Abstract
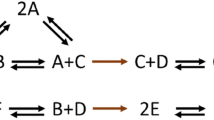
In a biochemical kinetic diagram1, the various discrete states of an enzyme or enzyme complex are represented by points, and interstate transitions are represented by lines. Figure 1 is an example. I show here that, for steady states near equilibrium, the phenomenological Onsager coefficients in the linear flux–force equations that are a consequence of any such diagram have a transparent physical interpretation: these coefficients are simple combinations of one-way cycle fluxes1 at equilibrium. The existence of this kind of relationship was pointed out recently in an appendix of a paper2 on another subject. I present here the explicit connection between the phenomenological coefficients and the one-way cycle fluxes of the diagram. These coefficients are considered by many scientists to be strictly empirical and incomprehensible. My report provides a simple interpretation of them, at the molecular level, for any biochemical kinetic diagram. This is analogous to the molecular interpretation of phenomenological equilibrium thermodynamics that is provided by statistical mechanics.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Hill, T. L. Free Energy Transduction in Biology (Academic, New York, 1977).
Hill, T. L. J. chem. Phys. 76, 1122 (1982).
Haase, R. Thermodynamics of Irreversible Processes, 121–133 (Addison-Wesley, Reading, Massachusetts, 1969).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hill, T. The linear Onsager coefficients for biochemical kinetic diagrams as equilibrium one-way cycle fluxes. Nature 299, 84–86 (1982). https://doi.org/10.1038/299084a0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/299084a0
This article is cited by
-
Bivectorial Nonequilibrium Thermodynamics: Cycle Affinity, Vorticity Potential, and Onsager’s Principle
Journal of Statistical Physics (2021)
-
Kinetic asymmetry allows macromolecular catalysts to drive an information ratchet
Nature Communications (2019)
-
Kinetic and mesoscopic non-equilibrium description of the Ca2+ pump: a comparison
European Biophysics Journal (2012)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.