Abstract
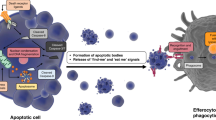
A cell is a potentially dangerous thing. In unicellular organisms, cells divide and multiply in a manner that is chiefly determined by the availability of nutritional substrates. In a multicellular organism, each cell has a distinct growth potential that is designed to subsume a role in the function of the whole body. Departure from this path to one of uncontrolled cellular proliferation leads to cancer. For this reason, evolution has endowed cells with an elaborate set of systems that cause errant cells to self-destruct. This process of cell suicide is known as apoptosis or programmed cell death and it plays a crucial role in the growth of both normal and malignant cells. In this review, we describe the mechanisms whereby programmed cell death is induced and executed. In particular, we concentrate on how anti-apoptotic signals generated by cytokines promote cell survival and how these signal transduction pathways may be involved in the pathogenesis of neoplasia. Understanding how these processes contribute to tumorigenesis may suggest new therapeutic options.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Frohlich KU, Madeo, F . Apoptosis in yeast – a monocellular organism exhibits altruistic behaviour FEBS Lett 2000 473: 6–9
Blagosklonny MV . Spotlight on apoptosis. Introduction Leukemia 2000 14: 1500–1501
Blagosklonny MV . Cell death beyond apoptosis Leukemia 2000 14: 1502–1508
Johnson DE . Programmed cell death regulation: basic mechanisms and therapeutic opportunities Leukemia 2000 14: 1340–1344
Johnson DE . Noncaspase proteases in apoptosis Leukemia 2000 14: 1695–1703
Hayflick L . Human cells and aging Sci Am 1968 218: 32–37
Arends MJ, Wyllie AH . Apoptosis: mechanisms and roles in pathology Int Rev Exp Pathol 1991 32: 223–254
Tata JR . Hormonal regulation of programmed cell death during amphibian metamorphosis Biochem Cell Biol 1994 72: 581–588
Wyllie AH . Cell death: the significance of apoptosis Int Rev Cytol 1980 68: 251–307
Scavo LM, Ertsey R, Chapin CJ, Allen L, Kitterman JA . Apoptosis in the development of rat and human fetal lungs Am J Respir Cell Mol Biol 1998 18: 21–31
Schittny JC, Djonov V, Fine A, Burri PH . Programmed cell death contributes to postnatal lung development Am J Respir Cell Mol Biol 1998 18: 786–793
Shibata S, Kyuwa S, Lee SK, Goto N . Apoptosis induced in mouse hepatitis virus-infected cells by a virus-specific CD8+ cytotoxic T-lymphocyte clone J Virol 1994 68: 7540–7545
Darmon AJ, Nicholson DW, Bleackley RC . Activation of the apoptotic protease CPP32 by cytotoxic T-cell-derived granzyme B Nature 1995 377: 4446–4448
Liebermann DA, Hoffman B, Steinman RA . Molecular controls of growth arrest and apoptosis: p53-dependent and independent pathways Oncogene 1995 11: 199–210
Green DR, Martin SJ . The killer and the executioner: how apoptosis controls malignancy Curr Opin Immunol 1995 7: 694–703
Jaattela M . Escaping cell death: survival proteins in cancer Exp Cell Res 1999 248: 30–43
Stambolic V, Mak TW, Woodgett JR . Modulation of cellular apoptotic potential: contributions to oncogenesis Oncogene 1999 18: 6094–6104
Klein G . Oncogenes and tumor suppressor genes Acta Oncol 1988 27: 427–437
Brugge JS, Steinbaugh PJ, Erikson RL . Characterization of the avian sarcoma virus protein p60src Virology 1978 91: 130–140
Downward J . The ras superfamily of small GTP-binding proteins Trends Biochem Sci 1990 15: 469–472
Rapp UR, Goldsborough MD, Mark GE, Bonner TI, Groffen J, Reynolds FH, Stephenson JR . Structure and biological activity of v-raf, a unique oncogene transduced by a retrovirus Proc Natl Acad Sci USA 1983 80: 4218–4222
Stanbridge EJ . A case for human tumor-suppressor genes Bioessays 1985 3: 252–255
Weinberg RA . Finding the anti-oncogene Sci Am 1988 259: 44–51
Harris H, Miller OJ, Klein G, Worst P, Tachibana T . Suppression of malignancy by cell fusion Nature 1969 223: 363–368
Stanbridge EJ . Suppression of malignancy in human cells Nature 1976 260: 17–20
Cohen GM . Caspases: the executioners of apoptosis Biochem J 1997 326: 1–6
Blalock WL, Weinstein-Oppenheimer C, Chang F, Hoyle PE, Wang XY, Algate PA, Franklin RA, Oberhaus SM, Steelman LS, McCubrey JA . Signal transduction, cell cycle regulatory, and anti-apoptotic pathways regulated by IL-3 in hematopoietic cells: possible sites for intervention with anti-neoplastic drugs Leukemia 1999 13: 1109–1166
Green DR . Apoptotic pathways: paper wraps stone blunts scissors Cell 2000 102: 1–4
Yuan J, Shaham S, Ledoux S, Ellis HM, Horvitz HR . The C. elegans cell death gene ced-3 encodes a protein similar to mammalian interleukin-1 beta-converting enzyme Cell 1993 75: 641–652
Thornberry NA, Bull HG, Calaycay JR, Chapman KT, Howard AD, Kostura MJ, Miller DK, Molineaux SM, Weidner JR, Aununs J et al. A novel heterodimeric cysteine protease is required for interleukin-1 beta processing in monocytes Nature 1992 356: 768–774
Cerretti DP, Kozlosky CJ, Mosley B, Nelson N, Van Ness K, Greenstreet TA, March CJ, Kronheim SR, Druck T, Cannizzaro LA, Huebner K, Black RA . Molecular cloning of the interleukin-1 beta converting enzyme Science 1992 256: 97–100
Gastman BR, Johnson DE, Whiteside TL, Rabinowich H . Caspase-mediated degradation of T-cell receptor zeta-chain Cancer Res 1999 59: 1422–1427
Kaufmann SH . Induction of endonucleolytic DNA cleavage in human acute myelogenous leukemia cells by etoposide, camptothecin, and other cytotoxic anticancer drugs: a cautionary note Cancer Res 1989 49: 5870–5878
Kaufmann SH, Desnoyers S, Ottaviano Y, Davidson NE, Poirier GG . Specific proteolytic cleavage of poly(ADP-ribose) polymerase: an early marker of chemotherapy-induced apoptosis Cancer Res 1993 53: 3976–3985
Enari M, Sakahira H, Yokoyama H, Okawa K, Iwamatsu A, Nagata S . A caspase-activated DNase that degrades DNA during apoptosis, and its inhibitor ICAD Nature 1998 391: 43–50
Song Q, Lees-Miller SP, Kumar S, Zhang N, Chan DW, Smith GCM, Jackson SP, Alnemri ES, Litwack G, Khanna KK, Lavin MF . DNA-dependent protein kinase catalytic subunit: a target for an ICE-like protease in apoptosis EMBO J 1996 15: 3238–3246
Casciola-Rosen L, Nicholson DW, Chong T, Rowan KR, Thornberry NA, Miller DK, Rosen AJ . Apopain/CPP32 cleaves proteins that are essential for cellular repair: a fundamental principle of apoptotic death J Exp Med 1996 183: 1957–1964
Lazebnik YA, Kaufmann SH, Desnoyers S, Poirier GG, Earnshaw WC . Cleavage of poly(ADP-ribose) polymerase by a proteinase with properties like ICE Nature 1994 371: 346–347
Chandler JM, Alnemri ES, Cohen GM, MacFarlane M . Activation of CPP32 and Mch3 alpha in wild-type p53-induced apoptosis Biochem J 1997 322: 19–23
Orth K, Chinnaiyan AM, Garg M, Froelich CJ, Dixit VM . The CED-3/ICE-like protease Mch2 is activated during apoptosis and cleaves the death substrate lamin A J Biol Chem 1996 271: 16443–16446
Takahashi A, Alnemri ES, Lazebnik YA, Fernandes-Alnemri T, Litwack G, Moir RD, Goldman RD, Poirier GG, Kaufmann SH, Earnshaw WC . Cleavage of lamin A by Mch2 alpha but not CPP32: multiple interleukin 1 beta-converting enzyme-related proteases with distinct substrate recognition properties are active in apoptosis Proc Natl Acad Sci USA 1996 93: 1220–1225
Martin SJ, O'Brien GA, Nishioka WK, McGahon AJ, Mahboubi A, Saido TC, Green DR . Proteolysis of fodrin (non-erythroid spectrin) during apoptosis J Biol Chem 1995 270: 6425–6428
Brancolini C, Benedetti M, Schneider C . Microfilament reorganization during apoptosis: the role of Gas2, a possible substrate for ICE-like proteases EMBO J 1995 14: 5179–5190
Mashima T, Naito M, Fujita N, Noguchi K, Tsuruo T . Identification of actin as a substrate of ICE and an ICE-like protease and involvement of an ICE-like protease but not ICE in VP-16-induced U937 apoptosis Biochem Biophys Res Commun 1995 217: 1185–1192
Kayalar C, Ord T, Testa MP, Zhong LT, Bredesen DE . Cleavage of actin by interleukin 1 beta-converting enzyme to reverse DNase I inhibition Proc Natl Acad Sci USA 1996 93: 2234–2238
Antoku K, Liu Z, Johnson DE . IL-3 withdrawal activates a CrmA-insensitive poly(ADP-ribose) polymerase cleavage enzyme in factor-dependent myeloid progenitor cells Leukemia 1998 12: 682–689
Antoku K, Liu Z, Johnson DE . Inhibition of caspase proteases by CrmA enhances the resistance of human leukemic cells to multiple chemotherapeutic agents Leukemia 1997 11: 1665–1672
Emoto Y, Manome Y, Meinhardt G, Kisaki H, Kharbanda S, Robertson M, Ghayur T, Wong WW, Kamen R, Weischelbaum RR, Kufe D . Proteolytic activation of protein kinase C-δ by an ICE-like protease in apoptotic cells EMBO J 1995 14: 6148–6156
Ghayur T, Hugunin M, Talanian RV, Ratnofsky S, Quinlan C, Emoto Y, Pandey P, Datta R, Huang Y, Kharbanda S, Allen H, Kamen R, Wong W, Kufe D . Proteolytic activation of protein kinase C-δ by an ICE/CED3-like protease induces characteristics of apoptosis J Exp Med 1996 184: 2399–2404
Datta R, Kojima H, Yoshida K, Kufe D . Caspase-3-mediated cleavage of protein kinase C-θ in induction of apoptosis J Biol Chem 1997 272: 20317–20320
Cardone MH, Salvesen GS, Widman C, Johnson G, Frisch SM . The regulation of anoikis: MEKK-1 activation requires cleavage by the caspases Cell 1997 90: 315–323
Lee N, MacDonald H, Reinhard C, Halenbeck R, Roulston A, Shi T, Williams LT . Activation of hPAK65 by caspase cleavage induces some of the morphological and biochemical changes of apoptosis Proc Natl Acad Sci USA 1997 94: 13642–13647
Widmann C, Gibson S, Johnson GL . Caspase-dependent cleavage of signaling proteins during apoptosis J Biol Chem 1998 273: 7141–7147
Browne SJ, Williams AC, Hague A, Butt AJ, Paraskeva C . Loss of APC protein expressed by human colonic epithelial cells and the appearance of a specific low-molecular-weight form is associated with apoptosis in vitro Int J Cancer 1994 59: 56–64
An B, Dou QP . Cleavage of retinoblastoma protein during apoptosis: an interleukin 1 beta-converting enzyme-like protease as candidate Cancer Res 1996 56: 438–442
Cheng EHY, Kirsch DG, Clem RJ, Ravi R, Kastan MB, Bedi A, Ueno K, Hardwick JM . Conversion of Bc1–2 to a Bax-like death effector by caspases Science 1997 278: 1966–1968
Clem R, Cheng E, Karp C, Kirsch D, Ueno K, Takahashi A, Kastan M, Griffin D, Earnshaw W, Veliuona M, Hardwick J . Modulation of cell death by Bcl-XL through caspase interaction Proc Natl Acad Sci USA 1998 95: 554–559
Yao R, Cooper GM . Requirement for phosphatidylinsoitol-3 kinase in the prevention of apoptosis by nerve growth factor Science 1995 267: 2003–2006
Arai K-I, Lee F, Miyajima A, Miyatake S, Arai N, Yokota T . Cytokines: coordinations of immune and inflammatory responses Annu Rev Biochem 1990 50: 783–836
Wang XY, McCubrey JA . Regulation of interleukin-3 expression in normal and autocrine transformed hematopoietic cells Int J Oncol 1997 10: 989–1001
Green DR, Reed JC . Mitochondria and apoptosis Science 1998 281: 1309–1316
Cai J, Yang J, Jones DP . Mitochondrial control of apoptosis: the role of cytochrome c Biochem Biophys Acta 1998 1366: 139–149
Susin SA, Zamzami N, Kroemer G . Mitochondria as regulators of apoptosis: doubt no more Biochem Biophys Acta 1998 1366: 151–165
Zou H, Henzel W, Liu X, Lutschg A, Wang X . Apaf-1, a human protein homologous to C. elegans CED-4, participates in cytochrome c-dependent activation of caspase-3 Cell 1997 90: 405–413
Li P, Nijhawan D, Budihardjo I, Srinivasula SM, Ahmad M, Alnemri ES, Wang X . Cytochrome c and dATP-dependent formation of Apaf-1/caspase-9 complex initiates an apoptotic protease cascade Cell 1997 91: 479–489
Cain K, Bratton SB, Langlais C, Walker G, Brown DG, Sun X-M, Cohen GM . Apaf-1 oligomerizes into biologically active approximately 700-kDa and inactive approximately 1.4-MDa apoptosome complexes J Biol Chem 2000 275: 6067–6070
Adams JM, Cory S . The Bcl-2 Protein family: arbiters of cell survival Science 1998 281: 1322–1326
Minn AJ, Swain RE, Ma A, Thompson CB . Recent progress on the regulation of apoptosis by Bcl-2 family members Adv Immunol 1998 70: 245–279
Gross A, McDonnell JM, Korsmeyer SJ . Bcl-2 family members and the mitochondria in apoptosis Genes Dev 1999 13: 1899–1911
Tsujimoto Y, Finger LR, Yunis J, Nowell PC, Croce CM . Cloning of the chromosome breakpoint of neoplastic B cells with the t(14;18) chromosome translocation Science 1984 226: 1097–1099
Vaux DL, Cory S, Adams JM . Bcl-2 gene promotes haemopoietic cell survival and cooperates with c-myc to immortalize pre-B cells Nature 1988 335: 440–442
Blalock WL, Moye PW, Chang F, Pearce M, Steelman LS, McMahon M, McCubrey JA . Combined effects of aberrant MEK1 activity and BCL2 overexpression on relieving the cytokine-dependency of human and murine hematopoietic cells Leukemia 2000 14: 1080–1097
McCubrey JA, Steelman LS, Moye PW, Hoyle PE, Weinstein-Oppenheimer C, Chang F, Pearce M, White MK, Franklin R, Blalock WL . Effects of deregulated Raf and MEK1 expression on the cytokine-dependency of hematopoietic cells Adv Enzyme Reg 2000 40: 305–337
Moye PW, Blalock WL, Hoyle PE, Change F, Franklin RA, Weinstein-Oppenheimer C, Pearce M, Steelman LS, McMahon M, McCubrey JA . Synergy between Raf and BCL2 in abrogating the cytokine dependency of hematopoietic cells Leukemia 2000 14: 1060–1079
McCubrey JA, May WS, Duronio V, Mufson A . Serine/threonine phosphorylation in cytokine signal transduction Leukemia 2000 14: 9–21
Vaux DL, Weissman IL, Kim SK . Prevention of programmed cell death in Caenorhabditis elegans by human bcl-2 Science 1992 258: 1955–1957
Hengartner MO, Horvitz HR . C. elegans cell survival gene ced-9 encodes a functional homolog of the mammalian proto-oncogene bcl-2 Cell 1994 76: 665–676
Oltvai ZN, Milliman CL, Korsmeyer SJ . Bcl-2 heterodimerizes in vivo with a conserved homolog, Bax, that accelerates programmed cell death Cell 1993 74: 609–619
Eskes R, Desagher S, Antonsson B, Martinou JC . Bid induces the oligomerization and insertion of Bax into the outer mitochondrial membrane Mol Cell Biol 2000 20: 929–935
Green DR . Apoptotic pathways: the roads to ruin Cell 1998 94: 695–698
Newmeyer DD, Bossy-Wetzel E, Kluck RM, Wolf BB, Beere HM, Green DR . Bcl-XL does not inhibit the function of Apaf-1 Cell Death Differ 2000 7: 402–407
Robertson LE, Plunkett W, McConnell K, Keating MJ, McDonnell TJ . Bcl-2 expression in chronic lymphocytic leukemia and its correlation with the induction of apoptosis and clinical outcome Leukemia 1996 10: 456–459
Meijerink JP . t(14;18), a journey to eternity Leukemia 1997 11: 2175–2187
Harada N, Hata H, Yoshida M, Soniki T, Nagasaki A, Kuribayashi N, Kimura T, Matsuzaki H, Mitsuya H . Expression of Bcl-2 family of proteins in fresh myeloma cells Leukemia 1998 12: 1817–1820
Puthier D, Pellat-Deceunynck C, Barille S, Robillard N, Rapp MJ, Juge-Morineau N, Harousseau JL, Bataille R, Amiot M . Differential expression of Bcl-2 in human plasma cell disorders according to proliferation status and malignancy Leukemia 1999 13: 289–294
Salomons GS, Smets LA, Verwijs-Janssen M, Hart AA, Haarman EG, Kaspers GJ, Wering EV, Der Does-Van Den Berg AV, Kamps WA . Bcl-2 family members in childhood acute lymphoblastic leukemia: relationships with features at presentation, in vitro and in vivo drug response and long-term clinical outcome Leukemia 1999 13: 1574–1580
Nuessler V, Stotzer O, Gullis E, Pelka-Fleischer R, Pogrebniak A, Gieseler F, Wilmanns W . Bcl-2, bax and bcl-xL expression in human sensitive and resistant leukemia cell lines Leukemia 1999 13: 1864–1872
Wuchter C, Karawajew L, Ruppert V, Buchner T, Schoch C, Haferlach T, Ratei R, Dorken B, Ludwig WD . Clinical significance of CD95, Bcl-2 and Bax expression and CD95 function in adult de novo acute myeloid leukemia in context of P-glycoprotein function, maturation stage, and cytogenetics Leukemia 1999 13: 1943–1953
Prokop A, Wieder T, Sturm I, Essmann F, Seeger K, Wuchter C, Ludwig WD, Henze G, Dorken B, Daniel PT . Relapse in childhood acute lymphoblastic leukemia is associated with a decrease of the Bax/Bcl-2 ratio and loss of spontaneous caspase-3 processing in vivo Leukemia 2000 14: 1606–1613
Stamatoullas A, Buchonnet G, Lepretre S, Lenain P, Lenormand B, Duval C, Callat MP, Gaulard P, Bastard C, Tilly H . De novo acute B cell leukemia/lymphoma with t(14;18) Leukemia 2000 14: 1960–1966
Winston JT, Coats SR, Wang Y, Pledger WJ . Regulation of the cell cycle machinery by oncogenic ras Oncogene 1996 12: 127–134
Barbacid M . Ras gene Annu Rev Biochem 1987 56: 779–827
Cahill MA, Janknecht R, Nordheim A . Signalling pathways: jack of all cascades Curr Biol 1996 6: 16–19
Franza BR Jr, Maruyama K, Garrels JI, Ruley HE . In vitro establishment is not a sufficient prerequisite for transformation by activated Ras oncogenes Cell 1986 44: 409–418
Hirakawa T, Ruley HE . Rescue of cells from ras oncogene-induced growth arrest by a second complementing oncogene Proc Natl Acad Sci USA 1988 85: 1519–1523
Ridley AJ, Davis JB, Stroobant P, Land H . Transforming growth factors-β1 and β2 are mitogens for rat Schwann cells J Cell Biol 1989 109: 3419–3424
Bar-Sagi D, Feramisco JR . Microinjection of the ras oncogene protein into PC12 cells induces morphological differentiation Cell 1985 42: 841–848
Benito M, Porras A, Nebreda AR, Santos E . Differentiation of 3T3-L1 fibroblasts to adipocytes induced by transfection of ras oncogenes Science 1991 253: 565–568
Serrano M, Lin AW, McCurrach ME, Beach D, Lowe S . Oncogenic ras provokes premature cell senescence associated with accumulation of p53 and p16ink4a Cell 1997 88: 593–602
Blagosklonny MV, Schulte TW, Nguyen P, Mimnaugh EG, Trepel J, Neckers L . Taxol induction of p21Waf1 and p53 requires c-raf-1 Cancer Res 1995 55: 4623–4626
Pumiglia KM, Decker SJ . Cell cycle arrest mediated by the MEK/mitogen-activated protein kinase pathway Proc Natl Acad Sci USA 1997 94: 448–452
Wood KW, Qi H, D'Arcangelo G, Armstrong RC, Roberts TM, Halegoua S . The cytoplasmic raf oncogene induces a neuronal phenotype in PC12 cells: a potential role for cellular raf kinases in neuronal growth factor signal transduction Proc Natl Acad Sci USA 1993 90: 5016–5020
Yen A, Williams M, Platko JD, Der C, Hisska M . Expression of activated Raf accelerates cell differentiation and PB protein down-regulation but not hypophosphorylation Eur J Cell Biol 1994 65: 103–113
O'Shea CC, Crompton T, Rosewell IR, Hayday AC, Owen MJ . Raf regulates positive selection Eur J Immunol 1996 26: 2350–2355
Samuels ML, McMahon M . Inhibition of platelet-derived growth factor- and epidermal growth-mediated mitogenesis and signaling in 3T3 cells expressing ΔRaf-ER, an estradiol-regulated from of Raf-1 Mol Cell Biol 1994 14: 7855–7866
McCarthy SA, Aziz N, McMahon M . Identification of immediate–early gene targets of the Raf-1 serine/threonine protein kinase using an estradiol-dependent fusion protein delta Raf-1:ER Meth Mol Biol 1997 85: 137–151
Ashkenazi A, Dixit VM . Death receptors: signaling and modulation Science 1998 281: 1305–1308
Ashkenazi A, Dixit VM . Apoptosis control by death and decoy receptors Curr Opin Cell Biol 1999 11: 255–260
Sheikh MS, Fornace AJ . Death and decoy receptors and p53-mediated apoptosis Leukemia 2000 14: 1509–1513
Boise LH, Thompson CB . Hierarchical control of lymphocyte survival Science 1996 274: 67–68
Smith CA, Farrah T, Goodwin RG . The TNF receptor superfamily of cellular and viral proteins: activation, costimulation, and death Cell 1994 76: 959–962
Tartaglia LA, Ayres TM, Wong GHW, Goeddel DV . A novel domain within the 55 kd TNF receptor signals cell death Cell 1993 74: 845–853
Nagata S . Apoptosis by death factor Cell 1997 88: 355–365
Chinnaiyan AM, O'Rourke K, Tewari M, Dixit VM . FADD, a novel death domain-containing protein, interacts with the death domain of Fas and initiates apoptosis Cell 1995 81: 505–512
Boldin MP, Mett IL, Varfolomeev EE, Chumakov I, Shemer-Avni Y, Camonis JH, Wallach D . Self-association of the ‘death domains’ of the p55 tumor necrosis factor (TNF) receptor and Fas/APO1 prompts signaling for TNF and Fas/APO1 effects J Biol Chem 1995 270: 387–391
Boldin M, Goncharov T, Goltsev Y, Wallach D . Involvement of MACH, a novel MORT1/FADD-interacting protease, in Fas/APO-1- and TNF receptor-induced cell death Cell 1996 85: 803–815
Muzio M, Chinnaiyan AM, Kischkel FC, O'Rourke K, Shevchenko A, Ni J, Scaffidi C, Bretz JD, Zhang M, Gentz R, Mann M, Krammer PH, Peter ME, Dixit VM . FLICE, a novel FADD-homologous ICE/CED-3-like protease, is recruited to the CD95 (Fas/APO-1) death-inducing signaling complex Cell 1996 85: 817–827
Pastorino JG, Tafani M, Farber JL . Tumor necrosis factor induces phosphorylation and translocation of BAD through a phosphatidylinositide-3-OH kinase-dependent pathway J Biol Chem 1999 274: 19411–19416
Pitti RM, Marsters SA, Ruppert S, Donahue CJ, Moore A, Askhenazi A . Induction of apoptosis by Apo-2 ligand, a new member of the tumor necrosis factor receptor family J Biol Chem 1996 271: 12687–12690
Wiley SR, Schooley K, Smolak PJ, Din WS, Huang CP, Nicholl JK, Sutherland GR, Smith TD, Rauch C, Smith CA et al. Identification and characterization of a new member of the TNF family that induces apoptosis Immunity 1995 3: 673–682
Deveraux QL, Takahashi R, Salvesen GS, Reed JC . X-linked IAP is a direct inhibitor of cell-death proteases Nature 1997 388: 300–304
Roy N, Deveraux QL, Takahashi R, Salvesen GS, Reed JC . The c-IAP-1 and c-IAP-2 proteins are direct inhibitors of specific caspases EMBO J 1997 16: 6914–6925
Crook NE, Clem RJ, Miller LK . An apoptosis-inhibiting baculovirus gene with a zinc finger-like motif J Virol 1993 67: 2168–2174
Miller LK . An exegesis of IAPs: salvation and surprises from BIR motifs Trends Cell Biol 1999 9: 323–328
Teodoro JG, Branton PE . Regulation of apoptosis by viral gene products J Virol 1997 71: 1739–1746
Uren AG, Coulsen EJ, Vaux DL . Conservation of baculovirus inhibitor of apoptosis repeat proteins (BIRPs) in viruses, nematodes, vertebrates and yeasts Trends Biochem Sci 1998 23: 159–162
LaCasse EC, Baird S, Korneluk RG, MacKenzie AE . The inhibitors of apoptosis (IAPs) and their emerging role in cancer Oncogene 1998 17: 3247–3259
Liston P, Roy N, Tamai K, Lefebvre C, Baird S, Cherton-Horvat G, Farahani R, McLean M, Ikeda JE, MacKenzie A, Korneluk RG . Suppression of apoptosis in mammalian cells by NAIP and a related family of IAP genes Nature 1996 379: 349–353
Ambrosini G, Adida C, Altieri DC . A novel anti-apoptosis gene, survivin, expressed in cancer and lymphoma Nat Med 1997 3: 917–921
Li F, Ambrosini G, Chu EY, Plescia J, Tognin S, Marchisio PC, Altieri DC . Control of apoptosis and mitotic spindle checkpoint by survivin Nature 1998 396: 580–584
Wang CY, Mayo MW, Korneluk RG, Goeddel DV, Baldwin AS . NF-kappaB antiapoptosis: induction of TRAF1 and TRAF2 and c-IAP1 and c-IAP2 to suppress caspase-8 inactivation Science 1998 281: 1680–1683
Freemont PS . RING for destruction? Curr Biol 2000 10: R84–R87
Yang Y, Fang S, Jensen JP, Weissman AM, Ashwell JD . Ubiquitin protein ligase activity of IAPs and their degradation in proteasomes in response to apoptotic stimuli Science 2000 288: 874–877
Du C, Fang M, Li Y, Li L, Wang X . Smac, a mitochondrial protein that promotes cytochrome c-dependent caspase activation by eliminating IAP inhibition Cell 2000 102: 33–42
Verhagen AM, Ekert PG, Pakusch M, Silke J Connoly LM, Reid GE, Moritz RL, Simpson RJ, Vaux DL . Identification of DIABLO, a mammalian protein that promotes apoptosis by binding to and antagonizing IAP proteins Cell 2000 102: 43–53
Kennedy SG, Wagner AJ, Conzen SD, Jordan J, Bellacosa A, Tsichilis PN, Hay N . The PI 3-kinase/Akt signaling pathway delivers an anti-apoptotic signal Genes Dev 1997 11: 701–703
Schlessinger J . New roles for src kinases in control of cell survival and angiogenesis Cell 2000 100: 293–296
White MK, Baireddy V, Strayer DS . Natural protection from apoptosis by surfactant protein-A in type II pneumocytes Exp Cell Res 2001 263: 183–192
Shepherd PR, Withers DJ, Siddle, K . Phosphoinositide 3-kinase: the key switch mechanism in insulin signalling Biochem J 1998 333: 471–490
Courtneidge SA, Heber A . An 81 kd protein complexed with middle T antigen and pp60c-src: a possible phosphatidylinositol kinase Cell 1987 50: 1031–1037
Auger KR, Serunian LA, Soltoff SP, Libby P, Cantley LC . PDGF-dependent tyrosine phosphorylation stimulates production of novel polyphosphoinositides in intact cells Cell 1989 57: 167–175
Sun XJ, Rothenberg P, Kahn CR, Backer JM, Araki E, Wilden PA, Cahill DA, Goldstein BJ, White MF . Structure of the insulin receptor substrate IRS-1 defines a unique signal transduction protein Nature 1991 352: 73–77
Pawson T, Gish GD . SH2 and SH3 domains: from structure to function Cell 1992 71: 359–362
Downward J . Mechanisms and consequences of activation of protein kinase B/Akt Curr Opin Cell Biol 1998 10: 262–267
Coffer PJ, Jin J, Woodgett JR . Protein kinase B (c-Akt): a multifunctional mediator of phospatidylinositol 3-kinase activation Biochem J 1998 335: 1–13
Stephens L, Anderson K, Stokoe D, Erdjument–Bromage H, Painter GF, Holmes AB, Gaffney PRJ, Reese CB, McCormick F, Tempst P, Coadwell J, Hawkins PT . Protein kinase B kinases that mediate phosphatidylinositol 3,4,5-triphosphate-dependent activation of protein kinase B Science 1998 279: 710–714
del Peso L, Gonzalez-Garcia M, Page C, Herrera R, Nunez G . Interleukin-3-induced phosphorylation of BAD through the protein kinase Akt Science 1997 278: 687–689
Datta SR, Dudek H, Tao X, Masters S, Fu H, Gotoh Y, Greenberg ME . Akt phosphorylation of BAD couples survival signals to the cell-intrinsic death machinery Cell 1997 91: 231–241
Toker A . Protein kinases as mediators of phosphoinositide 3-kinase signaling Mol Pharm 2000 57: 652–658
Cardone MH, Roy N, Stennicke HR, Salvesen GS, Franke TF, Stanbridge E, Frisch S, Reed JC . Regulation of cell death protease caspase-9 by phosphorylation Science 1998 282: 1318–1321
Rodriguez J, Chen HH, Lin SC, Lazebnik Y . Caspase phosphorylation, cell death, and species variability Science 2000 287: 1363a
Reed JC, Cardone MH, Roy N, Stennicke HR, Salvesen GS, Franke T, Stanbridge E . Caspase phosphorylation, cell death, and species variability. Response Science 2000 287: 1363a
Brunet A, Bonni A, Zigmond MJ, Lin MZ, Juo P, Hu LS, Anderson MJ, Arden KC, Blenis J, Greenberg ME . Akt promotes cell survival by phosphorylating and inhibiting a Forkhead transcription factor Cell 1999 96: 857–868
Wang R, Zagariya A, Ibarra-Sunga O, Gidea C, Ang E, Deshmukh S, Chaudhary G, Tang ED, Nunez G, Barr FG, Guan KL . Negative regulation of the forkhead transcription factor FKHR by Akt J Biol Chem 1999 274: 16741–16746
Pap M, Cooper GM . Role of glycogen synthase kinase-3 in the phosphatidylinositol 3-kinase/Akt cell survival pathway J Biol Chem 1998 273: 19929–19932
Cantley LC, Neel BG . New insights into tumor suppression: PTEN suppresses tumor formation by restraining the phosphoinositide 3-kinase/Akt pathway Proc Natl Acad Sci USA 1999 96: 4240–4245
Di Cristofano A, Pandolfi PP . The multiple roles of PTEN in tumor suppression Cell 2000 100: 387–390
Di Cristofano A, Pesce B, Cordon-Cardo C, Pandolfi PP . Pten is essential for embryonic development and tumour suppression Nat Genet 1998 19: 348–355
Liu TC, Lin PM, Chang JG, Lee JP, Chen TP, Lin SF . Mutation analysis of PTEN/MMAC1 in acute myeloid leukemia Am J Hematol 2000 63: 170–175
Sakai A, Thieblemont C, Wellmann A, Jaffe ES, Raffeld M . PTEN gene alterations in lymphoid neoplasms Blood 1998 92: 3410–3415
Butler MP, Wang SI, Chaganti RS, Parsons R, Dalla-Favera R . Analysis of PTEN mutations and deletions in B-cell non-Hodgkin's lymphomas Genes Chromosomes Cancer 1999 24: 322–327
Nakahara Y, Nagai H, Kinoshita T, Uchida T, Hatano S, Murate T, Saito H . Mutational analysis of the PTEN/MMAC1 gene in non-Hodgkin's lymphoma Leukemia 1998 12: 1277–1280
Scarisbrick JJ, Woolford AJ, Russell-Jones R, Whittaker SJ . Loss of heterozygosity on 10q and microsatellite instability in advanced stages of primary cutaneous T-cell lymphoma and possible association with homozygous deletion of PTEN Blood 2000 95: 2937–2942
Epinat JC, Gilmore TD . Diverse agents act at multiple levels to inhibit the Rel/NF-κB signal transduction pathway Oncogene 1999 18: 6896–6909
Barkett M, Gilmore TD . Control of apoptosis by RelA/NF-κB transcription factors Oncogene 18: 6910–6924
Whiteside ST, Israel A . I kappa B proteins: structure, function and regulation Semin Cancer Biol 1997 8: 75–82
Yamaoka S, Courtois G, Bessia C, Whiteside ST, Weil R, Agou F, Kirk HE, Kay RJ, Israel A . Complementation cloning of NEMO, a component of the IkappaB kinase complex essential for NF-kappaB activation Cell 1998 93: 1231–1240
Rayet B, Gelinas C . Aberrant rel/nfkb genes and activity in human cancer Oncogene 1999 18: 6938–6947
Gilmore TD . Multiple mutations contribute to the oncogenicity of the retroviral oncoprotein v-Rel Oncogene 1999 18: 6925–6937
Chen IS, Wilhelmsen KC, Temin HM . Structure and expression of c-rel, the cellular homolog to the oncogene of reticuloendotheliosis virus strain T J Virol 1983 45: 104–113
Neiman PE, Thomas SJ, Loring G . Induction of apoptosis during normal and neoplastic B-cell development in the bursa of Fabricius Proc Natl Acad Sci USA 1991 88: 5857–5861
White DW, Roy A, Gilmore TD . The v-Rel oncoprotein blocks apoptosis and proteolysis of I kappa B-alpha in transformed chicken spleen cells Oncogene 1995 10: 857–868
Sen R, Baltimore D . Inducibility of kappa immunoglobulin enhancer-binding protein Nf-kappa B by a posttranslational mechanism Cell 1986 47: 921–928
Sonenshein G . Rel/NF-kappaB transcription factors and the control of apoptosis Semin Cancer Biol 1997 8: 113–119
Foehr ED, Lin X, O'Mahony A, Geleziunas R, Bradshaw RA, Greene WC . NF-kappa B signaling promotes both cell survival and neurite process formation in nerve growth factor-stimulated PC12 cells J Neurosci 2000 20: 7556–7563
Mayo MW, Wang CY, Cogswell PC, Rogers-Graham KS, Lowe SW, Der CJ, Baldwin AS . Requirement of NF-kappaB activation to suppress p53-independent apoptosis induced by oncogenic Ras Science 1997 278: 1812–1815
Hsu H, Shu HB, Pan MG, Goeddel DV . TRADD-TRAF2 and TRADD-FADD interactions define two distinct TNF receptor 1 signal transduction pathways Cell 1996 84: 299–308
Ozes ON, Mayo LD, Gustin JA, Pfeffer SR, Pfeffer LM, Donner DB . NF-kappaB activation by tumor necrosis factor requires the Akt serine-threonine kinase Nature 1999 401: 82–85
Romashkova JA, Makarov SS . NF-kappaB is a target of Akt in anti-apoptotic PDGF signalling Nature 1999 401: 86–90
Bertrand F, Atfi A, Cadoret A, L'Allemain G, Robin H, Lascols O, Capeau J, Cherqui G . A role for nuclear factor kappaB in the antiapoptotic function of insulin J Biol Chem 1998 273: 2931–2938
Heck S, Lezoualc'h F, Engert S, Behl C . Insulin-like growth factor-1-mediated neuroprotection against oxidative stress is associated with activation of nuclear factor kappaB J Biol Chem 1999 274: 9828–9835
Foehr ED, Lin X, O'Mahony A, Geleziunas R, Bradshaw RA, Greene WC . NF-kappa B signaling promotes both cell survival and neurite process formation in nerve growth factor-stimulated PC12 cells J Neurosci 2000 20: 7556–7563
Wang CY, Mayo MW, Baldwin AS . TNF-and cancer therapy-induced apoptosis: potentiation by inhibition of NF-kappaB Science 1996 274: 784–787
Mayo MW, Baldwin AS . The transcription factor NF-κikB: control of oncogenesis and cancer therapy resistance Biochim Biophys Acta 2000 1470: M55–M62
Dasika GK, Lin SC, Zhao S, Sung P, Tomkinson A, Lee EY . DNA damage-induced cell cycle checkpoints and DNA strand break repair in development and tumorigenesis Oncogene 1999 18: 7883–7899
Kaelin WG . The p53 gene family Oncogene 1999 18: 7701–7705
Drexler HG . Review of alterations of the cyclin-dependent kinase inhibitor INK4 family p15, p16, p18 and p19 in human leukemia–lymphoma cells Leukemia 1998 12: 845–859
Agarwal ML, Taylor WR, Chernov MV, Chernova OB, Stark GR . The p53 network J Biol Chem 1998 273: 1–4
Lakin ND, Jackson SP . Regulation of p53 in response to DNA damage Oncogene 1999 18: 7644–7655
Sionov RV, Haupt Y . The cellular response to p53: the decision between life and death Oncogene 1999 18: 6145–6157
Acknowledgements
This work was supported by Public Health Services Grants DK45718 (MKW) and CA51025 (JAM).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
White, M., McCubrey, J. Suppression of apoptosis: role in cell growth and neoplasia. Leukemia 15, 1011–1021 (2001). https://doi.org/10.1038/sj.leu.2402143
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2402143
Keywords
This article is cited by
-
Hepatitis B virus regulates Raf1 expression in HepG2.2.15 cells by enhancing its promoter activity
Archives of Virology (2011)
-
Janus kinase 2 (V617F) mutation status, signal transducer and activator of transcription-3 phosphorylation and impaired neutrophil apoptosis in myelofibrosis with myeloid metaplasia
Leukemia (2006)
-
Caspase 8L, a novel inhibitory isoform of caspase 8, is associated with undifferentiated neuroblastoma
Apoptosis (2006)
-
NF-κB/Rel-mediated regulation of apoptosis in hematologic malignancies and normal hematopoietic progenitors
Leukemia (2004)
-
Requirement for the PI3K/Akt pathway in MEK1-mediated growth and prevention of apoptosis: identification of an Achilles heel in leukemia
Leukemia (2003)