Abstract
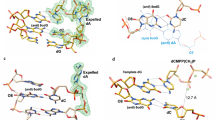
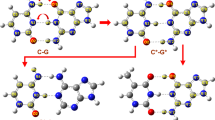
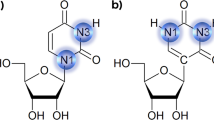
RECENT crystallographic studies of the dinucleosides ApU (ref. 1) and GpC (ref. 2) have given experimental proof for the base pairing arrangement proposed by Watson and Crick for the DNA double helix3. Another striking feature of this structure relates to the torsional angle about the C5′-C4′ bond in the phosphate–sugar backbone chain. In the Crick and Watson model4, this conformation is gauche–trans (GT). Crystal structures of 5′-nucleotides, dinucleosides and dinucleotides so far studied, however, have shown only the gauche–gauche (GG) conformation about this bond. The GG conformer is also the only one found in the refined models of the proposed structure of the double helical nucleic acids and polynucleotides5–7. The only nucleotide with a GT conformation is 6-azauridine-5′-phosphate8 which is not a normal monomer unit of nucleic acids. It is also reported that 5′-dGMP assumes preferentially GT conformation in solution9.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Rosenberg, J. M., Seeman, N. C., Kim, J. J. P., Suddath, F. L., Nicholas, H. B., and Rich, A., Nature, 243, 150 (1973).
Day, R. O., Seeman, N. C., Rosenberg, J. M., and Rich, A., Proc. natn. Acad. Sci. U.S.A., 70, 849 (1973).
Watson, J. D., and Crick, F. H. C., Nature, 171, 737 (1953).
Crick, F. H. C., and Watson, J. D., Proc. R. Soc., A 223, 80 (1954).
Arnott, S., Dover, S. D., and Wonacott, A. J., Acta Cryst., B 25, 2192 (1969).
Arnott, S., Prog. Biophys. molec. Biol., 21, 265 (1970).
Arnott, S., Wilkins, M. H. F., Fuller, W., and Langridge, R., J. molec. Biol., 27, 535 (1967).
Saenger, W., and Suck, D., Nature, 242, 610 (1973).
Sarma, R. H., Mynott, R. J., Wood, D. J., and Hruska, F. W., J. Am. Chem. Soc., 95, 6457 (1973).
Seshadri, T. P., and Viswamitra, M. A., Curr. Sci, 43, 111 (1974).
Germain, G., and Woolfson, M. M., Acta Cryst., B 24, 91 (1968).
Viswamitra, M. A., Reddy, S., Lin, B. G. H-.Y., and Sundaralingam, M., J. Am. Chem. Soc., 93, 4565 (1971).
Reddy, S. B., and Viswamitra, M. A., Cryst. Struct. Comm., 2, 9 (1973).
Trueblood, K. N., Horn, P., and Luzzati, V., Acta Cryst., 14, 965 (1961).
Sundaralingam, M., Jerusalem Symp. Quant. Chem. Biochem., V, 417 (1973).
Young, D. W., Tollin, P., Wilson, H. R., Nature, 248, 513 (1974).
Young, D. W., Tollin, P., Wilson, H. R., Acta Cryst., B 30, 2012 (1974).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
VISWAMITRA, M., SESHADRI, T. Molecular and crystal structure of deoxyguanosine 5′-phosphate. Nature 252, 176–177 (1974). https://doi.org/10.1038/252176a0
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1038/252176a0
This article is cited by
-
Antibodies to guanylic acid: Fractionation and specificities
Bioscience Reports (1984)
-
Is 3′-nucleotide rigid?
Nature (1980)
-
Structure of thymidylyl-3′, 5′-deoxyadenosine
Nature (1976)
-
Molecular structures of cytidine-5′-diphosphate and cytidine-5′-diphospho-choline, and their role in intermediary metabolism
Nature (1975)
-
An uncommon nucleotide conformation shown by molecular structure of deoxyuridine-5′-phosphate and nucleic acid stereochemistry
Nature (1975)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.