Abstract
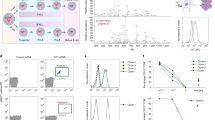
The novel fusion protein DT388IL3, composed of the catalytic and translocation domains of diphtheria toxin (DT388) fused with a Met-His linker to human interleukin 3 (IL-3), was tested for anti-leukemia efficacy in an in vivo model of differentiated human acute myeloid leukemia (AML). Six-week-old female SCID mice were irradiated with 350 cGy, inoculated 24 h later with 20 million (i.v., i.p., or s.c.) TF1 cells transfected with the v-SRC oncogene, and treated i.p., starting 24 h later, with up to five daily injections of saline, DT388IL3 (2 μg), DT388GMCSF (2 μg), DAB389IL2 (2 μg), or cytarabine (80 μg) or two weekly injections of anti-CD33-calicheamicin conjugate (5 μg). Animals were monitored twice daily, and moribund animals killed and necropsied. Control animals had a median disease-free survival (DFS) of 37 days (i.v., n = 45), 35 days (i.p., n = 20), and 21 days (s.c., n = 20), respectively. Only 5/49 (10%) of the DT388IL3 treated i.v. inoculated animals died with leukemia. Median DFS with i.v., i.p. and s.c. tumor inoculated animals was prolonged by fusion protein treatment to >120 days, 66 days and 31 days (P < 0.001, = 0.0003, and = 0.0006), respectively. Median DFS with s.c. tumor inoculated animals was also prolonged by other active anti-leukemia agents (DT388GMCSF, cytarabine and anti-CD33-calicheamicin) relative to controls by 67%, 172% and 47% (P < 0.0001, <0.0001, and =0.0004), respectively. In contrast, median DFS with s.c. tumor inoculated animals treated with DAB389IL2 non-significantly reduced by 13% relative to controls (P = 0.21). Thus, DT388IL3 fusion protein demonstrates in vivo anti-leukemia efficacy and warrants further preclinical development for treatment of chemo-resistant, IL-3 receptor positive AML patients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Schiffer, CA Acute myeloid leukemia in adults: where do we go from here?. Cancer Chemother Pharmacol, (2001). 48 (Suppl. 1), S45–S52.
Burnett, AK Evaluating the contribution of allogeneic and autologous transplantation to the management of acute myeloid leukemia in adults. Cancer Chemother Pharmacol, (2001). 48 (Suppl. 1), S53–S58.
Choe, S, Bennett, MJ, Fujii, G, Curmi, PMG, Kantardjieff, KA, Collier, RJ & Eisenberg, D The crystal structure of diphtheria toxin. Nature, (1992). 357, 216–222.
Brooke, JS & Cha, JH Molecular characterization of key diphtheria toxin: receptor interactions. Biochem. Biophys Res Commun, (2000). 275, 374–381.
Silverman, JA, Mindell, JA, Finkelstein, A, Shen, WH & Collier, RJ Mutational analysis of the helical hairpin region of diphtheria toxin transmembrane domain. J Biol Chem, (1994). 269, 22524–22532.
Bodley, JW, Dunlop, PC & Van Ness, BG Diphthamide in elongation factor 2: ADP-ribosylation, purification, and properties. Meth Enzymol, (1984). 106, 378–387.
vanderSpek, JC & Murphy, JR Fusion protein toxins based on diphtheria toxin: selective targeting of growth factor receptors of eukaryotic cells. Meth Enzymol, (2000). 327, 239–249.
Frankel, AE, Ramage, J, Latimer, A, Feely, T, Delatte, S, Hall, P, Tagge, E, Kreitman, RJ & Willingham, MC High level synthesis and maximal purification of the recombinant diphtheria fusion toxin DTGM for phase I clinical trials. Prot Exp Purif, (1999). 16, 190–201.
Frankel, AE, Powell, BL, Hall, PD, Case, LD & Kreitman, RJ Phase I trial of a novel diphtheria toxin/GM-CSF fusion protein (DT388GMCSF) for refractory or relapsed acute myeloid leukemia (AML). Clin Cancer Res, (2002). 8, 1004–1013.
Frankel, AE, Ramage, J, Gannaway, S, Kiser, M, Alexander, R, Kucera, G & Miller, MS Characterization of diphtheria fusion toxins targeted to the interleukin-3 receptor. Prot Eng, (2000). 13, 575–581.
Ailles, LE, Gerhard, B & Hogge, DE Detection and characterization of primitive malignant and normal progenitors in patients with acute myelogenous leukemia using long-term coculture with supportive feeder layers and cytokines. Blood, (1997). 90, 2555–2564.
Frankel, AE, McCubrey, J, Miller, MS, Delatte, S, Ramage, J, Kiser, M, Kucera, GL, Alexander, RL, Beran, M, Tagge, EP, Kreitman, RJ & Hogge, D Diphtheria toxin fused to human interleukin-3 is toxic to blasts from patients with myeloid leukemias. Leukemia, (2000). 14, 576–585.
Alexander, RL, Ramage, J, Kucera, GL, Caligiuri, MA & Frankel, AE High affinity interleukin-3 receptor expression on blasts from patients with acute myelogenous leukemia (AML) correlates with cytotoxicity of a diphtheria toxin/IL-3 fusion protein. Leukemia Res, (2001). 25, 875–881.
Alexander, RL, Kucera, GL, Klein, B & Frankel, AE In vitro interleukin-3 binding to leukemia cells predicts cytotoxicity of a diphtheria toxin/IL3 fusion protein. Bioconj Chem, (2000). 11, 564–568.
Feuring-Buske, M, Frankel, AE, Alexander, RL, Gerhard, B & Hogge, DE A diphtheria toxin-interleukin-3 fusion protein is cytotoxic to primitive acute myeloid leukemia (AML) progenitors but spares normal progenitors. Cancer Res, (2002). 62, 1730–1736.
Kiser, M, McCubrey, JA, Steelman, LS, Shelton, JG, Ramage, J, Alexander, RL, Kucera, GL, Pettenati, M, Willingham, MC, Miller, MS & Frankel, AE Oncogene-dependent engraftment of human myeloid leukemia cells in immunocompromised mice. Leukemia, (2001). 15, 814–818.
Blalock, WL, Weinstein-Oppenheimer, C, Chang, F, Hoyle, PE, Wang, XY, Algate, PA, Franklin, RA, Oberhaus, SM, Steelman, LS & McCubrey, JA Signal transduction, cell cycle regulatory, and anti-apoptotic pathways regulated by IL-3 in hematopoietic cells: possible sites for intervention with anti-neoplastic drugs. Leukemia, (1999). 13, 1109–1166.
Kitamura, T, Tange, T, Terasawa, T, Chiba, S, Kuwaiki, T, Miyagawa, K, Piao, YF, Miyazono, K, Urabe, A & Takaku, F Establishment and characterization of a unique human cell line that proliferates dependently on GM-CSF, IL-3 or erythropoietin. J Cell Physiol, (1989). 140, 323–334.
Vallera, DA, Seo, SY, Panoskaltsis-Mortari, A, Griffin, JD & Blazar, BR Targeting myeloid leukemia with a DT390-mIL-3 fusion immunotoxin: ex vivo and in vivo studies in mice. Prot Eng, (1999). 12, 779–785.
Sievers, EL, Larson, RA, Stadtmauer, EA, Estey, E, Lowenberg, B, Dombret, H, Karanes, C, Theobald, M, Bennett, JM, Sherman, ML, Berger, MS, Eten, CB, Loken, MR, van Dongen, JJ, Bernstein, ID & Appelbaumn, FR Efficacy and safety of gemtuzumab ozogamicin in patients with CD33-positive acute myeloid leukemia in first relapse. J Clin Oncol, (2001). 19, 3244–3254.
Acknowledgements
This work was supported by grants from the Leukemia and Lymphoma Society (Grant No. 6114–99), NIH (Grant No. R01CA76178, R21CA90550, and R01CA90263) to A Frankel, and from the Cancer Research Society of Canada and the National Cancer Institute of Canada to D Hogge.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Black, J., McCubrey, J., Willingham, M. et al. Diphtheria toxin-interleukin-3 fusion protein (DT388IL3) prolongs disease-free survival of leukemic immunocompromised mice. Leukemia 17, 155–159 (2003). https://doi.org/10.1038/sj.leu.2402744
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2402744
Keywords
This article is cited by
-
Novel strategies for targeting leukemia stem cells: sounding the death knell for blood cancer
Cellular Oncology (2017)
-
CD123 targeting oncolytic adenoviruses suppress acute myeloid leukemia cell proliferation in vitro and in vivo
Blood Cancer Journal (2014)
-
Targeting survival cascades induced by activation of Ras/Raf/MEK/ERK, PI3K/PTEN/Akt/mTOR and Jak/STAT pathways for effective leukemia therapy
Leukemia (2008)
-
Safety evaluation of DT388IL3, a diphtheria toxin/interleukin 3 fusion protein, in the cynomolgus monkey
Cancer Immunology, Immunotherapy (2005)