Abstract
Large B-cell lymphomas with IGH@BCL2 and MYC rearrangement, known as double-hit lymphoma (DHL), are clinically aggressive neoplasms with a poor prognosis. Some large B-cell lymphomas have concurrent abnormalities of MYC and BCL2 other than coexistent translocations. Little is known about patients with these lymphomas designated here as atypical DHL. We studied 40 patients of atypical DHL including 21 men and 19 women, with a median age of 60 years. Nine (23%) patients had a history of B-cell non-Hodgkin lymphoma. There were 30 diffuse large B-cell lymphoma (DLBCL), 7 B-cell lymphoma, unclassifiable, with features intermediate between DLBCL and Burkitt lymphoma, and 3 DLBCL with coexistent follicular lymphoma. CD10, BCL2, and MYC were expressed in 28/39 (72%), 33/35 (94%), and 14/20 (70%) cases, respectively. Patients were treated with standard (n=14) or more aggressive chemotherapy regimens (n=17). We compared the atypical DHL group with 76 patients with DHLand 35 patients with DLBCL lacking MYC and BCL2 abnormalities. The clinicopathologic features and therapies were similar between patients with atypical and typical DHL. The overall survival of patients with atypical double-hit lymphoma was similar to that of patients with double-hit lymphoma (P=0.47) and significantly worse than that of patients with DLBCL with normal MYC and BCL2 (P=0.02). There were some minor differences. Cases of atypical double-hit lymphoma more often have DLBCL morphology (P<0.01), less frequently expressed CD10 (P<0.01), and patients less often had an elevated serum lactate dehydrogenase level (P=0.01). In aggregate, these results support expanding the category of MYC/BCL2 DHL to include large B-cell lymphomas with coexistent MYC and BCL2 abnormalities other than concurrent translocations.
Similar content being viewed by others
Main
Double-hit lymphoma (DHL) is broadly defined as an aggressive B-cell lymphoma with a MYC translocation combined with an additional translocation involving other genes, such as BCL2, BCL3, BCL6, or CCND1.1 The most common type of double-hit lymphoma carries MYC/8q24 rearrangement and IGH@BCL2/t(14;18)(q32;q21), commonly referred to in the literature as double-hit B-cell lymphoma, MYC/BCL2, or BCL2+/MYC+ DHL. MYC, an oncogene located at 8q24, functions as a transcription factor controlling the expression of many genes, directly or indirectly, and is essential for many cellular physiologic functions.2, 3 BCL2, located at 18q21, encodes a protein that inhibits apoptosis and also is involved in physiological DNA repair. The cooperating effects of MYC and BCL2 in lymphomagenesis have been shown in many studies.4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14
Numerous case series of MYC/BCL2 double-hit lymphoma are reported in the literature.15, 16, 17, 18, 19, 20, 21, 22, 23, 24 MYC/BCL2 DHLs represent ∼10% of all lymphomas that otherwise resemble diffuse large B-cell lymphoma (DLBCL).10 Although different studies have focused on different aspects or details of double-hit lymphoma, all studies suggest that patients with these lymphomas commonly have an aggressive clinical course characterized by advanced stage disease, extranodal involvement, and high serum lactate dehydrogenase levels. Central nervous system and bone marrow involvement are common and the International Prognostic Index score (IPI score) is often high-intermediate or high. Most cases of double-hit lymphoma have morphologic features of DLBCL or B-cell lymphoma, unclassifiable, with features intermediate between DLBCL and Burkitt lymphoma. Virtually, all cases have a germinal center B-cell immunophenotype and a high proliferation rate. Despite a variety of therapeutic approaches that have been used to date, patients with DHL have a poor prognosis with a median overall survival (OS) of <2 years.
In daily practice we also have encountered cases of large B-cell lymphoma in which concurrent MYC and BCL2 abnormalities are present, but translocations involving MYC, BCL2, or both genes are not identified. These cases can be grouped into three types: (1) MYC translocation coexisting with extra copies of BCL2, but no evidence of t(14;18)(q32;q21)/IGH@BCL2; (2) t(14;18)(q32;q21)/IGH@BCL2 coexisting with extra copies of MYC, but without evidence of MYC translocation; and (3) extra copies of MYC and BCL2 without translocations involving either gene. For the purpose of this study, we have designated these tumors as atypical DHL in contrast with conventionally defined (hereafter referred to as typical) double-hit lymphoma.
Rarely, single or very few atypical DHL cases have been included in MYC/BCL2 DHL case series that have been reported.15, 20, 22, 23 To our knowledge, however, little is known about atypical DHL and there is no large study in the literature that has focused on atypical MYC/BCL2 double-hit lymphoma. In this study, we assess the clinicopathologic, immunophenotypic, and cytogenetic features of a large group of patients with atypical double-hit lymphoma, and compare this patient group to a group of patients who had typical double-hit lymphoma as well as a group of patients who had DLBCL not associated with either MYC or t(14;18)(q32;q21)/IGH@BCL2.
Materials and methods
Case Selection
A total of 151 cases of large B-cell lymphoma with MYC and BCL2 status tested by fluorescence in situ hybridization (FISH) are included in this study. These cases are derived from two institutions spanning the time interval of 1 January 2006 through 31 December 2013. Most of these cases were recognized in recent years after large B-cell lymphomas were screened routinely for MYC and BCL2 rearrangements by FISH testing.
The cases in this study were divided into three categories: typical double-hit lymphoma, atypical double-hit lymphoma, and DLBCL without MYC and BCL2 abnormalities as shown by FISH. Typical double-hit lymphoma cases included all cases of large B-cell lymphoma that met the classical definition of MYC/BCL2 double-hit lymphoma, namely rearrangements of both MYC and BCL2. Atypical double-hit lymphoma cases included all tumors with concurrent MYC and BCL2 abnormalities other than coexistent translocations and were further subcategorized into three groups: (1) MYC translocation coexisting with extra copies of BCL2, but lacking evidence of t(14;18)(q32;q21)/IGH@BCL2; (2) t(14;18)(q32;q21)/IGH@BCL2 coexisting with extra copies of MYC, but without evidence of MYC translocation; and (3) extra copies of MYC and BCL2 without translocations involving either gene. A total of 40 patients with atypical double-hit lymphoma were identified as the study group. These patients and their tumors were compared with 76 cases of typical double-hit lymphoma and 35 cases of DLBCL with no MYC and BCL2 abnormalities.
Corresponding medical records were reviewed to obtain clinical information, including a history of lymphoma, number and sites of involvement, Ann Arbor stage, IPI score, treatment regimens, response to therapy, and OS. Morphologic, immunophenotypic, and cytogenetic data were reviewed. All cases were classified according to the current World Health Organization (WHO) scheme, which includes the provisional category B-cell lymphoma, unclassifiable, with features intermediate between DLBCL and Burkitt lymphoma.25
Immunophenotypic Methods
Immunohistochemical studies were performed using formalin-fixed, paraffin-embedded tissue sections either at the time of diagnosis or retrospectively for this study. The panel of monoclonal antibodies included reagents specific for CD3, CD20 (Ventana Medical Systems, Tucson, AZ, USA); CD5, CD10, BCL2, BCL6, MUM1 (Leica Microsystems, Buffalo Grove, IL, USA); Ki-67 (MIB-1) (DAKO, Carpinteria, CA, USA); and MYC (Epitomics, Burlingame, CA, USA). The cutoffs for positivity for CD10 and BCL6 were both 30% and the cutoff for MUM1 was 60% as have been used in an earlier study.26 The cutoffs for a positive MYC or BCL2 results were ≥40% or ≥50% of cells, respectively, as used by others.27, 28
Flow cytometry immunophenotypic analysis was performed using standard multicolor analysis, mostly eight color. Analysis was performed using either a FACScanto II or FACSCalibur cytometer (Becton-Dickinson Biosciences, San Jose, CA, USA) as described previously.20 Lymphocytes were gated for analysis using side scatter versus forward scatter, and CD45 expression versus side scatter. The panel of antibodies included CD3, CD4, CD5, CD7, CD8, CD10, CD19, CD20, CD23, CD33, CD38, and immunoglobulin κ-and λ-light chains. All antibodies were obtained from Becton-Dickinson Biosciences.
Conventional Cytogenetics and FISH
Conventional G-band karyotype analysis was performed on five cases using cell suspensions of lymph node, extranodal tissue biopsy, or bone marrow aspirate specimens. The karyotypes were reported according to the 2013 International System for Human Cytogenetic Nomenclature.29
FISH analysis was performed on all cases using LSI MYC dual-color break-apart and LSI IGH@BCL2 dual-color, dual fusion probes (Abbott Laboratories, Des Plaines, IL, USA). For bone marrow aspirate specimens, FISH was performed by using a freshly dropped slide from a harvested bone marrow or a G-banded slide for metaphase mapping according to the manufacturer’s instructions. For formalin-fixed, paraffin-embedded tissue samples, FISH was performed on 4-μm-thick tissue sections and fixed onto slides according to the manufacturer’s protocol. The signals from 200 nuclei were analyzed.
The cutoffs for considering a tumor sample positive for MYC and BCL2 by FISH were generated by assessing 20 normal controls and choosing cutoffs two s.d. above the mean. These cutoffs were different in bone marrow smears versus fixed, paraffin-embedded tissue sections and were also slightly different between the laboratories of the two institutions. However, these cutoffs were low (all≤5%) and all case in this study designated as positive for MYC or BCL2 had these abnormalities in >25% of nuclei assessed.
Statistical Analysis
OS was calculated from the date of diagnosis to the date of death or last follow-up. Patient survival was analyzed using the Kaplan–Meier method and compared using the log rank test (GraphPad Prism 5 software). The Fisher exact test was used to compare differences between the atypical and typical double-hit lymphoma groups. A P-value of <0.05 was considered statistically significant.
Results
Atypical MYC/BCL2 Double-Hit Lymphoma
Clinical characteristics
We identified 40 patients with atypical DHL, 39 adults and 1 child. There were 21 men and 19 women with a median age of 60 years (range, 6–82 years). Twenty-nine (73%) patients presented with de novo disease, nine (23%) patients had a history or concurrent low-grade follicular lymphoma or other B-cell non-Hodgkin lymphoma, and two patients did not have a detailed history available for review. Serum LDH results were available for 29 patients, and 16 (55%) patients had an elevated level. Two patients were initially diagnosed by examination of bone marrow specimens, whereas all others were initially diagnosed in lymph node or other tissue site biopsy specimens. Fourteen of 27 (52%) patients had 2 or more extranodal sites of disease. The involved extranodal sites included the bone marrow, central nervous system, abdomen, gastrointestinal tract (small intestine or colon), liver, spleen, lung, pleural fluid, testis, ovaries, skin, and thyroid gland. Bone marrow involvement was present in 8 of 20 (40%) patients. Only 11 patients underwent spinal tap and cerebrospinal fluid analysis and 1 was involved by lymphoma. Twenty-two of 31 (76%) patients had advanced Ann Arbor stage (III/IV) disease, and 18 of 30 (60%) patients had a high-intermediate or high IPI score (Table 1).
Morphological and immunophenotypic features
The atypical DHL cases were classified as follows: 30 (75%) DLBCL, 7 (18%) B-cell lymphoma, unclassifiable, with features intermediate between DLBCL and Burkitt lymphoma, and 3 (8%) follicular lymphoma associated with DLBCL. The grade of the follicular lymphoma component in these cases was grade 1, grade 3A, and grade 3B in 1 case each (Table 2, Figures 1 and 2).
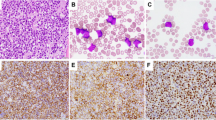
An atypical MYC/BCL2 double-hit lymphoma of B-cell lymphoma, unclassifiable, with features intermediate between diffuse large B-cell lymphoma and Burkitt lymphoma. (a) This lymphoma is positive for both MYC (70%), (b and c) BCL2 (100%), and has a Ki67 (d) proliferation rate of ∼90% (a–d: × 400); FISH showed BCL2 rearrangement (e) and extra copies of MYC (f).
All atypical DHLs were of B-cell lineage, positive for one or more pan-B-cell antigens and negative for pan-T-cell antigens. CD10 was positive in 28 of 39 (72%), BCL6 in 25 of 27 (93%), MUM1 in 5 of 16 (31%, all negative for CD10), and BCL2 in 33 of 35 (95%) cases. MYC expression was observed in 14 of 20 (70%) cases by using a 40% cutoff,28, 30 or 17 of 20 (85%) cases by using 10% as cutoff as reported by a recent study.31 On the basis of these results and by using the Hans algorithm,32 the 30 cases of DLBCL can be further subclassified as: 21 germinal center B-cell (GCB) type, 5 non-GCB type, and 4 unclassifiable due to incomplete data. The Ki67 proliferation rate in these lymphomas was variable, ranging from 20% up to nearly 100% (Table 2, Figures 1 and 2), with 28 of 32 (88%) cases having a proliferation rate >70%. Eleven of 20 (55%) cases were positive for both MYC and BCL2 by immunohistochemistry.
Cytogenetic characteristics
Conventional cytogenetic analysis was performed on five atypical DHLs. Each of these lymphomas had a complex karyotype with >3 numerical and/or structural aberrations (Table 3). Three cases had t(14;18)(q32;q21) and two cases had t(8;14)(q24;q32). No cases carried both translocations. FISH analysis showed 3 cases with MYC translocation and extra copies of BCL2, 22 cases with t(14;18)/IGH@BCL2 coexisting with extra copies of MYC, and 15 cases having extra copies of both MYC and BCL2, but no translocations involving either gene. The MYC copy number ranged from 3 to 8 and BCL2 copy number ranged from 3 to 7. In all three cases with MYC translocation, BCL6 was assessed by FISH and no case had a BCL6 translocation.
Treatment
Detailed therapy data were available for 31 of 40 patients with atypical DHL. All 31 patients were treated with combination chemotherapy (Table 4): 14 (45%) with rituximab, cyclophosphamide, daunorubicin, vincristine, and prednisone (R-CHOP), and 17 (55%) with more aggressive regimens. Seven of 17 patients received rituximab plus hyper-fractionated cyclophosphamide, vincristine, doxorubicin, dexamethasone alternating (R-hyper-CVAD), whereas 10 patients received rituximab plus one of the following regimens: EPOCH (etoposide, prednisone, vincristine, cyclophosphamide, and doxorubicin), ESHAP (etoposide, methylprednisolone (solumedrol), high-dose cytarabine (ara-C) and cisplatin), or RICE (rituximab, ifosfamide, carboplatin, and etoposide). Overall, there was no statistical significant difference in OS between patients who were treated with R-CHOP versus those treated with R-hyper-CVAD or other regimens (P=0.67). Five of 31 patients also received hematopoietic stem cell transplant, including 4 autologous and 1 allogeneic transplant.
Survival of patients with atypical double-hit lymphoma
Patients with tumors that had MYC amplification (defined as copy number ≥533, 34) were compared with tumors that had three to four copies of MYC, and there is no statistically significant difference in their OS (P=0.54). Similarly, patients with tumors that had BCL2 amplification versus lower copy number showed no statistically significant difference of OS (P=0.47). The OS of patients with the three subtypes of atypical double-hit lymphoma defined by FISH were further analyzed and there was no statistically significant difference in OS (P=0.27). Although the numbers for patients with each type of atypical double-hit lymphoma were small, based on these results we combined all patients with atypical double-hit lymphoma into a single group for comparison with patients with typical double-hit lymphoma and patients with DLBCL.
The three patients with atypical DHL in which MYC translocation was identified had poor survival. Although this is a small patient subset, the OS of these patients appeared to be similar to patients with typical DHL (P=0.70). In patients with atypical DHL in which extra copies of MYC were identified, the OS of patients in which their tumor carried a BCL2 translocation was not statistically significantly different from that of patients with tumors associated with extra copies of BCL2 (P=0.21). These data suggest that non-translocation BCL2 abnormalities have a negative impact on prognosis similar to BCL2 translocation in the setting of MYC cytogenetic abnormalities (translocation or extra copies).
Atypical versus Typical MYC/BCL2 DHL
The clinical, morphologic, immunophenotypic, and cytogenetic features, including proliferation rate and karyotypic complexity of patients with atypical DHL (n=40) were very similar to those of patients with typical DHL (n=76) (Tables 1, 2, 3). The few statistically significant differences were as follows: atypical double-hit lymphoma tumors more often had DLBCL morphology (P<0.01), were less often CD10 positive (P<0.01), and patients less often had an elevated serum LDH level (P=0.01). Comparison of therapies between the two groups revealed no statistically significant differences (P=0.11, Table 4). The OS of patients with atypical DHL was similar to that of patients with typical double-hit lymphoma (Figure 3a), with a 2-year OS rate of 54% in atypical double-hit lymphoma and 49% in typical double-hit lymphoma patients, respectively (P=0.47). Excluding patients with a history of low-grade B-cell lymphoma, there was no difference in OS between patients with de novo atypical double-hit lymphoma versus patients with typical double-hit lymphoma (Figures 3b, P=0.26).
Comparison of overall survival (OS). There is no statistically significant difference in OS between atypical MYC/BCL2 double-hit lymphoma (n=37) and typical MYC/BCL2 double-hit lymphoma (n=74) either including (a) or excluding transformed lymphomas (b); however, atypical double-hit lymphoma has a significant worse OS compared with diffuse large B-cell lymphoma without MYC and BCL2 FISH abnormalities (MYC/BCL2 NL) (c), similar to typical double-hit lymphoma (d).
We also compared patients with atypical double-hit lymphoma (n=40) to patients with DLBCL in which there was no evidence of MYC or BCL2 abnormalities shown by FISH (n=35). The DLBCL patient group showed similar ages and gender distribution (Table 5) and received similar therapies when compared with patients with atypical DHL. However, patients with atypical DHL more frequently presented with high stage disease, extranodal involvement, and high IPI score when compared with patients with DLBCL lacking MYC and BCL2 abnormalities (Tables 5, P<0.05). Especially, the OS of patients with atypical DHL was significantly worse than the survival of DLBCL patients (Figures 3c and d, P<0.05).
Discussion
Patients with large B-cell lymphoma with concurrent IGH@BCL2 and MYC rearrangements, known as MYC/BCL2 double-hit lymphoma (and referred to here as typical double-hit lymphoma), are known to have aggressive clinical course with a poor prognosis. It is also known that some cases of clinically aggressive large B-cell lymphoma have concurrent abnormalities of MYC and BCL2, but do not have concurrent MYC and BCL2 translocations (designated here as atypical DHL). The clinicopathologic features and prognostic implications of atypical double-hit lymphoma are not well understood and are the focus of this study.
We identified 40 patients with atypical DHL and compared them with 76 patients with typical DHL and 35 patients with DLBCL without MYC or BCL2 abnormalities. Overall, the clinical and pathologic features of patients with atypical and typical DHL were very similar with few statistically significant differences present. From a clinical perspective, there was only one difference; patients with atypical DHL less often had an elevated serum LDH level (P=0.01). From the pathologic perspective, atypical double-hit lymphoma cases more often showed DLBCL morphology and had a lower frequency of CD10 expression. The OS of patients with atypical DHL was poor and very similar to the OS of patients with typical double-hit lymphoma. Patients with atypical double-hit lymphoma had more aggressive clinical features and a significantly worse OS than patients with DLBCL without MYC and BCL2 abnormalities. In aggregate, these data suggest that atypical double-hit lymphoma is sufficiently distinct from DLBCL and more similar to typical double-hit lymphoma.
In the 2008 WHO classification, lymphoma cases of typical double-hit lymphoma were recognized and thought to commonly fit within the provisional disease category of B-cell lymphoma, unclassifiable, with features intermediate between DLBCL and Burkitt lymphoma. This does not appear to be the case for atypical double-hit lymphomas, however, which in this study more often had DLBCL morphology. Another apparent difference is the cell-of-origin classification. Nearly all typical DHLs have a germinal center B-cell origin and carry t(14;18)/IGH@BCL2. In contrast, a subset of atypical double-hit lymphomas lacks IGH@BCL2 and does not express CD10 (P<0.01).
It seems reasonable to hypothesize that cases of atypical DHL are a manifestation of the cooperative effects of MYC and BCL2 overexpression, and therefore affected patients have clinical and pathologic features and a poor prognosis similar to patients with typical double-hit lymphoma. Chromosomal translocations that involve MYC/8q24 and BCL2/18q21 are thought to result in upregulation of MYC and BCL2 expression. However, MYC and BCL2 can theoretically be activated through mechanisms other than translocation, and copy number increase is one possible mechanism. Data showing that DLBCL patients with MYC and BCL2 coexpression shown by immunohistochemistry have a poor prognosis in the absence of concurrent MYC and BCL2 translocations indirectly supports this hypothesis.28, 30, 34, 35, 36 In this study, similar proportions of typical and atypical DHLs showed MYC and BCL2 coexpression shown by immunohistochemistry, also supporting the above hypothesis. Of interest, not all cases of typical or atypical DHL showed MYC and BCL2 overexpression. Therefore, MYC or BCL2 abnormalities in atypical double-hit lymphoma do not always result in overexpression detectable by immunohistochemistry. In the literature, this observation also has been made in a subset of typical double-hit lymphoma. The pathogenesis of those cases needs further study.
Others have described cases of B-cell lymphoma with extra signals of MYC or BCL2 identified by FISH. Yoon et al37 studied 154 DLBCL cases. They reported 14 patients who had tumors with MYC rearrangement and 11 patients who had tumors with increased MYC copy number. Patients with tumors characterized by extra MYC copy number had a poorer prognosis, similar to patients with MYC rearrangement, and poorer than patients with DLBCL without MYC abnormalities. Mossafa et al33 studied 344 cases of non-Hodgkin lymphoma and identified 16 cases with multiple copies of MYC but no rearrangement. These 16 patients had a very poor prognosis: 9 patients died during a median follow-up of 11 months, although these include cases of different types of lymphoma. Testoni et al,36 in a series of 166 cases of DLBCL treated with R-CHOP, identified 17 with extra copies of MYC (all ≤4 copies) but no MYC translocations. All of these cases showed high MYC mRNA expression by gene expression profiling analysis. However, only patients with concomitant del(8p) had a poorer overall and progression-free survival. These findings are similar to and further support the results in this study. A recent study has suggested that MYC expression, translocation, or amplification (>4 copies) is associated with poorer prognosis in DLBCL patients, whereas tumors with three to four copies of MYC do not have a poorer prognosis.31 However, this study included only three cases with MYC amplification (>4 copies). Our results suggest that patients with atypical double-hit lymphoma that had three to four copies of MYC showed similar OS to patients with atypical DHL with more than four copies of MYC.
In the study by Yoon et al,37 increased BCL2 copy number was identified in 17 of 145 (12%) cases and was associated with worse prognosis. In the 20 cases of MYC/BCL2 double-hit lymphoma reported by Snuderl et al,15 one patient had MYC rearrangement with extra copies of BCL2 and had a poor prognosis. Jiang et al38 studied 60 cases of DLBCL and identified 18 cases with BCL2 gene amplification. All cases with BCL2 amplification demonstrated overexpression of BCL2 and were associated with a worse prognosis. Extra BCL2 copies were more commonly seen in the non-GCB type of DLBCL37, 39 and usually associated with overexpression of BCL2,40 which has been correlated with poorer prognosis in DLBCL in some studies.31 Overall, these studies directly or indirectly support the hypothesis that extra copies of MYC and BCL2 likely result in MYC and BCL2 overexpression in most cases and confer a poorer prognosis.
This study also has limitations. Our study group is relatively small compared with many studies on large B-cell lymphoma in the literature, although our study group is similar in number to most studies published on double hit-lymphoma. The limited number of cases in each of the three subtypes of atypical double-hit lymphoma also precludes a definitive conclusion that all three subgroups are similarly aggressive. A larger case series is needed to further confirm the results we present. In addition, not all cases in this study were assessed by FISH for BCL6 rearrangements and one could speculate that some cases in the atypical DHLs group are MYC/BCL6 double-hit lymphoma. However, BCL6 was assessed by FISH on the 3 cases had a MYC translocation among the 40 cases of atypical double-hit lymphoma and no BCL6 rearrangement were identified. Therefore, the possibility of MYC/BCL6 double-hit lymphoma defined using conventional criteria is excluded. Despite these limitations, we believe the clinical presentation of patients with atypical versus typical DHL is similar. Furthermore, the significant difference in behavior between patients with atypical double-hit lymphoma versus patients with DLBCL (DLBCL) lacking MYC and BCL2 abnormalities supports the idea that patients with atypical double-hit lymphoma have a poor prognosis similar to patients with typical DHL.
In conclusion, a small subset of large B-cell lymphomas have concurrent MYC and BCL2 abnormalities other than translocations, designated as atypical MYC/BCL2 double-hit lymphoma. The clinical and pathologic features of these patients were very similar to patients with typical MYC/BCL2 DHL in this study. In particular, we show that patients with atypical DHL have aggressive clinical features and a poor prognosis, similar to patients with typical double-hit lymphoma and significantly worse than patients with DLBCL lacking MYC and BCL2 FISH abnormalities. Although information about atypical DHL cases is sparse in the literature, our results suggest that these cases appear to fall within the biologic spectrum of MYC/BCL2 DHL. We acknowledge that our study size is somewhat small and that additional larger studies are needed to further confirm the findings presented.
References
Aukema SM, Siebert R, Schuuring E et al. Double-hit B-cell lymphomas. Blood 2011;117:2319–2331.
Nie Z, Hu G, Wei G et al. c-Myc is a universal amplifier of expressed genes in lymphocytes and embryonic stem cells. Cell 2012;151:68–79.
Smith SM, Anastasi J, Cohen KS et al. The impact of MYC expression in lymphoma biology: beyond Burkitt lymphoma. Blood Cells Mol Dis 2010;45:317–323.
Bueno MJ, Gomez de Cedron M, Gomez-Lopez G et al. Combinatorial effects of microRNAs to suppress the Myc oncogenic pathway. Blood 2011;117:6255–6266.
Chang TC, Yu D, Lee YS et al. Widespread microRNA repression by Myc contributes to tumorigenesis. Nat Genet 2008;40:43–50.
Frenzel A, Loven J, Henriksson MA . Targeting MYC-regulated miRNAs to combat cancer. Genes Cancer 2010;1:660–667.
Han SS, Yun H, Son DJ et al. NF-kappaB/STAT3/PI3K signaling crosstalk in iMyc E mu B lymphoma. Mol Cancer 2010;9:97.
Klapproth K, Wirth T . Advances in the understanding of MYC-induced lymphomagenesis. Br J Haematol 2010;149:484–497.
Leskov I, Pallasch CP, Drake A et al. Rapid generation of human B-cell lymphomas via combined expression of Myc and Bcl2 and their use as a preclinical model for biological therapies. Oncogene 2013;32:1066–1072.
Li S, Lin P, Young KH et al. MYC/BCL2 double-hit high-grade B-Cell lymphoma. Adv Anat Pathol 2013;20:315–326.
Marin MC, Hsu B, Stephens LC et al. The functional basis of c-myc and bcl-2 complementation during multistep lymphomagenesis in vivo. Exp Cell Res 1995;217:240–247.
Shaffer AL 3rd, Young RM, Staudt LM . Pathogenesis of human B cell lymphomas. Annu Rev Immunol 2012;30:565–610.
Tracey L, Perez-Rosado A, Artiga MJ et al. Expression of the NF-kappaB targets BCL2 and BIRC5/Survivin characterizes small B-cell and aggressive B-cell lymphomas, respectively. J Pathol 2005;206:123–134.
Zhang X, Zhao X, Fiskus W et al. Coordinated silencing of MYC-mediated miR-29 by HDAC3 and EZH2 as a therapeutic target of histone modification in aggressive B-Cell lymphomas. Cancer Cell 2012;22:506–523.
Snuderl M, Kolman OK, Chen YB et al. B-cell lymphomas with concurrent IGH-BCL2 and MYC rearrangements are aggressive neoplasms with clinical and pathologic features distinct from Burkitt lymphoma and diffuse large B-cell lymphoma. Am J Surg Pathol 2010;34:327–340.
Tomita N, Tokunaka M, Nakamura N et al. Clinicopathological features of lymphoma/leukemia patients carrying both BCL2 and MYC translocations. Haematologica 2009;94:935–943.
Johnson NA, Savage KJ, Ludkovski O et al. Lymphomas with concurrent BCL2 and MYC translocations: the critical factors associated with survival. Blood 2009;114:2273–2279.
Kanungo A, Medeiros LJ, Abruzzo LV et al. Lymphoid neoplasms associated with concurrent t(14;18) and 8q24/c-MYC translocation generally have a poor prognosis. Mod Pathol 2006;19:25–33.
Le Gouill S, Talmant P, Touzeau C et al. The clinical presentation and prognosis of diffuse large B-cell lymphoma with t(14;18) and 8q24/c-MYC rearrangement. Haematologica 2007;92:1335–1342.
Li S, Lin P, Fayad LE et al. B-cell lymphomas with MYC/8q24 rearrangements and IGH@BCL2/t(14;18)(q32;q21): an aggressive disease with heterogeneous histology, germinal center B-cell immunophenotype and poor outcome. Mod Pathol 2012;25:145–156.
Lin P, Medeiros LJ . High-grade B-cell lymphoma/leukemia associated with t(14;18) and 8q24/MYC rearrangement: a neoplasm of germinal center immunophenotype with poor prognosis. Haematologica 2007;92:1297–1301.
Macpherson N, Lesack D, Klasa R et al. Small noncleaved, non-Burkitt’s (Burkit-Like) lymphoma: cytogenetics predict outcome and reflect clinical presentation. J Clin Oncol 1999;17:1558–1567.
Niitsu N, Okamoto M, Miura I et al. Clinical features and prognosis of de novo diffuse large B-cell lymphoma with t(14;18) and 8q24/c-MYC translocations. Leukemia 2009;23:777–783.
Pedersen MO, Gang AO, Poulsen TS et al. Double-hit BCL2/MYC translocations in a consecutive cohort of patients with large B-cell lymphoma—a single centre’s experience. Eur J Haematol 2012;89:63–71.
Kluin PM, Harris NL, Stein H, Leoncini L et al. B-cell lymphoma, unclassifiable, with features intermediate between diffuse large B-cell lymphoma and Burkitt lymphoma In: Swerdlow SH, Campo E, Harris NL, et al (ed) WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues 4th edn IARC: Lyon, 2008, pp 265–267.
Visco C, Li Y, Xu-Monette ZY et al. Comprehensive gene expression profiling and immunohistochemical studies support application of immunophenotypic algorithm for molecular subtype classification in diffuse large B-cell lymphoma: a report from the International DLBCL Rituximab-CHOP Consortium Program Study. Leukemia 2012;26:2103–2113.
Visco C, Tzankov A, Xu-Monette ZY et al. Patients with diffuse large B-cell lymphoma of germinal center origin with BCL2 translocations have poor outcome, irrespective of MYC status: a report from an International DLBCL rituximab-CHOP Consortium Program Study. Haematologica 2013;98:255–263.
Johnson NA, Slack GW, Savage KJ et al. Concurrent Expression of MYC and BCL2 in Diffuse Large B-Cell Lymphoma Treated With Rituximab Plus Cyclophosphamide, Doxorubicin, Vincristine, and Prednisone. J Clin Oncol 2012;30:3452–3459.
Shaffer LG, McGowan-Jordan J, M, (eds) An International System for Human Cytogenetic Nomenclature. Karger: Basel, Switzerland, 2013.
Green TM, Young KH, Visco C et al. Immunohistochemical double-hit score is a strong predictor of outcome in patients with diffuse large B-cell lymphoma treated with rituximab plus cyclophosphamide, Doxorubicin, vincristine, and prednisone. J Clin Oncol 2012;30:3460–3467.
Valera A, Lopez-Guillermo A, Cardesa-Salzmann T et al. MYC protein expression and genetic alterations have prognostic impact in diffuse large B-cell lymphoma treated with immunochemotherapy. Haematologica 2013;98:1554–1562.
Hans CP, Weisenburger DD, Greiner TC et al. Confirmation of the molecular classification of diffuse large B-cell lymphoma by immunohistochemistry using a tissue microarray. Blood 2004;103:275–282.
Mossafa H, Damotte D, Jenabian A et al. Non-Hodgkin’s lymphomas with Burkitt-like cells are associated with c-Myc amplification and poor prognosis. Leuk Lymphoma 2006;47:1885–1893.
Valera A, Lopez-Guillermo A, Cardesa-Salzmann T et al. MYC protein expression and genetic alterations have prognostic impact in patients with diffuse large B-cell lymphoma treated with immunochemotherapy. Haematologica 2013;98:1554–1562.
Hu S, Xu-Monette ZY, Tzankov A et al. MYC/BCL2 protein co-expression contributes to the inferior survival of activated B-cell subtype of diffuse large B-cell lymphoma and demonstrates high-risk gene expression signatures: a report from The International DLBCL Rituximab-CHOP Consortium Program Study. Blood 2013;121:4021–4031.
Testoni M, Kwee I, Greiner TC et al. Gains of MYC locus and outcome in patients with diffuse large B-cell lymphoma treated with R-CHOP. Br J Haematol 2011;155:274–277.
Yoon SO, Jeon YK, Paik JH et al. MYC translocation and an increased copy number predict poor prognosis in adult diffuse large B-cell lymphoma (DLBCL), especially in germinal centre-like B cell (GCB) type. Histopathology 2008;53:205–217.
Jiang HY, Li HL, Hu H et al. [Detection of t (14; 18) translocation and bcl-2 amplification in diffuse large B-cell lymphoma]. Zhonghua Bing Li Xue Za Zhi 2007;36:84–89.
Kusumoto S, Kobayashi Y, Sekiguchi N et al. Diffuse large B-cell lymphoma with extra Bcl-2 gene signals detected by FISH analysis is associated with a ‘non-germinal center phenotype’. Am J Surg Pathol 2005;29:1067–1073.
Monni O, Joensuu H, Franssila K et al. BCL2 overexpression associated with chromosomal amplification in diffuse large B-cell lymphoma. Blood 1997;90:1168–1174.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Li, S., Seegmiller, A., Lin, P. et al. B-cell lymphomas with concurrent MYC and BCL2 abnormalities other than translocations behave similarly to MYC/BCL2 double-hit lymphomas. Mod Pathol 28, 208–217 (2015). https://doi.org/10.1038/modpathol.2014.95
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/modpathol.2014.95
This article is cited by
-
A comparison of the International Consensus and 5th World Health Organization classifications of mature B-cell lymphomas
Leukemia (2023)
-
High-grade B-cell lymphoma (HGBL)-NOS is clinicopathologically and genetically more similar to DLBCL/HGBL-DH than DLBCL
Leukemia (2023)
-
Diagnostic approaches and future directions in Burkitt lymphoma and high-grade B-cell lymphoma
Virchows Archiv (2023)
-
Blastoid high-grade B-cell lymphoma initially presenting in bone marrow: a diagnostic challenge
Modern Pathology (2022)
-
The “Burkitt-like” immunophenotype and genotype is rarely encountered in diffuse large B cell lymphoma and high-grade B cell lymphoma, NOS
Virchows Archiv (2021)