Abstract
Background:
Dietary medium-chain saturated fatty acids (MC-SFAs) have been shown to reduce total body fat. Previously, we showed that MC-SFAs prevent body fat accumulation, despite weight gain. Here, we aim to explore potential molecular mechanisms underlying the protective effect of MC-SFAs on body fat gain.
Methods:
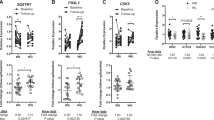
The DairyHealth study examined the long-term effects of milk protein and milk fat with a low or high content of MC-SFA. In this 12 week, randomized, double-blind, diet intervention study, participants consumed 60 g milk protein (whey or casein) and 63 g milk fat (high MC-SFA or low MC-SFA) daily in a two by two factorial design. We used microarrays to measure whole genome gene expression changes in subcutaneous adipose tissue in a subpopulation of 12 participants, 6 in the low MC-SFA+casein group and 6 in the high MC-SFA+casein group. Gene expression of several genes that were found to be changed by MC-SFAs was confirmed in the full study population using qPCR.
Results:
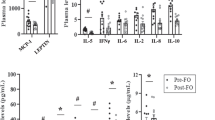
High MC-SFA resulted in an upregulation of gene expression related to citric acid cycle and oxidative phosphorylation, and a downregulation of gene expression related to complement system and inflammation.
Conclusions:
We hypothesize that the beneficial effects of MC-SFAs on prevention of fat accumulation are mediated via increased gene expression related to energy metabolism in the adipose tissue. Decreases in inflammation-related gene expression may have beneficial effects in relation to cardiometabolic diseases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Bueno NB, de Melo IV, Florencio TT, Sawaya AL . Dietary medium-chain triacylglycerols versus long-chain triacylglycerols for body composition in adults: systematic review and meta-analysis of randomized controlled trials. J Am Coll Nutr 2015; 34: 175–183.
Mumme K, Stonehouse W . Effects of medium-chain triglycerides on weight loss and body composition: a meta-analysis of randomized controlled trials. J Acad Nutr Diet 2015; 115: 249–263.
Bach AC, Babayan VK . Medium-chain triglycerides: an update. Am J Clin Nutr 1982; 36: 950–962.
Hill JO, Peters JC, Yang D, Sharp T, Kaler M, Abumrad NN et al. Thermogenesis in humans during overfeeding with medium-chain triglycerides. Metab: Clin Exp 1989; 38: 641–648.
Nosaka N, Maki H, Suzuki Y, Haruna H, Ohara A, Kasai M et al. Effects of margarine containing medium-chain triacylglycerols on body fat reduction in humans. J Atheroscler Thromb 2003; 10: 290–298.
Marten B, Pfeuffer M, Schrezenmeir J . Medium-chain triglycerides. Int Dairy J 2006; 16: 1374–1382.
Sarda P, Lepage G, Roy CC, Chessex P . Storage of medium-chain triglycerides in adipose tissue of orally fed infants. Am J Clin Nutr 1987; 45: 399–405.
Han JR, Hamilton JA, Kirkland JL, Corkey BE, Guo W . Medium-chain oil reduces fat mass and down-regulates expression of adipogenic genes in rats. Obes Res 2003; 11: 734–744.
Guo W, Lei T, Wang T, Corkey BE, Han J . Octanoate inhibits triglyceride synthesis in 3T3-L1 and human adipocytes. J Nutr 2003; 133: 2512–2518.
Bohl M, Bjornshave A, Rasmussen KV, Schioldan AG, Amer B, Larsen MK et al. Dairy proteins, dairy lipids, and postprandial lipemia in persons with abdominal obesity (DairyHealth): a 12-wk, randomized, parallel-controlled, double-blinded, diet intervention study. Am J Clin Nutr 2015; 101: 870–878.
Bohl M, Bjornshave A, Larsen MK, Gregersen S, Hermansen K . The effects of proteins and medium-chain fatty acids from milk on body composition, insulin sensitivity and blood pressure in abdominally obese adults. Eur J Clin Nutr 2017; 71: 76–82.
Weisbjerg MR, Wiking L, Kristensen NB, Lund P . Effects of supplemental dietary fatty acids on milk yield and fatty acid composition in high and medium yielding cows. J Dairy Res 2008; 75: 142–152.
Subramanian A, Tamayo P, Mootha VK, Mukherjee S, Ebert BL, Gillette MA et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci USA 2005; 102: 15545–15550.
Lin K, Kools H, de Groot PJ, Gavai AK, Basnet RK, Cheng F et al. MADMAX – Management and analysis database for multiple ~omics experiments. J Integr Bioinf 2011; 8: 160.
Dai M, Wang P, Boyd AD, Kostov G, Athey B, Jones EG et al. Evolving gene/transcript definitions significantly alter the interpretation of GeneChip data. Nucleic Acids Res 2005; 33: e175.
Irizarry RA, Bolstad BM, Collin F, Cope LM, Hobbs B, Speed TP . Summaries of Affymetrix GeneChip probe level data. Nucleic Acids Res 2003; 31: e15.
Ritchie ME, Phipson B, Wu D, Hu Y, Law CW, Shi W et al. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res 2015; 43: e47.
Merico D, Isserlin R, Stueker O, Emili A, Bader GD . Enrichment map: a network-based method for gene-set enrichment visualization and interpretation. PLoS One 2010; 5: e13984.
Wang X, Seed B . A PCR primer bank for quantitative gene expression analysis. Nucleic Acids Res 2003; 31: e154.
Wu Z, Puigserver P, Andersson U, Zhang C, Adelmant G, Mootha V et al. Mechanisms controlling mitochondrial biogenesis and respiration through the thermogenic coactivator PGC-1. Cell 1999; 98: 115–124.
Mandard S, Muller M, Kersten S . Peroxisome proliferator-activated receptor alpha target genes. Cell Mol Life Sci 2004; 61: 393–416.
Shinohara H, Wu J, Kasai M, Aoyama T . Randomly interesterified triacylglycerol containing medium- and long-chain fatty acids stimulates fatty acid metabolism in white adipose tissue of rats. Biosci Biotechnol Biochem 2006; 70: 2919–2926.
Barbu A, Hamad OA, Lind L, Ekdahl KN, Nilsson B . The role of complement factor C3 in lipid metabolism. Mol Immunol 2015; 67: 101–107.
Hertle E, Stehouwer CD, van Greevenbroek MM . The complement system in human cardiometabolic disease. Mol Immunol 2014; 61: 135–148.
Tilg H, Moschen AR . Adipocytokines: mediators linking adipose tissue, inflammation and immunity. Nat Rev Immunol 2006; 6: 772–783.
Geng S, Zhu W, Xie C, Li X, Wu J, Liang Z et al. Medium-chain triglyceride ameliorates insulin resistance and inflammation in high fat diet-induced obese mice. Eur J Nutr 2016; 55: 931–940.
Bohl M, Bjornshave A, Gregersen S, Hermansen K . Whey and casein proteins and medium-chain saturated fatty acids from milk do not increase low-grade inflammation in abdominally obese adults. Rev Diabet Stud 2016; 13: 148–157.
van Dijk SJ, Feskens EJ, Bos MB, Hoelen DW, Heijligenberg R, Bromhaar MG et al. A saturated fatty acid-rich diet induces an obesity-linked proinflammatory gene expression profile in adipose tissue of subjects at risk of metabolic syndrome. Am J Clin Nutr 2009; 90: 1656–1664.
McAllan L, Keane D, Schellekens H, Roche HM, Korpela R, Cryan JF et al. Whey protein isolate counteracts the effects of a high-fat diet on energy intake and hypothalamic and adipose tissue expression of energy balance-related genes. Br J Nutr 2013; 110: 2114–2126.
Acknowledgements
MB, SG, KH, and LAA: designed the trial; MB: conducted the trial; JCM and LAA: designed the research; JCM: conducted the research, analyzed the data and wrote the manuscript; and MB, SG, KH and LAA: critically reviewed the manuscript. This work was supported by the Danish Council for Strategic Research (DSF 0603-004193), Arla Foods Ingredients Group P/S and the Danish Dairy Research Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on International Journal of Obesity website
Rights and permissions
About this article
Cite this article
Matualatupauw, J., Bohl, M., Gregersen, S. et al. Dietary medium-chain saturated fatty acids induce gene expression of energy metabolism-related pathways in adipose tissue of abdominally obese subjects. Int J Obes 41, 1348–1354 (2017). https://doi.org/10.1038/ijo.2017.120
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2017.120
This article is cited by
-
Inflammatory crosstalk between saturated fatty acids and gut microbiota–white adipose tissue axis
European Journal of Nutrition (2023)
-
The effect of different sources of fish and camelina sativa oil on immune cell and adipose tissue mRNA expression in subjects with abnormal fasting glucose metabolism: a randomized controlled trial
Nutrition & Diabetes (2019)
-
Dietary fats and cardiometabolic disease: mechanisms and effects on risk factors and outcomes
Nature Reviews Cardiology (2019)