Abstract
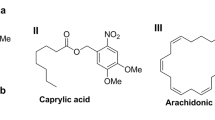
Two recent reviews of peroxidation of organic compounds1,2 cite the fact that α,β-unsaturated ketones are oxidized by peracids to enol esters. A priori, however, one might expect one or more of three possible products and their hydrolysates.  Thus, I and II would result from an attack of peracid at the carbonyl carbon followed by rearrangement according to the mechanism proposed by Criegee3, Friess4, and Doering5, while III Would be the consequence of an attack at the β-carbon of the conjugated system.
Thus, I and II would result from an attack of peracid at the carbonyl carbon followed by rearrangement according to the mechanism proposed by Criegee3, Friess4, and Doering5, while III Would be the consequence of an attack at the β-carbon of the conjugated system.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Swern, Chem. Rev., 45, 1 (1949).
Criegee, Fortschr. Chem. Forsch., 1, 508 (1950).
Criegee, Ann., 560, 127 (1948).
Friess, J. Amer. Chem. Soc., 71, 2571 (1949).
Doering and Speers, J. Amer. Chem. Soc., 72, 5515 (1950).
Böeseken and Kremer, Rec. trav. chim., 50, 827 (1931).
Böeseken and Soesman, Rec. trav. chim., 52, 874 (1933).
Böeseken and Jacobs, Rec. trav. chim., 55, 786 (1936).
Julian, Meyer and Ryden, J. Amer. Chem. Soc., 72, 367 (1950).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
WENKERT, E., RUBIN, M. Peroxidation of Unsaturated Ketones. Nature 170, 708–709 (1952). https://doi.org/10.1038/170708b0
Issue Date:
DOI: https://doi.org/10.1038/170708b0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.