Abstract
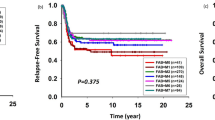
Despite substantial progress in the management of childhood acute myeloid leukemia (AML), only about 50% of patients are cured by intensive chemotherapy. The long-term results of clinical trials may reveal principles that can guide the development of future therapy. From 1980 to 2000, 251 patients <15 years of age with newly diagnosed AML were enrolled on one of the five consecutive St Jude AML studies. The median age of the 128 boys and 123 girls was 6.2 years; 193 were white, 45 black, and 13 of other racial groups. With the exception of one protocol (AML-83), outcomes improved in general over the two decades. The estimated 5-year event-free survival (±s.e.) was 30.8±5.6% for AML-80; 11.1±4.3% for AML-83; 35.9±7.4% for AML-87; 43.5±6.2% for AML-91; and 45.0±11.1% for AML-97. Resistant or relapsed AML caused the great majority of treatment failures. Increasing the intensity of chemotherapy (AML-87) did not improve outcome, partially because of toxicity, nor did prolonging postremission therapy by adding sequential myeloablative (AML-80) or nonmyeloablative (AML-83) chemotherapy cycles. We conclude that subtype-specific therapies are needed to replace the ‘one size fits all’ strategy of the past two decades.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Choi SI, Simone JV . Acute nonlymphocytic leukemia in 171 children. Med Pediatr Oncol 1976; 2: 119–146.
Weinstein HJ, Mayer RJ, Rosenthal DS, Camitta BM, Coral FS, Nathan DG et al. Treatment of acute myelogenous leukemia in children and adults. N Engl J Med 1980; 303: 473–478.
Athale UH, Razzouk BI, Raimondi SC, Tong X, Behm FG, Head DR et al. Biology and outcome of childhood acute megakaryoblastic leukemia: a single institution's experience. Blood 2001; 97: 3727–3732.
Dahl GV, Kalwinsky DK, Mirro Jr J, Look AT, Pui CH, Murphy SB et al. Allogeneic bone marrow transplantation in a program of intensive sequential chemotherapy for children and young adults with acute nonlymphocytic leukemia in first remission. J Clin Oncol 1990; 8: 295–303.
Kalwinsky D, Mirro Jr J, Schell M, Behm F, Mason C, Dahl GV . Early intensification of chemotherapy for childhood acute nonlymphoblastic leukemia: improved remission induction with a five-drug regimen including etoposide. J Clin Oncol 1988; 6: 1134–1143.
Arnaout MK, Radomski KM, Srivastava DK, Tong X, Belt JR, Raimondi SC et al. Treatment of childhood acute myelogenous leukemia with an intensive regimen (AML-87) that individualizes etoposide and cytarabine dosages: s. Leukemia 2000; 14: 1736–1742.
Pui CH, Mahmoud HH, Rivera GK, Hancock ML, Sandlund JT, Behm FG et al. Early intensification of intrathecal chemotherapy virtually eliminates central nervous system relapse in children with acute lymphoblastic leukemia. Blood 1998; 92: 411–415.
Krance RA, Hurwitz CA, Head DR, Raimondi SC, Behm FG, Crews KR et al. Experience with 2-chlorodeoxyadenosine in previously untreated children with newly diagnosed acute myeloid leukemia and myelodysplastic diseases. J Clin Oncol 2001; 19: 2804–2811.
Brenner MK, Rill DR, Moen RC, Krance RA, Mirro Jr J, Anderson WF et al. Gene-marking to trace origin of relapse after autologous bone-marrow transplantation. Lancet 1993; 341: 85–86.
Crews KR, Gandhi V, Srivastava DK, Razzouk BI, Tong X, Behm FG et al. Interim comparison of a continuous infusion vs a short daily infusion of cytarabine given in combination with cladribine for pediatric acute myeloid leukemia. J Clin Oncol 2002; 20: 4217–4224.
Klein JP, Moeschberger ML . Survival Analysis: Techniques for Censored and Truncated Data (Statistics for Biology and Health), 2nd edn. New York: Springer, 2003.
Smith MA, Ries LAG, Gurney JG, Ross JA . Leukemia. In: Ries LAG, Smith MA, Gurney JG et al (eds) Cancer Incidence and Survival among Children and Adolescents: United States SEER Program 1975–1995. Bethesda, MD: National Cancer Institute, 1999, pp 17–31.
Gatta G, Luksch R, Coleman MP, Corazziari I, EUROCARE Working Group. Survival from acute non-lymphocytic leukaemia (ANLL) and chronic myeloid leukaemia (CML) in European children since 1978: a population-based study. Eur J Cancer 2001; 37: 695–702.
Stevens RF, Hann IM, Wheatley K, Gray RG . Marked improvements in outcome with chemotherapy alone in paediatric acute myeloid leukemia: results of the United Kingdom Medical Research Council's 10th AML trial. MRC Childhood Leukaemia Working Party. Br J Haematol 1998; 101: 130–140.
Perel Y, Auvrignon A, Leblanc T, Vannier JP, Michel G, Nelken B et al. Impact of addition of maintenance therapy to intensive induction and consolidation chemotherapy for childhood acute myeloblastic leukemia: results of a prospective randomized trial, LAME 89/91. Leucamie Aique Myeloide Enfant. J Clin Oncol 2002; 20: 2774–2782.
Woods WG, Kobrinsky N, Buckley JD, Lee JW, Sanders J, Neudorf S et al. Timed-sequential induction therapy improves postremission outcome in acute myeloid leukemia: a report from the Children's Cancer Group. Blood 1996; 87: 4979–4989.
Lehrnbecher T, Varwig D, Kaiser J, Reinhardt D, Klingebiel T, Creutzig U . Infectious complications in pediatric acute myeloid leukemia: analysis of the prospective multi-institutional clinical trial AML-BFM 93. Leukemia 2004; 18: 72–77.
Leahey AM, Teunissen H, Friedman DL, Moshang T, Lange BJ, Meadows AT . Late effects of chemotherapy compared to bone marrow transplantation in the treatment of pediatric acute myeloid leukemia and myelodysplasia. Med Pediatr Oncol 1999; 32: 163–169.
Leung W, Hudson MM, Strickland DK, Phipps S, Srivastava DK, Ribeiro RC et al. Late effects of treatment in survivors of childhood acute myeloid leukemia. J Clin Oncol 2000; 18: 3273–3279.
Abbott BL, Rubnitz JE, Tong X, Srivastava DK, Pui CH, Ribeiro RC et al. Clinical significance of central nervous system involvement at diagnosis of pediatric acute myeloid leukemia: a single institution's experience. Leukemia 2003; 17: 2090–2096.
Kalwinsky DK, Raimondi SC, Schell MJ, Mirro Jr J, Santana VM, Behm F et al. Prognostic importance of cytogenetic subgroups in de novo pediatric acute nonlymphocytic leukemia. J Clin Oncol 1990; 8: 75–83.
Rubnitz JE, Raimondi SC, Tong X, Srivastava DK, Razzouk BI, Shurtleff SA et al. Favorable impact of the t(9;11) in childhood acute myeloid leukemia. J Clin Oncol 2002; 20: 2302–2309.
Martinez-Climent JA, Lane NJ, Rubin CM, Srivastava DK, Razzouk BI, Shurtleff SA et al. Clinical and prognostic significance of chromosomal abnormalities in childhood acute myeloid leukemia de novo. Leukemia 1995; 9: 95–101.
Swansbury GJ, Slater R, Bain BJ, Moorman AV, Secker-Walker LM . Hematological malignancies with t(9;11)(p21–22;q23) – a laboratory and clinical study of 125 cases. European 11q23 Workshop participants. Leukemia 1998; 12: 792–800.
Wheatley K . Current controversies: which patients with acute myeloid leukaemia should receive a bone marrow transplantation? – a statistician's view. Br J Haematol 2002; 118: 351–356.
Burnett AK, Wheatley K, Goldstone AH, Stevens RF, Hann IM, Rees JH et al. The value of allogeneic bone marrow transplant in patients with acute myeloid leukaemia at differing risk of relapse: results of the UK MRC AML 10 trial. Br J Haematol 2002; 118: 385–400.
Coustan-Smith E, Ribeiro RC, Rubnitz JE, Razzouk BI, Pui CH, Pounds S et al. Clinical significance of residual disease during treatment in childhood acute myeloid leukaemia. Br J Haematol 2003; 123: 243–252.
Acknowledgements
We thank Sharon Naron for editorial assistance. This work was supported in part by Cancer Center Support (CORE) Grant CA-21765 from the National Cancer Institute, USA, by a Center of Excellence grant from the State of Tennessee, USA, and by the American Lebanese Syrian Associated Charities (ALSAC), USA, C-H Pui is the American Cancer Society FM Kirby Clinical Research Professor.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information
Supplementary Information accompanies the paper on the Leukemia website (http://www.nature.com/leu).
Rights and permissions
About this article
Cite this article
Ribeiro, R., Razzouk, B., Pounds, S. et al. Successive clinical trials for childhood acute myeloid leukemia at St Jude Children's Research Hospital, from 1980 to 2000. Leukemia 19, 2125–2129 (2005). https://doi.org/10.1038/sj.leu.2403872
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2403872
Keywords
This article is cited by
-
Effect of RNAi-induced down regulation of nuclear factor kappa-B p65 on acute monocytic leukemia THP-1 cells in vitro and vivo
Molecular and Cellular Biochemistry (2012)
-
The heterogeneity of pediatric MLL-rearranged acute myeloid leukemia
Leukemia (2011)
-
Childhood Acute Myeloid Leukemia
Current Treatment Options in Oncology (2008)
-
Pediatric acute myeloid leukemia with NPM1 mutations is characterized by a gene expression profile with dysregulated HOX gene expression distinct from MLL-rearranged leukemias
Leukemia (2007)
-
Predictive value of multidrug resistance proteins and cellular drug resistance in childhood relapsed acute lymphoblastic leukemia
Journal of Cancer Research and Clinical Oncology (2007)