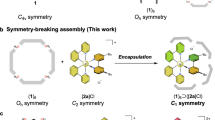
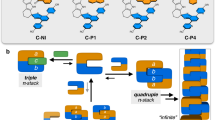
Abstract
‘Container’ complexes in which a guest molecule is held mechanically within a cage-like host have been known for over a decade1,2. They provide a means to stabilize reactive intermediates3 and to create new forms of stereoisomerism4. Molecular capsules held together by hydrogen bonds are more recent; they are formed reversibly on timescales of milliseconds to hours, long enough formolecular motions5 and even reactions6 to be seen for the encapsulated species. Here we describe the synthesis and characterization of a hydrogen-bonded molecular capsule of nanometre dimensions, which is large enough to encapsulate two different molecules. This allows us to explore the size- and shape-selectivity of the encapsulation process: we see, for example, the exclusive formation of the hetero-guest pair when benzene and p -xylene are both added to a solution. This presumably reflects optimal occupancy of the capsule—two benzene guests leave too much empty space in the interior, and two p -xylene molecules make it too crowded.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Cram, D. J. & Cram, J. M. Container Molecules and Their Guests(RSC, Cambridge, MA, (1994)).
Collet, A.et al. Cyclotriveratrylenes and cryptophanes: their synthesis and applications to host–guest chemistry and to the design of new materials. Top. Curr. Chem. 165, 103–129 (1993).
Warmuth, R. o -Benzyne: strained alkyne or cumulene?—NMR characterization in a molecular container. Angew. Chem. Int. Edn Engl. 36, 1347–1350 (1997).
Timmerman, P., Verboom, W., van Veggel, F. C. J. M., van Duynhoven, J. P. M. & Reinhoudt, D. N. Anovel type of stereoisomerism in calix[4]arene-based carceplexes. Angew. Chem. Int. Edn Engl. 33, 2345–2348 (1994).
Rebek, J. J Assembly and encapsulation with self-complementary molecules. Chem. Soc. Rev. 255–264 (1996).
Kang, J. & Rebek, J. J Acceleration of a Diels–Alder reaction by a self-assembled molecular capsule. Nature 385, 50–52 (1997).
Cram, D. J., Choi, H.-J., Bryant, J. A. & Knobler, C. B. Solvophobic and entropic driving forces for forming velcraplexes, which are four-fold, lock–key dimers in organic media. J. Am. Chem. Soc. 114, 7748–7765 (1992).
Moran, J. R.et al. Vases and kites as cavitands. J. Am. Chem. Soc. 113, 5707–5714 (1991).
Rudkevich, D. M., Hilmersson, G. & Rebek, J. J Intramolecular hydrogen bonding controls the exchange rates of guests in a cavitand. J. Am. Chem. Soc. 119, 9911–9912 (1997).
Mohamadi, F.et al. MacroModel—an integrated software system for modeling organic and bioorganic molecules using molecular mechanisms. J. Comput. Chem. 11, 440–467 (1990).
Chapman, R. G. & Sherman, J. C. Study of templation and molecular encapsulation using highly stable and guest-selective self-assembling structures. J. Am. Chem. Soc. 117, 9081–9082 (1995).
Acknowledgements
We thank the Skaggs Research Foundation and the National Institutes of Health for financial support. T.H. is grateful to the Swiss National Science Foundation and the Ciba-Geigy-Jubiläums-Stiftung for the fellowships.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Heinz, T., Rudkevich, D. & Rebek, J. Pairwise selection of guests in a cylindrical molecular capsule of nanometre dimensions. Nature 394, 764–766 (1998). https://doi.org/10.1038/29501
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/29501
This article is cited by
-
Synthesis of covalent organic pillars as molecular nanotubes with precise length, diameter and chirality
Nature Synthesis (2023)
-
Creation of kinetically-controlled supramolecular systems based on coordination chemistry
Journal of Inclusion Phenomena and Macrocyclic Chemistry (2023)
-
Coordination cages as permanently porous ionic liquids
Nature Chemistry (2020)
-
Polyaromatic molecular peanuts
Nature Communications (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.