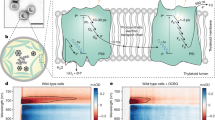
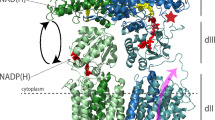
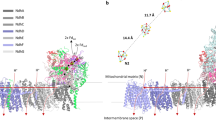
Abstract
Energy-transducing membranes of living organisms couple spontaneous to non-spontaneous processes through the intermediacy of protonmotive force (p.m.f.) — an imbalance in electrochemical potential of protons across the membrane. In most organisms, p.m.f. is generated by redox reactions that are either photochemically driven, such as those in photosynthetic reaction centres, or intrinsically spontaneous, such as those of oxidative phosphorylation in mitochondria. Transmembrane proteins (such as the cytochromes and complexes I, III and IV in the electron-transport chain in the inner mitochondrial membrane) couple the redox reactions to proton translocation, thereby conserving a fraction of the redox chemical potential as p.m.f. Many transducer proteins couple p.m.f. to the performance of biochemical work, such as biochemical synthesis and mechanical and transport processes. Recently, an artificial photosynthetic membrane was reported in which a photocyclic process was used to transport protons across a liposomal membrane, resulting in acidification of the liposome's internal volume1. If significant p.m.f. is generated in this system, then incorporating an appropriate transducer into the liposomal bilayer should make it possible to drive a non-spontaneous chemical process. Here we report the incorporation of FOF1-ATP synthase into liposomes containing the components of the proton-pumping photocycle. Irradiation of this artificial membrane with visible light results in the uncoupler- and inhibitor-sensitive synthesis of adenosine triphosphate (ATP) against an ATP chemical potential of ∼12 kcal mol−1, with a quantum yield of more than 7%. This system mimics the process by which photosynthetic bacteria convert light energy into ATP chemical potential.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Steinberg-Yfrach, G. et al. Conversion of light energy to proton potential in liposomes by artificial photosynthetic reactions centres. Nature 385, 239–241 (1997).
Gust, D., Moore, T. A. & Moore, A. L. Molecular mimicry of photosynthetic energy and electron transfer. Acc. Chem. Res. 26, 198–205 (1993).
Schmidt, G. & Gräber, P. The rate of ATP synthesis by reconstituted CF0F1liposomes. Biochim. Biophys. Acta 808, 46–51 (1985).
Sigalat, C., Pitard, B. & Haraux, F. Proton coupling is preserved in membrane-bound chloroplast ATPase activated by high concentrations of tentoxin. FEBS Lett. 368, 253–256 (1995).
Hangarter, R. P. & Good, N. E. Energy thresholds for ATP synthesis in chloroplasts. Biochim. Biophys. Acta 681, 397–404 (1982).
Richard, P. & Gräber, P. Kinetics of ATP synthesis catalyzed by the H+-ATPase from chloroplasts (CF0F1) reconstituted into liposomes and coreconstituted with bacteriorhodopsin. Eur. J. Biochem. 210, 287–291 (1992).
Richard, P., Pitard, B. & Rigaud, J.-L. ATP synthesis by the F0F1-ATPase from the thermophilic Bacillus PS3 co-reconstituted with bacteriorhodopsin into liposomes. J. Biol. Chem. 270, 21571–21578 (1995).
Fromme, P. & Gräber, P. Activatin/inactivation and uni-site catalysis by the reconstituted ATP-synthase from chloroplasts. Biochim. Biophys. Acta 1016, 29–42 (1990).
Girault, G., Berger, G., Galmiche, J.-M. & Andre, F. Characterization of six nucleotide-binding sites on chloroplast coupling factor 1 and one site on its purified β subunit. J. Biol. Chem. 263, 14690–14695 (1988).
Cladera, J. et al. Functional reconstitution of photosystem I reaction center from cyanobacterium Synechocystis sp PCC6803 into liposomes using a new reconstitution procedure. J. Bioenerg. Biomembr. 28, 503–515 (1996).
Pitard, B., Richard, P., Duñach, M. & Rigaud, J.-L. ATP synthesis by the F0F1ATP synthase from thermophilic Bacillus PS3 reconstituted into liposomes with bacteriorhodopsin. Eur. J. Biochem. 235, 779–788 (1996).
Richard, P., Rigaud, J.-L. & Gräber, P. Reconstitution of CF0F1into liposomes using a new reconstitution procedure. Eur. J. Biochem. 193, 921–925 (1990).
Petitou, M., Tuy, F. & Rosenfeld, C. Asimplified procedure for organic phosphorus determination from phospholipids. Anal. Biochem. 91, 350–353 (1978).
Acknowledgements
We thank F. Haraux for the gift of purified CF0F1, and both F. Haraux and W. Frasch for discussions. This work was supported by the US DOE.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Steinberg-Yfrach, G., Rigaud, JL., Durantini, E. et al. Light-driven production of ATP catalysed by F0F1-ATP synthase in an artificial photosynthetic membrane. Nature 392, 479–482 (1998). https://doi.org/10.1038/33116
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/33116
This article is cited by
-
Ratcheting synthesis
Nature Reviews Chemistry (2023)
-
Bioinspired photocatalytic systems towards compartmentalized artificial photosynthesis
Communications Chemistry (2023)
-
Controlled electron transfer by molecular wires embedded in ultrathin insulating membranes for driving redox catalysis
Photosynthesis Research (2023)
-
Enhanced photo-driven ion pump through silver nanoparticles decorated graphene oxide membranes
Nano Research (2022)
-
A self-organized synthetic morphogenic liposome responds with shape changes to local light cues
Nature Communications (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.