Abstract
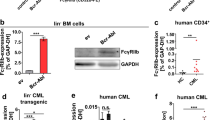
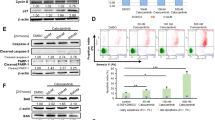
Overcoming resistance against BCR-ABL-inhibitors in chronic myeloid leukemia (CML) is central to prevent progression to advanced phase disease. Kinase mutations of BCR-ABL and cytokine-mediated modulation of response to tyrosine kinase inhibitors (TKIs) are key mechanisms governing clinical response to imatinib and second generation TKIs. Omacetaxine mepesuccinate is effective in imatinib-resistant CML with reported stem cell activity. We specifically thought to explore omacetaxine in the context of the pan-resistant mutant T315I, and in its potential to modify cytokine-dependent resistance. Omacetaxine was investigated in cell lines and primary CD34+ enriched progenitor cells from patients with CML. Addition of cytokines, shown to revert the efficacy of TKIs in BCR-ABL-positive cells, does not affect omacetaxine mediated antiproliferative activity, neither in cell lines nor in primary CML CD34+ progenitor cells. Looking at potential mechanisms, we found marked downregulation of the common β-subunit c of the cytokine-receptors (cCRβc) for IL3, IL5 and GM-CSF by omacetaxine in cell lines and primary progenitor cultures. The observed cytokine-independent in-vitro cytotoxicity of omacetaxine may be explained by downregulation of cCRβc. Whether this can be used clinically as a means to optimize the stem cell activity of TKIs merits further evaluation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Quintas-Cardama A, Cortes J . Molecular biology of bcr-abl1-positive chronic myeloid leukemia. Blood 2009; 113: 1619–1630.
Daley GQ, Baltimore D . Transformation of an interleukin 3-dependent hematopoietic cell line by the chronic myelogenous leukemia-specific P210bcr/abl protein. Proc Natl Acad Sci USA 1988; 85: 9312–9316.
Druker BJ, Guilhot F, O′Brien SG, Gathmann I, Kantarjian H, Gattermann N et al. Five-year follow-up of patients receiving imatinib for chronic myeloid leukemia. N Engl J Med 2006; 355: 2408–2417.
Apperley JF . Part I: mechanisms of resistance to imatinib in chronic myeloid leukaemia. Lancet Oncol 2007; 8: 1018–1029.
Cortes J, Hochhaus A, Hughes T, Kantarjian H . Front-line and salvage therapies with tyrosine kinase inhibitors and other treatments in chronic myeloid leukemia. J Clin Oncol 2011; 29: 524–531.
Nicolini FE, Mauro MJ, Martinelli G, Kim DW, Soverini S, Muller MC et al. Epidemiologic study on survival of chronic myeloid leukemia and Ph(+) acute lymphoblastic leukemia patients with BCR-ABL T315I mutation. Blood 2009; 114: 5271–5278.
Chu S, Holtz M, Gupta M, Bhatia R . BCR/ABL kinase inhibition by imatinib mesylate enhances MAP kinase activity in chronic myelogenous leukemia CD34+ cells. Blood 2004; 103: 3167–3174.
Wang Y, Cai D, Brendel C, Barett C, Erben P, Manley PW et al. Adaptive secretion of granulocyte-macrophage colony-stimulating factor (GM-CSF) mediates imatinib and nilotinib resistance in BCR/ABL+ progenitors via JAK-2/STAT-5 pathway activation. Blood 2007; 109: 2147–2155.
Druker BJ, Tamura S, Buchdunger E, Ohno S, Segal GM, Fanning S et al. Effects of a selective inhibitor of the Abl tyrosine kinase on the growth of Bcr-Abl positive cells. Nat Medic 1996; 2: 561–566.
Härtel N, Klag T, Hanfstein B, Mueller MC, Schenk T, Erben P et al. Enhanced ABL-inhibitor-induced MAPK-activation in T315I-BCR-ABL-expressing cells: a potential mechanism of altered leukemogenicity. J Cancer Res Clin Oncol; e-pub ahead of print 17 November 2011; PMID:22089930.
Härtel N, Klag T, Hochhaus A, La Rosée P . Overexpression of the IL3/IL5/GM-CSFRβ as potential mechanism leading to survival factor activation in response to ABL-inhibitor treatment. Haematologica 2009; 93 (S1): 382(abstract).
Nicolini FE, Chomel JC, Roy L, Legros L, Chabane K, Ducastelle S et al. The durable clearance of the T315I BCR-ABL mutated clone in chronic phase chronic myelogenous leukemia patients on omacetaxine allows tyrosine kinase inhibitor rechallenge. Clin Lymphoma Myeloma Leuk 2010; 10: 394–399.
Chen Y, Hu Y, Michaels S, Segal D, Brown D, Li S . Inhibitory effects of omacetaxine on leukemic stem cells and BCR-ABL-induced chronic myeloid leukemia and acute lymphoblastic leukemia in mice. Leukemia 2009; 23: 1446–1454.
Wetzler M, Segal D . Omacetaxine as an anticancer therapeutic: what is old is new again. Curr Pharm Des 2011; 17: 59–64.
Palacios R, Steinmetz M . Il-3-dependent mouse clones that express B-220 surface antigen, contain Ig genes in germ-line configuration, and generate B lymphocytes in vivo. Cell 1985; 41: 727–734.
La Rosée P, Corbin AS, Stoffregen EP, Deininger MW, Druker BJ . Activity of the Bcr-Abl kinase inhibitor PD180970 against clinically relevant Bcr-Abl isoforms that cause resistance to imatinib mesylate (Gleevec, STI571). Cancer Res 2002; 62: 7149–7153.
Greenberger JS, Sakakeeny MA, Humphries RK, Eaves CJ, Eckner RJ . Demonstration of permanent factor-dependent multipotential (erythroid/neutrophil/basophil) hematopoietic progenitor cell lines. Proc Natl Acad Sci USA 1983; 80: 2931–2935.
Beran M, Pisa P, O′Brien S, Kurzrock R, Siciliano M, Cork A et al. Biological properties and growth in SCID mice of a new myelogenous leukemia cell line (KBM-5) derived from chronic myelogenous leukemia cells in the blastic phase. Cancer Res 1993; 53: 3603–3610.
Ricci C, Scappini B, Divoky V, Gatto S, Onida F, Verstovsek S et al. Mutation in the ATP-binding Pocket of the ABL kinase domain in an STI571-resistant BCR/ABL-positive cell line. Cancer Res 2002; 62: 5995–5998.
Lozzio CB, Lozzio BB . Human chronic myelogenous leukemia cell-line with positive Philadelphia chromosome. Blood 1975; 45: 321–334.
Bhatia R, McGlave PB, Dewald GW, Blazar BR, Verfaillie CM . Abnormal function of the bone marrow microenvironment in chronic myelogenous leukemia: role of malignant stromal macrophages. Blood 1995; 85: 3636–3645.
Donato NJ, Wu JY, Zhang L, Kantarjian H, Talpaz M . Down-regulation of interleukin-3/granulocyte-macrophage colony-stimulating factor receptor beta-chain in BCR-ABL(+) human leukemic cells: association with loss of cytokine-mediated Stat-5 activation and protection from apoptosis after BCR-ABL inhibition. Blood 2001; 97: 2846–2853.
Dorsey JF, Cunnick JM, Lanehart R, Huang M, Kraker AJ, Bhalla KN et al. Interleukin-3 protects Bcr-Abl-transformed hematopoietic progenitor cells from apoptosis induced by Bcr-Abl tyrosine kinase inhibitors. Leukemia 2002; 16: 1589–1595.
Tang R, Faussat AM, Majdak P, Marzac C, Dubrulle S, Marjanovic Z et al. Semisynthetic homoharringtonine induces apoptosis via inhibition of protein synthesis and triggers rapid myeloid cell leukemia-1 down-regulation in myeloid leukemia cells. Mol Cancer Ther 2006; 5: 723–731.
Copland M, Hamilton A, Elrick LJ, Baird JW, Allan EK, Jordanides N et al. Dasatinib (BMS-354825) targets an earlier progenitor population than imatinib in primary CML but does not eliminate the quiescent fraction. Blood 2006; 107: 4532–4539.
Konig H, Copland M, Chu S, Jove R, Holyoake TL, Bhatia R . Effects of dasatinib on SRC kinase activity and downstream intracellular signaling in primitive chronic myelogenous leukemia hematopoietic cells. Cancer Res 2008; 68: 9624–9633.
Konig H, Holyoake TL, Bhatia R . Effective and selective inhibition of chronic myeloid leukemia primitive hematopoietic progenitors by the dual Src/Abl kinase inhibitor SKI-606. Blood 2008; 111: 2329–2338.
Konig H, Holtz M, Modi H, Manley P, Holyoake TL, Forman SJ et al. Enhanced BCR-ABL kinase inhibition does not result in increased inhibition of downstream signaling pathways or increased growth suppression in CML progenitors. Leukemia 2008; 22: 748–755.
Chu S, Xu H, Shah NP, Snyder DS, Forman SJ, Sawyers CL et al. Detection of BCR-ABL kinase mutations in CD34+ cells from chronic myelogenous leukemia patients in complete cytogenetic remission on imatinib mesylate treatment. Blood 2005; 105: 2093–2098.
Jiang X, Zhao Y, Smith C, Gasparetto M, Turhan A, Eaves A et al. Chronic myeloid leukemia stem cells possess multiple unique features of resistance to BCR-ABL targeted therapies. Leukemia 2007; 21: 926–935.
La Rosee P, Deininger MW . Resistance to imatinib: mutations and beyond. Semin Hematol 2010; 47: 335–343.
Robert F, Carrier M, Rawe S, Chen S, Lowe S, Pelletier J . Altering chemosensitivity by modulating translation elongation. PLoS ONE 2009; 4: e5428.
Liu J, Joha S, Idziorek T, Corm S, Hetuin D, Philippe N et al. BCR-ABL mutants spread resistance to non-mutated cells through a paracrine mechanism. Leukemia 2008; 22: 791–799.
Allan EK, Holyoake TL, Craig AR, Jorgensen HG . Omacetaxine may have a role in chronic myeloid leukaemia eradication through downregulation of Mcl-1 and induction of apoptosis in stem/progenitor cells. Leukemia 2011; 25: 985–994.
Corbin AS, Agarwal A, Loriaux M, Cortes J, Deininger MW, Druker BJ . Human chronic myeloid leukemia stem cells are insensitive to imatinib despite inhibition of BCR-ABL activity. J Clin Invest 2011; 121: 396–409.
Acknowledgements
We would like to thank Ms Susanne Brendel and Ms Silke Will for expert technical assistance. Chemgenex Pharmaceuticals (Menlo Park, CA, USA) provided Omacetaxine. AH received research support by Novartis Pharmaceuticals, Nürnberg, Germany and Chemgenex Pharmaceuticals. This work was funded by the Max Eder program, Deutsche Krebshilfe, Germany (PL).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
AH received research support by Novartis Pharmaceuticals, Nürnberg, Germany and Chemgenex Pharmaceuticals (Menlo Park, CA, USA). PL receives speaker honorarium from Novartis.
Additional information
Supplementary Information accompanies the paper on the Leukemia website
Supplementary information
Rights and permissions
About this article
Cite this article
Klag, T., Härtel, N., Erben, P. et al. Omacetaxine mepesuccinate prevents cytokine-dependent resistance to nilotinib in vitro: potential role of the common β-subunit c of cytokine receptors. Leukemia 26, 1321–1328 (2012). https://doi.org/10.1038/leu.2011.380
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2011.380