Abstract
Diagnosing breast carcinoma that has metastasized to body cavity fluids can be difficult. Recently, immunostaining for the facultative glucose transporter Glut-1 has been described as a sensitive and specific means of detecting carcinomas in effusions. However, only five cases of breast carcinoma were studied. We examined Glut-1 specifically as a means of detecting breast carcinoma in effusion cytology. Using avidin-biotin immunocytochemistry, cell block material from 31 cases of breast carcinoma metastatic to body cavity effusions and 33 cases of benign effusions were studied. All cases were immunostained with the Glut-1 antibody. An additional set of slides from these same cases was stained for mucin using the Mayer's mucicarmine technique. Slides were graded for percentage of cells exhibiting immunoreactivity for Glut-1 or for the presence of mucin. Results of staining for both Glut-1 alone and in combination with mucicarmine were compared between the benign and malignant groups. Of the breast cancer cases, 19 of 31 (61%) were immunoreactive for Glut-1, and 25 of 31 (81%) were positive for either Glut-1 or mucicarmine. One of the 33 (3%) benign cases was immunoreactive for Glut-1, and none were positive for mucin. These data suggest that using Glut-1 as a single immunostain or in conjunction with mucicarmine is a specific but modestly sensitive means of detecting breast carcinoma in this cytologic setting.
Similar content being viewed by others
INTRODUCTION
The cytologic dilemma of distinguishing malignant cells from benign mesothelium has attracted a variety of novel diagnostic techniques, none of which are widely accepted for clinical use. The time, expense, or technical demands of such techniques as polymerase chain reaction (1), in situ hybridization (2, 3, 4), silver nuclear organizing region staining (5, 6), or flow cytometry (7) limit their usefulness in most hospitals. Immunostaining with a diversity of antibodies has been reported over the years (8, 9), Included in this investigative effort are antibodies against CA15–3 (10), MOC-31 (11), the fragile histidine triad (12), and p53 oncogene products (13). For all this, Bedrossian (14) has suggested a simple panel of periodic acid–Schiff with diastase and carcinoembryonic antigen to detect adenocarcinoma in serous effusions.
The Glut-1 protein is one of at least six glucose transport proteins found in various human cells (15). Glut-1 is normally expressed in human blood–brain barrier, placenta, and erythrocytes (16). Recent investigations have also described its expression in skin and its adnexae (17). Much investigation of Glut-1 expression has involved various non–small cell carcinomas of the lung and, to a lesser extent, the colon and other organs (18, 19, 20). Burstein et al. (21) examined Glut-1 expression in serous effusions and found Glut-1 expression in 93.5% of malignant and 20% of benign cases studied. This study included five cases of breast carcinoma, four (80%) of which exhibited immunoreactivity to Glut-1. Because metastatic breast carcinoma may defy easy diagnosis, we examined the panel of Glut-1 and mucicarmine as a means of detecting this malignancy in serous effusions.
MATERIALS AND METHODS
Thirty-one cases of malignant effusions from patients with known breast carcinoma were studied. These patients had no history of any other malignancy. Thirty-three additional cases of benign serous effusions served as controls. These control cases had no clinical history or suspicion of malignancy. The medical history of the benign cases is summarized in Table 1. Some of these cases have been reported previously (10, 12, 22, 23). The cell blocks had cellular material present on the last hematoxylin and eosin–stained slide cut. Four-micrometer sections were cut onto positively charged glass. After drying, the slides were deparaffinized in xylene and rehydrated in graded ethanol. No antigen retrieval was performed.
Staining for Glut-1 was performed using a modified avidin-biotin complex method on an Optimax Plus automated immunostainer (Biogenex, San Ramon CA). Briefly, slides were incubated with hydrogen peroxide for 5 minutes, followed by a 10-minute incubation with CAS protein block (Zymed Laboratories, South San Francisco, CA) for 10 minutes. Incubation with the Glut-1 antibody was for 1 hour at room temperature, with an antibody dilution of 1:100. After incubation with the primary antibody, the slides were incubated with a multilink secondary antibody (Zymed) for 30 minutes, followed by incubation with a horseradish-peroxidase conjugate (Zymed) for another 30 minutes. Diaminobenzidine (Zymed) was applied for 5 minutes, and the slides were then counterstained with hematoxylin. Sections of normal cerebral cortex were used as a positive control. Well-preserved erythrocytes, when present in the tissue section, served as internal positive controls. Mucicarmine staining was performed using the Mayer's method and normal colon as a positive control.
Slides stained for Glut-1 were evaluated for the presence of a well-defined membranous staining pattern using a 10 × or 20 × objective. Cytoplasmic staining without distinct membranous staining was disregarded. Slides stained with mucicarmine were assessed for the presence of cytoplasmic staining. One cell with clearly defined mucin present rendered the entire case positive for malignancy. Evaluation of slides was performed in a blinded fashion. After evaluation, the diagnoses were unblinded, the two groups of cases compared, and the χ2 test used to determine statistical significance.
RESULTS
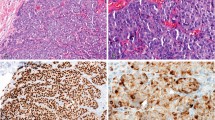
All cases were successfully stained for Glut-1 and all but one malignant case, for mucicarmine. The results are shown in Table 2. Nineteen of 31 (61%) cases of breast carcinoma exhibited some immunoreactivity for Glut-1, with an average of 26% of tumor cell exhibiting immunoreactivity (Fig. 1). In six cases, <10% of tumor cells were immunoreactive. Fifteen of 30 (50%) malignant effusions exhibited mucin staining; six of these cases were negative for Glut-1. Altogether, 25 of 31 (81%) of malignant cases were detected by this panel of stains.
Only one of 33 (3%) benign cases contained immunoreactive cells, and these comprised <1% of all cells present (Fig. 2). These immunoreactive cells were also morphologically benign. None of the benign mesothelium cases exhibited mucin staining. Overall, the sensitivity of Glut-1 for detection of metastatic breast carcinoma in serous effusions was 61%, and its specificity compared with detection of benign mesothelium was 97%. When used in combination with mucicarmine, the sensitivity was 81%, and the specificity was 97% (P < .001).
DISCUSSION
The challenge of discriminating malignant cells from reactive mesothelium has led cytopathologists to investigate a variety of immunostains and laboratory techniques as diagnostic adjuncts. Typically, an antibody panel of carcinoembryonic antigen, Ber-EP4, Leu M-1, and B72.3 is employed. In 1998, Bedrossian (14) reported a panel of carcinoembryonic antigen and periodic acid–Schiff-D to be the most reliable panel for discriminating mesothelium from carcinoma. More recently described antibodies such as CA15–3 (10), MOC-31 (11), and p53 (clone DO-7; 13), although showing promise, will have to pass the tests of time and further study. Simply stated, a reliable, easy, and relatively inexpensive method of detecting adenocarcinomas in serous effusions has yet to be found.
The task of identifying breast carcinoma in serous effusions can be especially difficult. A previous report described the performance of Glut-1 in serous effusions and reported a high sensitivity and specificity in this cytologic setting (21). Included in this work were five cases of metastatic breast carcinoma, four (80%) of which were reported as positive. In contrast, a study of surgical pathology material examined 11 cases of breast carcinoma and found only 45% of them to express Glut-1 (24). Data presented here falls midway between these two prior studies. Specifically, 19 of 31 (61%) cases were immunoreactive for Glut-1. The combination of mucicarmine and Glut-1 detected 81% of breast tumors. Although respectable, this performance is less than that of other individual immunostains such as CA15–3 and BCA-225 (10, 23). Of the 16 breast carcinoma cases studied with both Glut-1 and CA15–3, 100% stained with CA15–3, compared with 10 of 16 (63%) for Glut-1. Similarly, of those cases studied with BCA-225 and Glut-1, 17 of 19 (90%) stained with BCA-225, and 12 of 19 (63%) stained with Glut-1.
Even though Glut-1 is not as sensitive as CA15–3, BCA-225, and other immunostains, its specificity may prove clinically useful for detecting breast carcinoma in this cytologic setting. There is also evidence in the surgical pathology literature to suggest that Glut-1 may prove to be a sensitive means of detecting lung or colon carcinoma in serous effusions. Further study will be needed to determine whether Glut-1 is superior to carcinoembryonic antigen and periodic acid–Schiff-D or other panels for detecting these other malignancies in this cytologic setting.
CONCLUSION
Glut-1 is a glucose transport protein frequently expressed in many malignancies, including breast carcinoma. However, data presented here suggest that a modest number of metastatic breast carcinomas express this antigen in serous effusions. Thus, Glut-1 may be of limited diagnostic value in this cytologic setting.
References
Yamashita K, Kuba T, Shinoda H, Takahashi E, Okayasu I . Detection of K-ras point mutations in the supernatants of peritoneal and pleural effusions for diagnosis complementary to cytologic examination. Am J Clin Pathol 1998; 109: 704–711.
Zimmerman RL, Bibbo M . Clinical utility of chromosome 17 alpha satellite probe in distinguishing benign mesothelium from malignant cells: a pilot study using routinely fixed specimens. Oncol Rep 1999; 6: 695–698.
Athanassiadou PP, Veneti SZ, Kyrkou KA, Athanassiades PH . Detection of c-Ha-ras oncogene expression in pleural and peritoneal smear effusions by in situ hybridization. Cancer Detect Prev 1993; 17: 585–590.
Zojer N, Angeles J, Mullauer L, Gsur A, Roka S, Pechrstorfer M, et al. Interphase fluorescence in situ hybridization improves the detection of malignant cells in effusions from breast cancer patients. Br J Cancer 1977; 75: 403–407.
Trevisan MS, Souza MI, Magna LA . Nucleolar organiser regions of mesothelial and carcinomatous cells in effusions. Diagn Cytopathol 1993; 9: 492–497.
Carrillo R, Sneige N, el-Naggar AK . Interphase nucleolar organizer regions in the evaluation of serosal cavity effusions. Acta Cytol 1994; 38: 367–372.
Chen LM, Lazcano O, Katzmann JA, Kimlinger TK, Li CY . The role of conventional cytology, immunocytochemistry, and flow cytometric DNA ploidy in the evaluation of body cavity fluids: a prospective study of 52 patients. Am J Clin Pathol 1998; 109: 712–721.
Ordóñez NG . Value of calretinin immunostaining in differentiating epithelial mesotheliomas from lung adenocarcinomas. Mod Pathol 1998; 11: 929–933.
Osborn M, Wenancjusz D . Special techniques in cytopathology. In: Bibbo M, ed. Comprehensive cytopathology. 2nd ed. Philadelphia: Saunders; 1996; p. 1058–1060.
Zimmerman RL, Fogt F, Goonewardene S . Diagnostic value of a second generation CA15–3 antibody to detect adenocarcinoma in body cavity effusions. Cancer Cytopathol 2000; 90: 230–234.
Morgan RL, De Young BR, McGaughty VR, Niemann TH . MOC-31 aids in the differentiation between adenocarcinoma and reactive mesothelial cells. Cancer Cytopathol 1999; 87: 390–394.
Zimmerman RL . The FHIT protein is expressed in benign mesothelium and has no clinical value in detecting carcinoma in body cavity effusions. Appl Immunohistochem Mol Morphol 2000; 8: 154–157.
Pindzola JA, Kovatich AJ, Bibbo M . p53 immunohistochemistry for distinguishing reactive mesothelium from low grade ovarian carcinoma. Acta Cytol 2000; 44: 31–36.
Bedrossian CWM . Special stains, the old and the new: the impact of immunocytochemistry in effusion cytology. Diagn Cytopathol 1998; 18: 141–149.
Pessin JE, Bell GI . Mammalian facultative glucose transporter family: structure and molecular regulation. Annu Rev Physiol 1992; 54: 511–530.
Fogt F, Vortmeyer AO, De Girolami U, Cukor B, Ahn G, Loda M . Blood-brain barrier immunophenotype of microvessels within neuroectodermal tissue in mature ovarian teratomas. Appl Immunohistochem 1994; 2: 268–273.
Reisser C, Eichhorn K, Herold-Mende C, Born AI, Bannasch P . Expression of facilitative glucose transport proteins during development of squamous cell carcinomas of the head and neck. Int J Cancer 1999; 80: 194–198.
Ito T, Noguchi Y, Satoh S, Hayashi H, Inayama Y, Kitamura H . Expression of facilitative glucose isoforms in lung carcinomas: its relation to histologic type, differentiation grade, and tumor stage. Mod Pathol 1998; 11: 437–443.
Brown RS, Leung JY, Kison PV, Zasadney KR, Flint A, Wahl RL . Glucose transporters and FDG uptake in untreated primary human non-small cell lung cancer. J Nucl Med 1999; 40: 556–565.
Haber RS, Rathan A, Weiser KR, Pritsker A, Itzkowitz SH, Bodian C, et al. GLUT1 glucose transporter expression in colorectal carcinoma: a marker for poor prognosis. Cancer 1998; 83: 34–40.
Burstein DE, Reder I, Weiser K, Tong T, Pritsker A, Haber RS . GLUT1 glucose transporter: a highly sensitive marker of malignancy in body cavity effusions. Mod Pathol 1998; 11: 392–396.
Zimmerman RL, Fogt F, Bibbo M . Diagnostic utility of sialosyl-Tn in discriminating carcinomatous cells from benign mesothelium in body cavity effusions. Acta Cytol 1999; 43: 1079–1084.
Zimmerman RL, Fogt F, Goonewardene S . Diagnostic utility of BCA-225 in detecting adenocarcinoma in serous effusions. Anal Quant Cytol Histol 2000; 22: 353–357.
Younes M, Lechago LV, Somoano JR, Mosharaf M, Lechago J . Wide expression of the human erythrocyte glucose transporter Glut1 in human cancers. Cancer Res 1996; 56: 1164–1167.
Author information
Authors and Affiliations
Corresponding author
Additional information
This study was presented in abstract form at the 14th International Congress of Cytopathology, hosted by the International Academy of Cytopathology, May 27–31, 2001, Amsterdam, the Netherlands.
Rights and permissions
About this article
Cite this article
Zimmerman, R., Goonewardene, S. & Fogt, F. Glucose Transporter Glut-1 Is of Limited Value for Detecting Breast Carcinoma in Serous Effusions. Mod Pathol 14, 748–751 (2001). https://doi.org/10.1038/modpathol.3880384
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/modpathol.3880384