Abstract
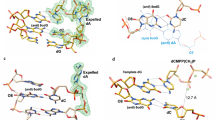
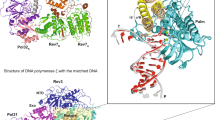
THE DNA polymerase from Thermus aquaticus (Taq polymerase) is homologous to Escherichia coli DNA polymerase I (Pol I) and likewise has domains responsible for DNA polymerase and 5′ nuclease activities1,2. The structures of the polymerase domains of Taq polymerase and of the Klenow fragment (KF) of Pol I are almost identical, whereas the structure of a vestigial editing 3′–5′ exonuclease domain of Taq polymerase that lies between the other two domains is dramatically altered, resulting in the absence of this activity in the thermostable enzyme2. The structures have been solved for editing complexes between KF and single-stranded DNA3,4 and for duplex DNA with a 3′ overhanging single strand5, but not for a complex containing duplex DNA at the polymerase active-site. Here we present the co-crystal structure of Taq polymerase with a blunt-ended duplex DNA bound to the polymerase active-site cleft; the DNA neither bends nor goes through the large polymerase cleft, and the structural form of the bound DNA is between the B and A forms. A wide minor groove allows access to protein side chains that hydrogen-bond to the N3 of purines and the O2 of pyrimidines at the blunt-end terminus. Part of the DNA bound to the polymerase site shares a common binding site with DNA bound to the exonuclease site, but they are translated relative to each other by several ångströms along their helix axes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Lawyer, F. C. et al. J. biol. Chem. 264, 6427–6437 (1989).
Kim, Y. et al. Nature 376, 612–616 (1995).
Freemont, P. S. et al. Proc. natn. Acad. Sci. U.S.A. 85, 8924–8928 (1988).
Beese, L. S. & Steitz, T. A. EMBO J. 10, 25–33 (1991).
Beese, L. S., Derbyshire, V. & Steitz, T. A. Science 260, 352–355 (1993).
Ollis, D. L., Brick, P., Hamlin, R., Xuong, N. G. & Steitz, T. A. Nature 313, 762–766 (1985).
Joyce, C. M. & Steitz, T. A. A. Rev. Biochem. 63, 777–822 (1994).
Pelletier, H. et al. Science 264, 1891–1903 (1994).
Steitz, T. A., Smerdon, S. J., Jaeger, J. & Joyce, C. M. Science 266, 2022–2025 (1994).
Cowtan, K. Joint CCP4 and ESF-EACBM Newsletter on Protein Crystallography 31, 34–38 (1994).
Steitz, T. A. Curr. Opin. struct. Biol. 3, 31–38 (1993).
Braithwaite, D. K. & Ito, J. Nucleic Acids Res. 21, 787–802 (1993).
Astatke, M., Grindley, N. D. F. & Joyce, C. M. J. molec. Biol. 270, 1945–1954 (1995).
Polesky, A. H., Steitz, T. A., Grindley, N. D. F. & Joyce, C. M. J. biol. Chem. 265, 14579–14591 (1990).
Polesky, A. H., Dahlberg, M. E., Benkovic, S. J., Grindley, N. F. D. & Joyce, C. M. J. biol. Chem. 267, 8417–8428 (1992).
Tabor, S. & Richardson, C. C. Proc. natn. Acad. Sci. U.S.A. 92, 6339–6343 (1995).
Conner, B. N. et al. Nature 295, 294–299 (1982).
Seeman, N. C., Rosenberg, J. M. & Rich, A. Proc. natn. Acad. Sci. U.S.A. 73, 804–808 (1976).
Steitz, T. A. et al. Cold Spring Harbor Symp. quant. Biol. 52, 465–471 (1987).
Steitz, T. A. in Biological Organization: Macromolecular Interactions at High Resolution 45–55 (Academic, New York, 1992).
Georgiadis, M. M. et al. Structure 3, 879–892 (1995).
Jacobo-Molina, A. et al. Proc. natn. Acad. Sci. U.S.A. 90, 6320–6324 (1993).
Kohlstaedt, L. A., Wang, J., Friedman, J. M., Rice, P. A. & Steitz, T. A. Science 256, 1783–1790 (1992).
Brutlag, D. & Kornberg, A. J. biol. Chem. 247, 241–248 (1972).
Joyce, C. M. J. biol. Chem. 264, 10858–10866 (1989).
Otwinowski, Z. in Data Collection and Processing (eds Sawyer, L., Isaacs, N. & Burley, S.) 56–62 (SERC Daresbury Laboratory, UK, 1993).
Brünger, A. T. X-PLOR version 3.1 (Yale Univ. Press, New Haven, CT, 1992).
Read, R. J. Acta crystallogr. A42, 140–149 (1986).
Lee, B. K. & Richards, F. M. J. molec. Biol. 55, 379–400 (1971).
Tabor, S., Huber, H. E. & Richardson, C. C. J. biol. Chem. 262, 16212–16223 (1987).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Eom, S., Wang, J. & Steitz, T. Structure of Taq polymerase with DNA at the polymerase active site. Nature 382, 278–281 (1996). https://doi.org/10.1038/382278a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/382278a0
This article is cited by
-
Thermostable enzyme research advances: a bibliometric analysis
Journal of Genetic Engineering and Biotechnology (2023)
-
Application of a modified tetra-primer ARMS–PCR assay for rapid Panax species identity authentication in ginseng products
Scientific Reports (2023)
-
Molecular evolution steered structural adaptations in the DNA polymerase III α subunit of halophilic bacterium Salinibacter ruber
Extremophiles (2023)
-
Customized multiple sequence alignment as an effective strategy to improve performance of Taq DNA polymerase
Applied Microbiology and Biotechnology (2023)
-
Probing conformational transitions towards mutagenic Watson–Crick-like G·T mismatches using off-resonance sugar carbon R1ρ relaxation dispersion
Journal of Biomolecular NMR (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.