Abstract
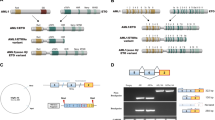
The human AML1 gene (also named CBFA2 or RUNX1), located in the 21q22 chromosomal band, encodes for one of the two subunits forming a heterodimeric transcription factor, the human core binding factor (CBF). AML1 protein contains a highly evolutionary conserved domain of 128 amino acids called runt domain, responsible for both heterodimerization with the β subunit of CBF and for DNA binding. AML1 is normally expressed in all hematopoietic lineages and acts to regulate the expression of various genes specific to hematopoiesis playing a pivotal role in myeloid differentiation. AML1 is one of the genes most frequently deregulated in leukemia through different mechanisms including translocation, mutation and amplification. Translocations lead to the formation of fusion genes encoding for chimerical proteins such as AML1-ETO which induces leukemogenesis. Recently, new mechanisms of AML1 deregulation by point mutations or amplification have been reported. To our knowledge, 51 patients (among 805 studied) with AML1 point mutations have been described. Forty of them have acute myeloid leukemia (AML) most often M0 AML. In this subtype of AML, the frequency of AML1 mutation is significantly higher; 21.5% of patients mutated (34/158). Mutations have also been found with lower frequency in other FAB subtype AML (6 cases), in myeloproliferative disorders (6 cases), in myelodysplastic syndrome (3 cases) and rarely in acute lymphoblastic leukemia (1 case). AML1 gene amplification has been found essentially in childhood ALL (12 cases) and more rarely in myeloid malignancies (4 cases). Here, we reviewed all these cases of AML1 point mutations and amplification and focused on the mechanisms of AML1 deregulation induced by these alterations.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Miyoshi, H, Shimizu, K, Kozu, T, Maseki, N, Kaneko, Y & Ohki, M t(8;21) breakpoints on chromosome 21 in acute myeloid leukemia are clustered within a limited region of a single gene, AML1. Proc Natl Acad Sci USA, (1991). 88, 1031–1034.
Levanon, D, Negreanu, V, Bernstein, Y, Bar–Am, I, Avivi, L & Groner, Y AML1, AML2, and AML3, the human members of the runt domain gene-family: cDNA structure, expression, and chromosomal localization. Genomics, (1994). 23, 425–432.
Speck, NA, Stacy, T, Wang, Q, North, T, Gu, TL, Miller, J, Binder, M & Marin-Padilla, M Core-binding factor: a central player in hematopoiesis and leukemia. Cancer Res, (1999). 59, 1789s–1793s.
Ogawa, E, Maruyama, M, Kagoshima, H, Inuzuka, M, Lu, J, Satake, M, Shigesada, K & Ito, Y PEBP2/PEA2 represents a family of transcription factors homologous to the products of the Drosophila runt gene and the human AML1 gene. Proc Natl Acad Sci USA, (1993). 90, 6859–6863.
Downing, JR AML1/CBFbeta transcription complex: its role in normal hematopoiesis and leukemia. Leukemia, (2001). 15, 664–665.
Wang, Q, Stacy, T, Miller, JD, Lewis, AF, Gu, TL, Huang, X, Bushweller, JH, Bories, JC, Alt, FW, Ryan, G, Liu, PP, Wynshaw-Boris, A, Binder, M, Marin-Padilla, M, Sharpe, AH & Speck, NA The CBFbeta subunit is essential for CBFalpha2 (AML1) function in vivo. Cell, (1996). 87, 697–708.
Tanaka, T, Tanaka, K, Ogawa, S, Kurokawa, M, Mitani, K, Nishida, J, Shibata, Y, Yazaki, Y & Hirai, H An acute myeloid leukemia gene, AML1, regulates hemopoietic myeloid cell differentiation and transcriptional activation antagonistically by two alternative spliced forms. EMBO J, (1995). 14, 341–350.
Lutterbach, B & Hiebert, SW Role of the transcription factor AML-1 in acute leukemia and hematopoietic differentiation. Gene, (2000). 245, 223–235.
Rhoades, KL, Hetherington, CJ, Rowley, JD, Hiebert, SW, Nucifora, G, Tenen, DG & Zhang, DE Synergistic up-regulation of the myeloid-specific promoter for the macrophage colony-stimulating factor receptor by AML1 and the t(8;21) fusion protein may contribute to leukemogenesis. Proc Natl Acad Sci USA, (1996). 93, 11895–11900.
Uchida, H, Zhang, J & Nimer, SD AML1A and AML1B can transactivate the human IL-3 promoter. J Immunol, (1997). 158, 2251–2258.
Wang, Q, Stacy, T, Binder, M, Marin-Padilla, M, Sharpe, AH & Speck, NA Disruption of the Cbfa2 gene causes necrosis and hemorrhaging in the central nervous system and blocks definitive hemapoiesis. Proc Natl Acad Sci USA, (1996). 93, 3444–3449.
Sasaki, K, Yagi, H, Bronson, RT, Tominaga, K, Matsunashi, T, Deguchi, K, Tani, Y, Kishimoto, T & Komori, T Absence of fetal liver hematopoiesis in mice deficient in transcriptional coactivator core binding factor beta. Proc Natl Acad Sci USA, (1996). 93, 12359–12363.
Okuda, T, van Deursen, J, Hiebert, SW, Grosveld, G & Downing, JR AML1, the target of multiple chromosomal translocations in human leukemia, is essential for normal fetal liver hematopoiesis. Cell, (1996). 84, 321–330.
Nucifora, G & Rowley, JD AML1 and the 8;21 and 3;21 translocations in acute and chronic myeloid leukemia. Blood, (1995). 86, 1–14.
Romana, SP, Poirel, H, Leconiat, M, Flexor, MA, Mauchauffe, M, Jonveaux, P, Macintyre, EA, Berger, R & Bernard, OA High frequency of t(12;21) in childhood B-lineage acute lymphoblastic leukemia. Blood, (1995). 86, 4263–4269.
Okuda, T, Takeda, K, Fujita, Y, Nishimura, M, Yagyu, S, Yoshida, M, Akira, S, Downing, JR & Abe, T Biological characteristics of the leukemia-associated transcriptional factor AML1 disclosed by hematopoietic rescue of AML1-deficient embryonic stem cells by using a knock-in strategy. Mol Cell Biol, (2000). 20, 319–328.
Westendorf, JJ, Yamamoto, CM, Lenny, N, Downing, JR, Selsted, ME & Hiebert, SW The t(8;21) fusion product, AML-1-ETO, associates with C/EBP-alpha, inhibits C/EBP-alpha-dependent transcription, and blocks granulocytic differentiation. Mol Cell Biol, (1998). 18, 322–333.
Tanaka, K, Tanaka, T, Kurokawa, M, Imai, Y, Ogawa, S, Mitani, K, Yazaki, Y & Hirai, H The AML1/ETO(MTG8) and AML1/Evi-1leukemia-associated chimeric oncoproteins accumulate PEBP2beta(CBFbeta) in the nucleus more efficiently than wild-type AML1. Blood, (1998). 91, 1688–1699.
Osato, M, Asou, N, Abdalla, E, Hoshino, K, Yamasaki, H, Okubo, T, Suzushima, H, Takatsuki, K, Kanno, T, Shigesada, K & Ito, Y Biallelic and heterozygous point mutations in the runt domain of the AML1/PEBP2alphaB gene associated with myeloblastic leukemias. Blood, (1999). 93, 1817–1824.
Preudhomme, C, Warot-Loze, D, Roumier, C, Grardel-Duflos, N, Garand, R, Lai, JL, Dastugue, N, Macintyre, E, Denis, C, Bauters, F, Kerckaert, JP, Cosson, A & Fenaux, P High incidence of biallelic point mutations in the Runt domain of the AML1/PEBP2 alpha B gene in Mo acute myeloid leukemia and in myeloid malignancies with acquired trisomy 21. Blood, (2000). 96, 2862–2869.
Imai, Y, Kurokawa, M, Izutsu, K, Hangaishi, A, Takeuchi, K, Maki, K, Ogawa, S, Chiba, S, Mitani, K & Hirai, H Mutations of the AML1 gene in myelodysplastic syndrome and their functional implications in leukemogenesis. Blood, (2000). 96, 3154–3160.
Steer, EJ, Goldman, JM & Cross, NC Mutations of the transcription factor AML1/CBFA2 are uncommon in blastic transformation of chronic myeloid leukaemia. Leukemia, (2001). 15, 476–477.
Langabeer, SE, Gale, RE, Rollinson, SJ, Morgan, GJ & Linch, DC Mutations of the AML1 gene in acute myeloid leukaemia of FAB types M0 and M7. Genes Chromosomes Cancer, (2002). 34, 24–32.
Yeoh, A, Williams, K, Behm, F, Lenny, N, Shih, L, Harada, Y, Gilliland, G & Downing, J Somatic mutations of the AML1 gene are frequent in acute myeloid leukemia with FAB M0 morphology. Blood, (2000). 96, (Abstr. 389)
Song, WJ, Sullivan, MG, Legare, RD, Hutchings, S, Tan, X, Kufrin, D, Ratajczak, J, Resende, IC, Haworth, C, Hock, R, Loh, M, Felix, C, Roy, DC, Busque, L, Kurnit, D, Willman, C, Gewirtz, AM, Speck, NA, Bushweller, JH, Li, FP, Gardiner, K, Poncz, M, Maris, JM & Gilliland, DG Haploinsufficiency of CBFA2 causes familial thrombocytopenia with propensity to develop acute myelogenous leukaemia. Nat Genet, (1999). 23, 166–175.
Niini, T, Kanerva, J, Vettenranta, K, Saarinen-Pihkala, UM & Knuutila, S AML1 gene amplification: a novel finding in childhood acute lymphoblastic leukemia. Haematologica, (2000). 85, 362–366.
Dal Cin, P, Atkins, L, Ford, C, Ariyanayagam, S, Armstrong, SA, George, R, Cleary, A & Morton, CC Amplification of AML1 in childhood acute lymphoblastic leukemias. Genes Chromosomes Cancer, (2001). 30, 407–409.
Streubel, B, Valent, P, Lechner, K & Fonatsch, C Amplification of the AML1(CBFA2) gene on ring chromosomes in a patient with acute myeloid leukemia and a constitutional ring chromosome 21. Cancer Genet Cytogenet, (2001). 124, 42–46.
Kakazu, N, Taniwaki, M, Horiike, S, Nishida, K, Tatekawa, T, Nagai, M, Takahashi, T, Akaogi, T, Inazawa, J, Ohki, M & Abe, T Combined spectral karyotyping and DAPI banding analysis of chromosome abnormalities in myelodysplastic syndrome. Genes Chromosomes Cancer, (1999). 26, 336–345.
Mathew, S, Rao, PH, Dalton, J, Downing, JR & Raimondi, SC Multicolor spectral karyotyping identifies novel translocations in childhood acute lymphoblastic leukemia. Leukemia, (2001). 15, 468–472.
Busson-Le Coniat, M, Khac, FN, Daniel, MT, Bernard, OA & Berger, R Chromosome 21 abnormalities with AML1 amplification in acute lymphoblastic leukemia. Genes Chromosomes Cancer, (2001). 32, 244–249.
Ma, SK, Wan, TS, Cheuk, AT, Fung, LF, Chan, GC, Chan, SY, Ha, SY & Chan, LC Characterization of additional genetic events in childhood acute lymphoblastic leukemia with TEL/AML1 gene fusion: a molecular cytogenetics study. Leukemia, (2001). 15, 1442–1447.
Bennett, JM, Catovsky, D, Daniel, MT, Flandrin, G, Galton, DA, Gralnick, HR & Sultan, C Proposals for the classification of the acute leukaemias. French–American–British (FAB) co-operative group. Br J Haematol, (1976). 33, 451–458.
Penther, D, Preudhomme, C, Talmant, P, Roumier, C, Godon, A, Méchinaud, F, Milpied, N, Bataille, R & Avet-Loiseau, H Amplification of aml1 gene is present in chilhood acute lymphoblastic leukemia but not in adult, and is not associated with aml1 gene mutation. Leukemia, (2002). 16, 1131–1134.
Bravo, J, Li, Z, Speck, NA & Warren, AJ The leukemia-associated AML1 (Runx1) – CBF beta complex functions as a DNA-induced molecular clamp. Nat Struct Biol, (2001). 8, 371–378.
Imai, Y, Kurokawa, M, Tanaka, K, Friedman, AD, Ogawa, S, Mitani, K, Yazaki, Y & Hirai, H TLE, the human homolog of groucho, interacts with AML1 and acts as a repressor of AML1-induced transactivation. Biochem Biophys Res Commun, (1998). 252, 582–589.
Mukouyama, Y, Chiba, N, Hara, T, Okada, H, Ito, Y, Kanamaru, R, Miyajima, A, Satake, M & Watanabe, T The AML1 transcription factor functions to develop and maintain hematogenic precursor cells in the embryonic aorta–gonad–mesonephros region. Dev Biol, (2000). 220, 27–36.
Lutterbach, B, Westendorf, JJ, Linggi, B, Isaac, S, Seto, E & Hiebert, SW A mechanism of repression by acute myeloid leukemia-1, the target of multiple chromosomal translocations in acute leukemia. J Biol Chem, (2000). 275, 651–656.
Bruckert, P, Kappler, R, Scherthan, H, Link, H, Hagmann, F & Zankl, H Double minutes and c-MYC amplification in acute myelogenous leukemia: Are they prognostic factors?. Cancer Genet Cytogenet, (2000). 120, 73–79.
Andersen, MK, Christiansen, DH, Kirchhoff, M & Pedersen-Bjergaard, J Duplication or amplification of chromosome band 11q23, including the unrearranged MLL gene, is a recurrent abnormality in therapy-related MDS and AML, and is closely related to mutation of the TP53 gene and to previous therapy with alkylating agents. Genes Chromosomes Cancer, (2001). 31, 33–41.
Kurokawa, M, Tanaka, T, Tanaka, K, Ogawa, S, Mitani, K, Yazaki, Y & Hirai, H Overexpression of the AML1 proto-oncoprotein in NIH3T3 cells leads to neoplastic transformation depending on the DNA-binding and transactivational potencies. Oncogene, (1996). 12, 883–892.
Acknowledgements
Supported by the Centre Hospitalier de Lille (PHRC 1997), and the Ligue National contre le Cancer (Comité du Nord et de l’Aisne) and the Fondation de France (Comité leucémie).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Roumier, C., Fenaux, P., Lafage, M. et al. New mechanisms of AML1 gene alteration in hematological malignancies. Leukemia 17, 9–16 (2003). https://doi.org/10.1038/sj.leu.2402766
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2402766
Keywords
This article is cited by
-
iAMP21 in acute myeloid leukemia is associated with complex karyotype, TP53 mutation and dismal outcome
Modern Pathology (2020)
-
Trisomy 21 with t(5; 11) chromosomal translocation as new unfavorable cytogenetic abnormalities in pediatric acute myeloid leukemia type M2: One case report of nine-year follow-up and literature review
Current Medical Science (2017)
-
Internal tandem duplication of FLT3 deregulates proliferation and differentiation and confers resistance to the FLT3 inhibitor AC220 by Up-regulating RUNX1 expression in hematopoietic cells
International Journal of Hematology (2016)
-
RUNX1 mutation associated with clonal evolution in relapsed pediatric acute myeloid leukemia with t(16;21)(p11;q22)
International Journal of Hematology (2014)
-
RUNX1 meets MLL: epigenetic regulation of hematopoiesis by two leukemia genes
Leukemia (2013)