Abstract
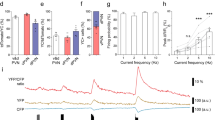
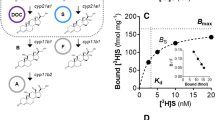
CORTICOTROPIN-RELEASING factor (CRF), a peptide first isolated from mammalian brain1, is critical in the regulation of the pituitary–adrenal axis, and in complementary stress-related endocrine, autonomic and behavioural responses2. Fish urotensin I and amphibian sauvagine were considered to be homologues3 of CRF until peptides even more closely related to CRF were identified in these same vertebrate classes4,5. We have characterized another mammalian member of the CRF family and have localized its urotensin-like immunoreactivity to, and cloned related complementary DNAs from, a discrete rat midbrain region. The deduced protein encodes a peptide that we name urocortin, which is related to urotensin (63% sequence identity) and CRF (45% sequence identity). Synthetic urocortin evokes secretion of adrenocortico-tropic hormone (ACTH) both in vitro and in vivo and binds and activates transfected type-1 CRF receptors6–9, the subtype expressed by pituitary corticotropes. The coincidence of urotensin-like immunoreactivity with type-2 CRF receptors10–13 in brain, and our observation that urocortin is more potent than CRF at binding and activating type-2 CRF receptors, as well as at inducing c-Fos (an index of cellular activation) in regions enriched in type-2 CRF receptors, indicate that this new peptide could be an endogenous ligand for type-2 CRF receptors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Vale, W., Spiess, J., Rivier, C. & Rivier, J. Science 213, 1394–1397 (1981).
Vale, W. et al. Chemical and Biological Characterization of Corticotropin Releasing Factor in Recent Progress in Hormone Research, Vol. 39, 245–270 (Academic, San Diego, 1983).
Lederis, K., Fryer, J. N., Okawara, Y., Schonrock, C. & Richter, D. in Fish Physiology (ed. Farrell, A.) 67–100 (Academic, San Diego, 1994).
Okawara, Y. et al. Proc. natn. Acad. Sci. U.S.A. 85, 8439–8443 (1988).
Stenzel-Poore, M. P., Heldwein, K. A., Stenzel, P., Lee, S. & Vale, W. W. Molec. Endocr. 6, 1716–1724 (1992).
Chen, R., Lewis, K. A., Perrin, M. H. & Vale, W. W. Proc. natn. Acad. Sci. U.S.A. 90, 8967–8971 (1993).
Perrin, M. H., Donaldson, C. J., Chen, R., Lewis, K. A. & Vale, W. W. Endocrinology 133, 3058–3061 (1993).
Chang, C., Pearse, R., O'Connell, S. & Rosenfeld, M. G. Neuron 11, 1187–1195 (1993).
Vita, N. et al. FEBS Lett. 335, 1–5 (1993).
Lovenberg, T. W. et al. Proc. natn. Acad. Sci. U.S.A. 92, 836–840 (1995).
Kishimoto, T., Pearse, R. V. II, Lin, C. R. & Rosenfeld, M. G. Proc. natn. Acad: Sci. U.S.A. 92, 1108–1112 (1995).
Perrin, M. et al. Proc. natn. Acad. Sci. U.S.A. 92, 2969–2973 (1995).
Stenzel, P. et al. Molec. Endocr. 9, 637–645 (1995).
Imaki, J., Imaki, T., Vale, W. & Sawchenko, P. E. Brain Res. 547, 28–36 (1991).
Lovenberg, T. W., Chalmers, D. T., Liu, C. & De Souza, E. B. Endocrinology 136, 3351–3355 (1995).
Potter, E. et al. Proc. natn. Acad. Sci. U.S.A. 91, 8777–8781 (1994).
Ishida, I., Ichikawa, T., & Deguchi, T. Proc. natn. Acad. Sci. U.S.A. 83, 803–812 (1986).
Thompson, R. C., Seasholtz, A. F. & Herbert, E. Molec. Endocr. 1, 363–370 (1987).
Potter, E. et al. Proc. natn. Acad. Sci. U.S.A. 89, 4192–4196 (1992).
Muglia, L. Jacobson, L., Dikkes, P. & Majzoub, J. A. Nature 373, 427–432 (1995).
Loewy, A. D. in Central Regulation of Autonomic Functions (eds Loewy, A. D. & Spyer, K. M.) 268–285 (Oxford Univ. Press, New York. 1990).
Loewy, A. D., Saper, C. B. & Yamodis, N. D. Brain Res. 141, 153–159 (1978).
Webster, W. R. in The Rat Nervous System (ed. Paxinos, G.) 797–831 (Academic, San Diego. 1995).
Saper, C. B., Swanson, L. W. & Cowan, W. M. J. comp. Neuro. 183, 689–706 (1979).
Morgan, J. I. & Curran, T. A. Rev. Neurosci. 14, 421–451 (1991).
Lederis, K. et al. Rec. Prog. Horm. Res. 41, 553–576 (1985).
Vale, W. et al. Meth. Enzym. 103, 565–577 (1983).
Sutton, S. W. et al. Endocrinology 136, 1097–1102 (1995).
Chung, R. Y., Mason, P., Strassman, A., Maciewicz, R. Neurosci. Lett. 83, 13–19 (1987).
Chan, R. K. W., Brown, E. R., Ericsson, A., Kovács, K. J. & Sawchenko, P. E. J. Neurosci. 13, 5125–5138 (1993).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Vaughan, J., Donaldson, C., Bittencourt, J. et al. Urocortin, a mammalian neuropeptide related to fish urotensin I and to corticotropin-releasing factor. Nature 378, 287–292 (1995). https://doi.org/10.1038/378287a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/378287a0
This article is cited by
-
Neurodegeneration in the centrally-projecting Edinger–Westphal nucleus contributes to the non-motor symptoms of Parkinson’s disease in the rat
Journal of Neuroinflammation (2022)
-
Cancer-Specific Loss of Urocortin 3 in Human Renal Cancer
Advances in Therapy (2020)
-
Urocortins in the mammalian endocrine system
Acta Veterinaria Scandinavica (2019)
-
Urocortins and their unfolding role in mammalian social behavior
Cell and Tissue Research (2019)
-
Cardiovascular Effects of Urocortin-2: Pathophysiological Mechanisms and Therapeutic Potential
Cardiovascular Drugs and Therapy (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.