from mutation of key signalling molecules involved in this regulatory system highlights its importance to human health. Although each new signalling pathway discovered in the hypothalamus is a potential target for drug development in the treatment of obesity, the growing number of such signalling molecules indicates that food intake is controlled by a highly complex process. To better understand how energy homeostasis can be achieved, we describe a model that delineates the roles of individual hormonal and neuropeptide signalling pathways in the control of food intake and the means by which obesity can arise from inherited or acquired defects in their function.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Edholm, O. G. Energy balance in man. J. Human Nutr. 31, 413–431 (1977).
Montague, C. et al. Congenital leptin deficiency is associated with severe early-onset obesity in humans. Nature 387, 903– 908 (1997).
Clement, K. et al. A mutation in the human leptin receptor gene causes obesity and pituitary dysfunction. Nature 392, 330 –331 (1998).
Vaisse, C., Clement, K., Guy-Grand, B. & Froguel, P. A frameshift mutation in human MC4R is associated with a dominant form of obesity. Nature Genet. 20, 113– 114 (1998).
Yeo, G. et al. A frameshift mutation in MC4R associated with dominantly inherited human obesity. Nature Genet. 20, 111– 112 (1998).
Harris, R. B. S., Kasser, T. R. & Martin, R. J. Dynamics of recovery of body composition after overfeeding, food restriction or starvation of mature female rats. J. Nutr. 116, 2536–2546 ( 1986).
Kennedy, G. C. The role of depot fat in the hypothalamic control of food intake in the rat . Proc. R. Soc. Lond. B 140, 579– 592 (1953).
Gibbs, J., Young, R. C. & Smith, G. P. Cholecystokinin decreases food intake in rats. J. Comp. Physiol. Psychol. 84, 488– 495 (1973).
Baura, G. et al. Saturable transport of insulin from plasma into the central nervous system of dogs in vivo: a mechanism for regulated insulin delivery to the brain. J. Clin. Invest. 92, 1824– 1830 (1993).
Woods, S., Lotter, E., McKay, L. & Porte, D. J. Chronic intracerebroventricular infusion of insulin reduces food intake and body weight of baboons. Nature 282, 503–505 ( 1979).
Zhang, Y. et al. Positional cloning of the mouse obese gene and its human homologue . Nature 372, 425–432 (1994).
Bagdade, J. D., Bierman, E. L. & Porte, D. Jr The significance of basal insulin levels in the evaluation of the insulin response to glucose in diabetic and nondiabetic subjects. J. Clin. Invest. 46, 1549– 1557 (1967).
Considine, R.V. et al. Serum immunoreactive-leptin concentrations in normal-weight and obese humans. New Eng. J. Med. 334, 292–295 (1996).
Schwartz, M.W. et al. Cerebrospinal fluid leptin levels: relationship to plasma levels and to adiposity in humans. Nature Med. 2, 589–593 (1996).
Baskin, D. G., Wilcox, B. J., Figlewicz, D. P. & Dorsa, D. M. Insulin and insulin-like growth factors in the CNS. Trends Neurosci. 11, 107–111 ( 1988).
Baskin, D., Breininger, J. & Schwartz, M. Leptin receptor mRNA identifies a subpopulation of neuropeptide Y neurons activated by fasting in rat hypothalamus. Diabetes 48, 828–833 (1999).
Cheung, C., Clifton, D. & Steiner, R. Proopimelanocortin neurons are direct targets for leptin in the hypothalamus. Endocrinology 138, 4489–4492 (1997).
Weigle, D.S. et al. Recombinant ob protein reduces feeding and body weight in the ob/ob mouse. J. Clin. Invest. 96, 2065–2070 (1995).
Campfield, L., Smith, F., Gulsez, Y., Devos, R. & Burn, P. Mouse OB protein: evidence for a peripheral signal linking adiposity and central neural networks. Science 269, 546– 549 (1995).
Sipols, A. J., Baskin, D. G. & Schwartz, M. W. Effect of intracerebroventricular insulin infusion on diabetic hyperphagia and hypothalamic neuropeptide gene expression. Diabetes 44, 147–151 ( 1995).
Porte, D. J., Woods, S., Baskin, D., Seeley, R. & Schwartz, M. Obesity, diabetes and the central nervous system. Diabetologia 41, 863–881 (1998).
Schwartz, M.W. et al. Evidence that plasma leptin and insulin levels are associated with body adiposity via different mechanisms. Diabetes Care 20, 1476–1481 (1997).
Kahn, S.E. et al. Quantification of the relationship between insulin sensitivity and B-cell function in human subjects: evidence for a hyperbolic function . Diabetes 42, 1663–1672 (1993).
Polonsky, K. S., Given, B. D. & VanCauter, E. Twenty-four hour profiles and pulsatile patterns of insulin secretion in normal and obese subjects. J. Clin. Invest. 81, 442–448 ( 1988).
Mueller, W. et al. Evidence that glucose metabolism regulates leptin secretion from cultured rat adipocytes. Endocrinology 139, 551–558 (1998).
Wang, J., Liu, R., Hawkins, M., Barzilai, N. & Rossetti, L. A nutrient-sensing pathway regulates leptin gene expression in muscle and fat. Nature 393, 684– 688 (1998).
Leedom, L. J. & Meehan, W. P. The psychoneuroendocrinology of diabetes mellitus in rodents. Psychoneuroendocrinology 14, 275–294 (1989).
Havel, P. et al. Marked and rapid decreases of circulating leptin in streptozotocin diabetic rats: reversal by insulin. Am. J. Physiol. 274, R1482–R1491 (1998).
Hathout, E. et al. Changes in plasma leptin during the treatment of diabetic ketoacidosis. J. Clin. Endocrinol. Metab. 84, 4545–4548 (1999).
Sindelar, D. et al. Low plasma leptin levels contribute to diabetic hyperphagia in rats. Diabetes 48, 1275– 1280 (1999).
Chua, S.C. et al. Phenotypes of mouse diabetes and rat fatty due to mutations in the OB (Leptin) receptor. Science 271, 994–996 (1996).
Mantzoros, C. et al. Severe leptin resistance in brown fat-deficient uncoupling protein promoter-driven diphtheria toxin A mice despite suppression of hypothalamic neuropeptide Y and circulating corticosterone. Diabetes 47, 230–238 (1998).
Marsh, D.J. et al. Response of melanocortin-4 receptor-deficient mice to anorectic and orexigenic peptides. Nature Genet. 21, 119–122 (1999).
Wilson, B. et al. Physiological and anatomical circuitry between Agouti-related protein and leptin signaling. Endocrinology 140, 2387–2397 (1999).
Bjorbaek, C. et al. Expression of leptin receptor isoforms in rat brain microvessels . Endocrinology 139, 3485– 3491 (1998).
Caro, J.F. et al. Decreased cerebrospinal-fluid/serum leptin ratio in obesity: a possible mechanism for leptin resistance. Lancet 348, 159–161 (1996).
Bjorbaek, C., Elmquist, J., Frantz, J., Shoelson, S. & Flier, J. Identification of SOCS-3 as a potential mediator of central leptin resistance. Mol. Cell 1, 619–625 (1998).
Stanley, B. G., Kyrkouli, S. E., Lampert, S. & Leibowitz, S. F. Neuropeptide Y chronically injected into the hypothalamus: a powerful neurochemical inducer of hyperphagia and obesity. Peptides 7, 1189–1192 (1986).
Billington, C. J., Briggs, J. E., Grace, M. & Levine, A. S. Effects of intracerebroventricular injection of neuropeptide Y on energy metabolism. Am. J. Physiol. 260, R321–R327 ( 1991).
Zarjevski, N., Cusin, I., Vetter, R., Rohner-Jeanrenaud, F. & Jeanrenaud, B. Chronic intracerebroventricular neuropeptide-Y administration to normal rats mimics hormonal and metabolic changes of obesity . Endocrinology 133, 1753– 1758 (1993).
Kalra, S. P., Dube, M. G., Sahu, A., Phelps, C. P. & Kalra, P. Neuropeptide Y secretion increases in the paraventricular nucleus in association with increased appetite for food. Proc. Natl Acad. Sci. USA 88, 10931–10935 (1991).
White, J. D. & Kershaw, M. Increased hypothalamic neuropeptide Y expression following food deprivation. Mol. Cell. Neurosci. 1, 41–48 (1989).
Wilding, J. P.H. et al. Increased neuropeptide-Y messenger ribonucleic acid (mRNA) and decreased neurotensin mRNA in the hypothalamus of the obese (ob/ ob) mouse. Endocrinology 132, 1939– 1944 (1993).
Schwartz, M.W. et al. Specificity of leptin action on elevated blood glucose levels and hypothalamic neuropeptide Y gene expression in ob/ob mice. Diabetes 45, 531–535 ( 1996).
Stephens, T.W. et al. The role of neuropeptide Y in the antiobesity action of the obese gene product. Nature 377, 530– 534 (1995).
Erickson, J. C., Hollopeter, G. & Palmiter, R. D. Attenuation of the obesity syndrome of ob/ob mice by the loss of neuropeptide Y. Science 274, 1704–1707 (1996).
Williams, G. et al. Increased neuropeptide Y concentrations in specific hypothalamic regions of streptozocin-induced diabetic rats. Diabetes 38, 321–327 (1989).
Erickson, J. C., Clegg, K. E. & Palmiter, R. D. Sensitivity to leptin and susceptibility to seizures of mice lacking neuropeptide Y. Nature 381, 415–418 (1996).
Cone, R. et al. The melanocortin receptors: agonists, antagonists, and the hormonal control of pigmentation. Rec. Prog. Horm. Res. 51, 287–318 (1996).
Bray, G. A., Fisler, J. & York, D. A. Neuroendocrine control of the development of obesity: understanding gained from studies of experimental animal models. Front. Neuroendocrinol. 11, 128–181 (1990).
Kow, L. & Pfaff, D. The effects of the TRH-metabolite cyclo(His-Pro) and its analogs on feeding. Pharmacol. Biochem. Behav. 38, 359–364 ( 1991).
Kristensen, P. et al. Hypothalamic CART is a new anorectic peptide regulated by leptin. Nature 393, 72– 76 (1998).
Luheshi, G., Gardner, J., Rushforth, D., Loudon, A. & Rothwell, N. Leptin actions on food intake and body temperature are mediated by IL-1. Proc. Natl Acad. Sci. USA 96, 7047–7052 ( 1999).
Mountjoy, K., Mortrud, M., Low, M., Simerly, R. & Cone, R. Localization of the melanocortin-4 receptor (MC4-R) in neuroendocrine and autonomic control circuits in the brain. Mol. Endocrinol. 8, 1298–1308 (1994).
Fan, W., Boston, B., Kesterson, R., Hruby, V. & Cone, R. Role of melanocortinergic neurons in feeding and the agouti obesity syndrome. Nature 385 , 165–168 (1997).
Huszar, D. et al. Targeted disruption of the melanocortin-4 receptor results in obesity in mice. Cell 88, 131– 141 (1997).
Miller, M. et al. Cloning of the mouse agouti gene predicts a secreted protein ubiquitously expressed in mice carrying the lethal yellow mutation. Genes Dev. 7, 454–467 ( 1993).
Shutter, J. et al. Hypothalamic expression of ART, a novel gene related to agouti, is up-regulated in obese and diabetic mutant mice. Genes Dev. 11, 593–602 (1997).
Ollmann, M. et al. Antagonism of central melanocortin receptors in vitro and in vivo by agouti-related protein. Science 278, 135–138 (1997).
Hahn, T., Breininger, J., Baskin, D. & Schwartz, M. Coexpression of Agrp and NPY in fasting-activated hypothalamic neurons. Nature Neurosci. 1, 271–272 (1998).
Broberger, C., Johansen, J., Johasson, C., Schalling, M. & Hokfelt, T. The neuropeptide Y/agouti gene-related protein (AGRP) brain circuitry in normal, anorectic, and monosodium glutamate-treated mice. Proc. Natl Acad. Sci. USA 95, 15043 –15048 (1998).
Hagan, M. et al. Effect of agouti-related peptide (83-132) on food intake: competitive melanocortin-receptor antagonism on short but not long term effects. Am. J. Physiol. (in the press).
Rossi, M. et al. A C-terminal fragment of Agouti-related protein increases feeding and antagonizes the effect of alpha-melanocyte stimulating hormone in vivo . Endocrinology 139, 4428– 4431 (1998).
Stellar, E. The physiology of motivation. Psychol. Rev. 61, 5 (1954).
Woods, S., Seeley, R., Porte, D. J. & Schwartz, M. Signals that regulate food intake and energy homeostasis. Science 280, 1378–1383 (1998).
Elias, C. et al. Leptin activates hypothalamic CART neurons projecting to the spinal cord. Neuron 21, 1375– 1385 (1998).
Satoh, N. et al. The arcuate nucleus as a primary site of satiety effect of leptin in rats. Neurosci. Lett. 224, 149– 152 (1997).
Tang-Christensen, M., Holst, J., Hartmann, B. & Vrang, N. The arcuate nucleus is pivotal in mediating the anorectic effects of centrally administered leptin . Neuroreport 10, 1183– 1187 (1999).
Dawson, R., Pelleymounter, M., Millard, W., Liu, S. & Eppler, B. Attenuation of leptin-mediated effects by monosodium glutamate-induced arcuate nucleus damage. Am. J. Physiol. 273, E202–E206 (1997).
Schwartz, M. et al. Leptin increases hypothalamic proopiomelanocortin (POMC) mRNA expression in the rostral arcuate nucleus. Diabetes 46, 2119–2123 (1997).
Thornton, J., Cheung, C., Clifton, D. & Steiner, R. Regulation of hypothalamic proopiomelanocortin mRNA by leptin in ob/ob mice. Endocrinology 138, 5063–5066 (1997).
Hagan, M. et al. Role of the CNS melanocortin system in the response to overfeeding . J. Neurosci. 19, 2362– 2367 (1999).
Seeley, R. et al. Melanocortin receptors in leptin effects. Nature 390, 349 (1997).
Elmquist, J., Maratos-Flier, E., Saper, C. & Flier, J. Unraveling the central nervous system pathways underlying responses to leptin . Nature Neurosci. 1, 445– 450 (1998).
Elmquist, J., Elias, C. & Saper, C. From lesions to leptin: hypothalamic control of food intake and body weight. Neuron 22, 221– 232 (1999).
Stanley, B. G., Willett, V. L. III, Donias, H. W., Ha, L. H. & Spears, L. C. The lateral hypothalamus: a primary site mediating excitatory amino acid-elicited eating . Brain Res. 630, 41–49 (1993).
Dallman, M.F. et al. Feast and famine: Critical role of glucocorticoids with insulin in daily energy flow. Front. Neuroendocrinol. 14, 303–347 (1993).
Verbalis, J., Blackburn, R., Hoffman, G. & Stricker, E. Establishing behavioral and physiological functions of central oxytocin: insights from studies of oxytocin and ingestive behaviors. Adv. Exp. Med. Biol. 395, 209–225 ( 1995).
Qu, D. et al. A role for melanin-concentrating hormone in the central regulation of feeding behaviour. Nature 380, 243– 247 (1996).
Shimada, M., Tritos, N., Lowell, B., Flier, J. & Maratos-Flier, E. Mice lacking melanin-concentrating hormone are hypophagic and lean. Nature 39, 670– 674 (1998).
Saito, Y. et al. Molecular characterization of the melanin-concentrating-hormone receptor. Nature 400, 265– 269 (1999).
Chambers, J. et al. Melanin-concentrating hormone is the cognate ligand for the orphan G-protein-coupled receptor SLC-1. Nature 400 , 261–265 (1999).
de Lecea, L. et al. The hypocretins: hypothalamus-specific peptides with neuroexcitatory activity. Proc. Natl Acad. Sci. USA 95, 322–327 (1998).
Sakurai, T. et al. Orexins and orexin receptors: a family of hypothalamic neuropeptides and G-protein-coupled receptors that regulate feeding behavior. Cell 92, 573–585 ( 1998).
Hagan, J. et al. Orexin A activates locus coeruleus cell firing and increases arousal in the rat. Proc. Natl Acad. Sci. USA 96, 10911–10916 (1999).
Chemelli, R. et al. Narcolepsy in orexin knockout mice: molecular genetics of sleep regulation. Cell 98, 437– 451 (1999).
Figlewicz, D. P., Stein, L. J., West, D., Porte, D. Jr & Woods, S. C. Intracisternal insulin alters sensitivity to CCK-induced meal suppression in baboons. Am. J. Physiol. 250, R856–R860 (1986).
Woods, S. C. & Strubbe, J. H. The psychobiology of meals. Psychonomic Bull. Rev. 1, 141–155 (1994).
Leibowitz, S. & Alexander, J. Analysis of neuropeptide Y-induced feeding: dissociation of Y1 and Y2 receptor effects on natural meal patterns . Peptides 12, 1251–1260 (1991).
Flynn, M., Scott, T., Pritchard, T. & Plata-Salaman, C. Mode of action of OB protein (leptin) on feeding. Am. J. Physiol. 275, R174–R179 (1998).
Ritter, S., Dinh, T. & Friedman, M. Induction of Fos-like immunoreactivity (Fos-li) and stimulation of feeding by 2,5-anhydro-D-mannitol (2,5-AM) require the vagus nerve. Brain Res. 646, 53– 64 (1994).
Travers, S. & Norgren, R. Gustatory neural processing in the hindbrain. Annu. Rev. Neurosci. 10, 595– 632 (1987).
Friedman, M., Harris, R., Ji, H., Ramirez, I. & Tordoff, M. Fatty acid oxidation affects food intake by altering hepatic energy status. Am. J. Physiol. 276, R1046–R1053 (1999).
Moran, T. & Schwartz, G. Neurobiology of cholecystokinin . Crit. Rev. Neurobiol. 9, 1– 28 (1994).
Grill, H. & Smith, G. Cholecystokinin decreases sucrose intake in chronic decerebrate rats. Am. J. Physiol. 254, R853–R856 (1988).
Matson, C. & Ritter, R. Long-term CCK-leptin synergy suggests a role for CCK in the regulation of body weight. Am. J. Physiol. 276, R1038–R1045 ( 1999).
Emond, M., Schwartz, G., Ladenheim, E. & Moran, T. Central leptin modulates behavioral and neural responsivity to CCK. Am. J. Physiol. 276, R1545–R1549 (1999).
Ter Horst, G., de Boer, P., Luiten, P. & van Willigen, J. Ascending projections from the solitary tract nucleus to the hypothalamus. A Phaseolus vulgaris lectin tracing study in the rat. Neuroscience 31, 785–797 (1989).
Grill, H., Ginsberg, A., Seeley, R. & Kaplan, J. Brainstem application of melanocortin receptor ligands produces long-lasting effects on feeding and body weight. J. Neurosci. 18, 10128– 10135 (1998).
Mercer, J., Moar, K. & Hoggard, N. Localization of leptin receptor (Ob-R) messenger ribonucleic acid in the rodent hindbrain. Endocrinology 139, 29–34 (1998).
Bronstein, D., Schafer, M., Watson, S. & Akil, H. Evidence that beta-endorphin is synthesized in cells in the nucleus tractus solitarius: detection of POMC mRNA. Brain Res. 587, 269– 275 (1992).
Leibowitz, S., Roossin, P. & Rosenn, M. Chronic norepinephrine injection into the hypothalamic paraventricular nucleus produces hyperphagia and increased body weight in the rat. Pharmacol. Biochem. Behav. 21, 801–808 (1984).
Oltmans, G. Norepinephrine and dopamine levels in hypothalamic nuclei of the genetically obese mouse (ob/ob). Brain Res. 273, 369–373 (1983).
Brunetti, L., Michelotto, B., Orlando, G. & Vacca, M. Leptin inhibits norepinephrine and dopamine release from rat hypothalamic neuronal endings. Eur. J. Pharmacol. 372, 237–240 (1999).
Salamone, J., Mahan, K. & Rogers, S. Ventrolateral striatal dopamine depletions impair feeding and food handling in rats. Pharmacol. Biochem. Behav. 44, 605–610 (1993).
Szczypka, M. et al. Feeding behavior in dopamine-deficient mice. Proc. Natl Acad. Sci. USA 96, 12138–12143 (1999).
Pothos, E., Creese, I. & Hoebel, B. Restricted eating with weight loss selectively decreases extracellular dopamine in the nucleus accumbens and alters dopamine response to amphetamine, morphine, and food intake. J. Neurosci. 15, 6640–6650 (1995).
Figlewicz, D., Endocrine regulation of neurotransmitter transporters. Epilepsy Res. 37, 203–210 ( 1999).
Patterson, T. et al. Food deprivation decreases mRNA and activity of the rat dopamine transporter. Neuroendocrinology 68, 11– 20 (1998).
Leibowitz, S. & Alexander, J. Hypothalamic serotonin in control of eating behavior, meal size, and body weight. Biol. Psychiatry 44, 851–864 (1998).
Nonogaki, K., Strack, A., Dallman, M. & Tecott, L. Leptin-independent hyperphagia and type 2 diabetes in mice with a mutated serotonin 5-HT 2C receptor gene. Nature Med. 4, 1152 –1156 (1998).
Calapai, G. et al. Leptin increases serotonin turnover by inhibition of brain nitric oxide synthesis. J. Clin. Invest. 104, 975–982 (1999).
Yaswen, L., Diehl, N., Brennam, M. & Hochgeschwender, V. Obesity in the mouse model of pro-opiomelanocortin deficiency responds to peripheral melanocortin. Nature Med. 5, 1066– 1070 (1999).
Farooqi, I. et al. Effects of recombinant leptin therapy in a child with congenital leptin deficiency. N. Engl. J. Med. 341, 879–884 (1999).
Cheatham, B. & Kahn, C. Insulin action and the insulin signaling network. Endocr. Rev. 16, 117– 142 (1995).
Baskin, D. G., Sipols, A. J., Schwartz, M. W. & White, M. F. Insulin receptor substrate-1 (IRS-1) expression in rat brain. Endocrinology 134, 1952–1955 (1994).
Kapeller, R. et al. Tyrosine phosphorylation of tub and its association with Src homology 2 domain-containing proteins implicate tub in intracellular signaling by insulin. J. Biol. Chem. 274, 24980– 24986 (1999).
Tartaglia, L.A. et al. Identification and expression cloning of a leptin receptor, OB-R. Cell 83, 1263–1271 (1995).
Tartaglia, L. A. The leptin receptor. J. Biol. Chem. 272, 6093–6096 (1997).
Spanswick, D., Smith, M., Groppi, V., Logam, S. & Ashford, M. Leptin inhibits hypothalamic neurons by activation of ATP-sensitive potassium channels. Nature 390, 521–525 (1997).
Dunn- Meynell, A., Rawson, N. & Levin, B. Distribution and phenotype of neurons containing the ATP-sensitive K+ channel in rat brain. Brain Res. 814, 41–54 (1998).
Acknowledgements
Assistance in manuscript preparation provided by L. Walters and in figure preparation by M. Baskin is gratefully acknowledged. This work was supported by NIH grants, the Diabetes Endocrinology Research Center and Clinical Nutrition Research Unit of the University of Washington, and the Department of Veterans Affairs.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Schwartz, M., Woods, S., Porte, D. et al. Central nervous system control of food intake. Nature 404, 661–671 (2000). https://doi.org/10.1038/35007534
Issue Date:
DOI: https://doi.org/10.1038/35007534
This article is cited by
-
Sex-specific neurobehavioral and biochemical effects of developmental exposure to Malathion in offspring mice
Naunyn-Schmiedeberg's Archives of Pharmacology (2024)
-
Polymorphisms and expressions of ADSL, MC4R and CAPN1 genes and their effects on economic traits in Egyptian chicken breeds
Molecular Biology Reports (2024)
-
Breast feeding, obesity, and asthma association: clinical and molecular views
Clinical and Molecular Allergy (2023)
-
GADD45A regulates subcutaneous fat deposition and lipid metabolism by interacting with Stat1
BMC Biology (2023)
-
Effect of acute and long-term exercise on leptin levels in depressed outpatients
BMC Public Health (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.