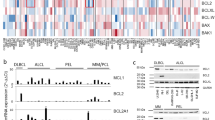
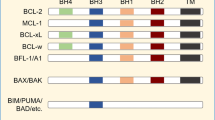
Abstract
Bcr-Abl is a constitutively active tyrosine kinase involved in the development and progression of chronic myeloid leukaemia (CML). It has been demonstrated that Bcr-Abl-positive cells can be uniquely resistant to apoptosis induced by different types of stimuli, but the mechanism by which this is achieved is not defined. In this study we have investigated how cells expressing high expression levels of Bcr-Abl may gain resistance to cytotoxic drugs. We have established cell lines expressing low and high expression levels of Bcr-Abl. Cells expressing elevated Bcr-Abl are resistant to cytotoxic drugs. In drug-sensitive 32D-parental and low Bcr-Abl expressing cells, pro-apoptotic Bcl-2 family members, Bax and Bad translocate from the cytosol to the mitochondrion following a cytotoxic insult. In contrast, high Bcr-Abl expression prevents the early translocation of these pro-apoptotic proteins to the mitochondrion, mitochondrial membrane potential is retained and caspases are inactive. We also demonstrate that IL-3 can contribute to drug resistance in low Bcr-Abl expressing cells, however, independent inhibition of IL-3 activated pathways (PI3K/AKT and Jak/STAT) does not sensitise cells to apoptosis. This study demonstrates that the subcellular translocation of Bax and Bad can be regulated by elevated Bcr-Abl expression and this may be a key event in the abrogation of an apoptotic response following a cytotoxic insult.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Keeshan K, Mills KI, Cotter TG, McKenna SL . Elevated Bcr-Abl levels are sufficient for a haematopoietic cell line to acquire a drug resistant phenotype Leukemia 2001 15: 1823–1833
Amarante-Mendes GP, McGahon AJ, Nishioka WK, Afar DE, Witte ON, Green DR . Bcl-2-independent Bcr-Abl-mediated resistance to apoptosis: protection is correlated with up regulation of Bcl-xL Oncogene 1998 16: 1383–1390
McGahon A, Bissonnette R, Schmitt M, Cotter KM, Green DR, Cotter TG . BCR-ABL maintains resistance of chronic myelogenous leukemia cells to apoptotic cell death [published erratum appears in Blood 1994 Jun 15; 83(12): 3835] Blood 1994 83: 1179–1187
Oetzel C, Jonuleit T, Gotz A, van der Kuip H, Michels H, Duyster J, Hallek M, Aulitzky WE . The tyrosine kinase inhibitor CGP 57148 (ST1 571) induces apoptosis in BCR-ABL-positive cells by down-regulating BCL-X Clin Cancer Res 2000 6: 1958–1968
Di Bacco A, Keeshan K, McKenna SL, Cotter TG . Molecular abnormalities in chronic myeloid leukemia: deregulation of cell growth and apoptosis Oncologist 2000 5: 405–415
Wilson-Rawls J, Liu J, Laneuville P, Arlinghaus RB . P210 Bcr-Abl interacts with the interleukin-3 beta c subunit and constitutively activates Jak2 Leukemia 1997 11 (Suppl. 3): 428–431
Xie S, Wang Y, Liu J, Sun T, Wilson MB, Smithgall TE, Arlinghaus RB . Involvement of Jak2 tyrosine phosphorylation in Bcr-Abl transformation Oncogene 2001 20: 6188–6195
Sakai I, Kraft AS . The kinase domain of Jak2 mediates induction of bcl-2 and delays cell death in hematopoietic cells J Biol Chem 1997 272: 12350–12358
de Groot RP, Raaijmakers JA, Lammers JW, Jove R, Koenderman L . STAT5 activation by BCR-Abl contributes to transformation of K562 leukemia cells Blood 1999 94: 1108–1112
Nieborowska-Skorska M, Wasik MA, Slupianek A, Salomoni P, Kitamura T, Calabretta B, Skorski T . Signal transducer and activator of transcription (STAT)5 activation by BCR/ABL is dependent on intact Src homology (SH)3 and SH2 domains of BCR/ABL and is required for leukemogenesis J Exp Med 1999 189: 1229–1242
Gesbert F, Griffin JD . Bcr/Abl activates transcription of the Bcl-X gene through STAT5 Blood 2000 96: 2269–2276
Skorski T, Bellacosa A, Nieborowska-Skorska M, Majewski M, Martinez R, Choi JK, Trotta R, Wlodarski P, Perrotti D, Chan TO, Wasik MA, Tsichlis PN, Calabretta B . Transformation of hematopoietic cells by BCR/ABL requires activation of a PI-3k/Akt-dependent pathway EMBO J 1997 16: 6151–6161
Mathieu AL, Gonin S, Leverrier Y, Blanquier B, Thomas J, Dantin C, Martin G, Baverel G, Marvel J . Activation of the phosphatidylinositol 3-kinase/Akt pathway protects against interleukin-3 starvation but not DNA damage-induced apoptosis J Biol Chem 2001 276: 10935–10942
Rosa Santos SC, Dumon S, Mayeux P, Gisselbrecht S, Gouilleux F . Cooperation between STAT5 and phosphatidylinositol 3-kinase in the IL-3- dependent survival of a bone marrow derived cell line Oncogene 2000 19: 1164–1172
Antonsson B, Martinou JC . The Bcl-2 protein family Exp Cell Res 2000 256: 50–57
Salomoni P, Condorelli F, Sweeney SM, Calabretta B . Versatility of BCR/ABL-expressing leukemic cells in circumventing proapoptotic BAD effects Blood 2000 96: 676–684
Salomoni P, Wasik MA, Riedel RF, Reiss K, Choi JK, Skorski T, Calabretta B . Expression of constitutively active Raf-1 in the mitochondria restores antiapoptotic and leukemogenic potential of a transformation-deficient BCR/ABL mutant J Exp Med 1998 187: 1995–2007
Neshat MS, Raitano AB, Wang HG, Reed JC, Sawyers CL . The survival function of the Bcr-Abl oncogene is mediated by Bad- dependent and -independent pathways: roles for phosphatidylinositol 3-kinase and Raf Mol Cell Biol 2000 20: 1179–1186
Majewski M, Nieborowska-Skorska M, Salomoni P, Slupianek A, Reiss K, Trotta R, Calabretta B, Skorski T . Activation of mitochondrial Raf-1 is involved in the antiapoptotic effects of Akt Cancer Res 1999 59: 2815–2819
Jia L, Patwari Y, Srinivasula SM, Newland AC, Fernandes-Alnemri T, Alnemri ES, Kelsey SM . Bax translocation is crucial for the sensitivity of leukaemic cells to etoposide-induced apoptosis Oncogene 2001 20: 4817–4826
Gonzalez-Garcia M, Perez-Ballestero R, Ding L, Duan L, Boise LH, Thompson CB, Nunez G . bcl-XL is the major bcl-x mRNA form expressed during murine development and its product localizes to mitochondria Development 1994 120: 3033–3042
Gross A, Jockel J, Wei MC, Korsmeyer SJ . Enforced dimerization of BAX results in its translocation, mitochondrial dysfunction and apoptosis EMBO J 1998 17: 3878–3885
Hsu YT, Wolter KG, Youle RJ . Cytosol-to-membrane redistribution of Bax and Bcl-X(L) during apoptosis Proc Natl Acad Sci USA 1997 94: 3668–3672
Wolter KG, Hsu YT, Smith CL, Nechushtan A, Xi XG, Youle RJ . Movement of Bax from the cytosol to mitochondria during apoptosis J Cell Biol 1997 139: 1281–1292
Oliver L, Priault M, Tremblais K, LeCabellec M, Meflah K, Manon S, Vallette FM . The substitution of the C-terminus of bax by that of bcl-xL does not affect its subcellular localization but abrogates its pro-apoptotic properties FEBS Lett 2000 487: 161–165
Nouraini S, Six E, Matsuyama S, Krajewski S, Reed JC . The putative pore-forming domain of Bax regulates mitochondrial localization and interaction with Bcl-X(L) Mol Cell Biol 2000 20: 1604–1615
Zha J, Harada H, Osipov K, Jockel J, Waksman G, Korsmeyer SJ . BH3 domain of BAD is required for heterodimerization with BCL-XL and pro-apoptotic activity J Biol Chem 1997 272: 24101–24104
Datta SR, Dudek H, Tao X, Masters S, Fu H, Gotoh Y, Greenberg ME . Akt phosphorylation of BAD couples survival signals to the cell-intrinsic death machinery Cell 1997 91: 231–241
Zha J, Harada H, Yang E, Jockel J, Korsmeyer SJ . Serine phosphorylation of death agonist BAD in response to survival factor results in binding to 14–3-3 not BCL-X(L) Cell 1996 87: 619–628
Cheng EH, Kirsch DG, Clem RJ, Ravi R, Kastan MB, Bedi A, Ueno K, Hardwick JM . Conversion of Bcl-2 to a Bax-like death effector by caspases Science 1997 278: 1966–1968
Harris MH, Thompson CB . The role of the Bcl-2 family in the regulation of outer mitochondrial membrane permeability Cell Death Differ 2000 7: 1182–1191
Shimizu S, Tsujimoto Y . Proapoptotic BH3-only Bcl-2 family members induce cytochrome c release, but not mitochondrial membrane potential loss, and do not directly modulate voltage-dependent anion channel activity Proc Natl Acad Sci USA 2000 97: 577–582
Polster BM, Kinnally KW, Fiskum G . Bh3 death domain peptide induces cell type-selective mitochondrial outer membrane permeability J Biol Chem 2001 276: 37887–37894
Bossy-Wetzel E, Newmeyer DD, Green DR . Mitochondrial cytochrome c release in apoptosis occurs upstream of DEVD-specific caspase activation and independently of mitochondrial transmembrane depolarization EMBO J 1998 17: 37–49
Martinou JC, Green DR . Breaking the mitochondrial barrier Nat Rev Mol Cell Biol 2001 2: 63–67
Guinn BA, Evely RS, Walsh V, Gilkes AF, Burnett AK, Mills KI . An in vivo and in vitro comparison of the effects of b2-a2 and b3-a2 p210BCR-ABL splice variants on murine 32D cells Leuk Lymphoma 2000 37: 393–404
Gisslinger H, Kurzrock R, Wetzler M, Tucker S, Kantarjian H, Robertson B, Talpaz M . Apoptosis in chronic myelogenous leukemia: studies of stage-specific differences Leuk Lymphoma 1997 25: 121–133
Liu RY, Fan C, Garcia R, Jove R, Zuckerman KS . Constitutive activation of the JAK2/STAT5 signal transduction pathway correlates with growth factor independence of megakaryocytic leukemic cell lines Blood 1999 93: 2369–2379
Meydan N, Grunberger T, Dadi H, Shahar M, Arpaia E, Lapidot Z, Leeder JS, Freedman M, Cohen A, Gazit A, Levitzki A, Roifman CM . Inhibition of acute lymphoblastic leukaemia by a Jak-2 inhibitor Nature 1996 379: 645–648
Tang X, Downes CP, Whetton AD, Owen-Lynch PJ . Role of phosphatidylinositol 3-kinase and specific protein kinase B isoforms in the suppression of apoptosis mediated by the Abelson protein-tyrosine kinase J Biol Chem 2000 275: 13142–13148
Sun X, Layton JE, Elefanty A, Lieschke GJ . Comparison of effects of the tyrosine kinase inhibitors AG957, AG490, and STI571 on BCR-ABL--expressing cells, demonstrating synergy between AG490 and STI571 Blood 2001 97: 2008–2015
Amarante-Mendes GP, Finucane DM, Martin SJ, Cotter TG, Salvesen GS, Green DR . Anti-apoptotic oncogenes prevent caspase-dependent and independent commitment for cell death Cell Death Differ 1998 5: 298–306
Craddock BL, Orchiston EA, Hinton HJ, Welham MJ . Dissociation of apoptosis from proliferation, protein kinase B activation, and BAD phosphorylation in interleukin-3-mediated phosphoinositide 3-kinase signaling J Biol Chem 1999 274: 10633–10640
Ilaria RL Jr, Van Etten RA . P210 and P190(BCR/ABL) induce the tyrosine phosphorylation and DNA binding activity of multiple specific STAT family members J Biol Chem 1996 271: 31704–31710
Wei MC, Zong WX, Cheng EH, Lindsten T, Panoutsakopoulou V, Ross AJ, Roth KA, MacGregor GR, Thompson CB, Korsmeyer SJ . Proapoptotic BAX and BAK: a requisite gateway to mitochondrial dysfunction and death Science 2001 292: 727–730
Yang E, Zha J, Jockel J, Boise LH, Thompson CB, Korsmeyer SJ . Bad, a heterodimeric partner for Bcl-xL and Bcl-2, displaces Bax and promotes cell death Cell 1995 80: 285–291
Nechushtan A, Smith CL, Hsu YT, Youle RJ . Conformation of the Bax C-terminus regulates subcellular location and cell death EMBO J 1999 18: 2330–2341
Murphy KM, Ranganathan V, Farnsworth ML, Kavallaris M, Lock RB . Bcl-2 inhibits Bax translocation from cytosol to mitochondria during drug-induced apoptosis of human tumor cells Cell Death Differ 2000 7: 102–111
Zhou XM, Liu Y, Payne G, Lutz RJ, Chittenden T . Growth factors inactivate the cell death promoter BAD by phosphorylation of its BH3 domain on Ser155 J Biol Chem 2000 275: 25046–25051
Acknowledgements
We are grateful to Dr Ken Mills for cell lines and constructs. We thank Dr Ruaidhri Carmody for helpful discussions and critical reading of the manuscript. This work was funded by grants from the Irish Cancer Society. We are also grateful to the Children's Leukaemia Research Project, the Health Research Board, Ireland and Enterprise Ireland for financial support.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Keeshan, K., Cotter, T. & McKenna, S. High Bcr-Abl expression prevents the translocation of Bax and Bad to the mitochondrion. Leukemia 16, 1725–1734 (2002). https://doi.org/10.1038/sj.leu.2402576
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2402576
Keywords
This article is cited by
-
The application of BH3 mimetics in myeloid leukemias
Cell Death & Disease (2021)
-
Increased acetylation of lysine 317/320 of p53 caused by BCR-ABL protects from cytoplasmic translocation of p53 and mitochondria-dependent apoptosis in response to DNA damage
Apoptosis (2012)
-
p53 stabilization induces apoptosis in chronic myeloid leukemia blast crisis cells
Leukemia (2011)
-
BCR–ABL: a multi-faceted promoter of DNA mutation in chronic myelogeneous leukemia
Leukemia (2010)
-
Antitumor effect of betulinic acid on human acute leukemia K562 cells in vitro
Journal of Huazhong University of Science and Technology [Medical Sciences] (2010)