Abstract
OBJECTIVES: To examine sociodemographic factors associated with long-term adult weight gain and current general and central adiposity in women.
DESIGN: Cross-sectional analysis based on data from the Malmö Diet and Cancer prospective cohort Study (MDCS), Sweden.
SUBJECTS: 5464 women aged 45–73 y, who participated in the MDCS between 1994 and 1996.
MEASUREMENTS: Weight change was defined as the difference between measured weight and recalled weight at age 20. Body composition was estimated from bioelectrical impedance analysis. Waist circumference and waist-hip ratio (WHR) were indicators of central obesity. Sociodemographic factors studied included reproductive, lifestyle and socioeconomic characteristics obtained from a questionnaire.
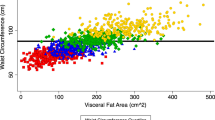
RESULTS: At current age 13% of all women were obese (BMI≥30), and nearly one-third had a body fat content of >33%. Since age 20, the majority of women (77%) had gained more than 10% of their initial weight. On average, the mean weight change was 12.6±10.0 kg during adult life. Age-adjusted group mean comparisons identified a number of significant sociodemographic variables predicting past weight gain and current obesity. In multivariate analysis, significant independent correlates of weight gain were: age, initial small body size, high parity, early menarche, non-use of hormone therapy, low self-rated health, non-smoking, no consumption of alcohol, sedentary leisure activity, past change in diet, retirement, low education, low socioeconomic status, and low socioeconomic status of origin. Many of the same variables were independently associated with current waist circumference, WHR, and percentage body fat.
CONCLUSIONS: Large weight gain during adult life and a high rate of overweight were observed in this group of Swedish women. Many sociodemographic characteristics were associated with long-term weight gain and recent obesity indices. Initial BMI, smoking, age, physical activity and early socioeconomic status accounted for most of the explained variance in weight change. The findings indicate the importance of identifying environmental determinants of both weight gain and attained fatness, as well as fat distribution.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
World Health Organization . Obesity: preventing and managing the global epidemic. Report of a WHO consultation on obesity, Geneva, 3–5 June 1997. WHO/NUT/NCD/98.1 WHO: Geneva 1998.
Rippe JM, Crossley S, Ringer R . Obesity as a chronic disease: modern medical and lifestyle management J Am Diet Assoc 1998 98: S9–S15.
French SA, Jeffery RW, Folsom AR, Williamson DF, Byers T . Relation of weight variability and intentionality of weight loss to disease history and health-related variables in a population-based sample of women aged 55–69 years Am J Epidemiol 1995 142: 1306–1314.
Magnusson C, Baron J, Persson I, Wolk A, Bergström R, Trichopoulous D, Adami HO . Body size in different periods of life and breast cancer risk in post-menopausal women Int J Cancer 1998 76: 29–34.
Barnes-Josiah D, Potter JD, Sellers TA, Himes JH . Early body size and subsequent weight gain as predictors of breast cancer incidence (Iowa, United States) Cancer Causes Control 1995 6: 112–118.
Manson JE, Willett WC, Stampfer MJ, Colditz GA, Hunter DJ, Hankinson SE, Hennekens CH, Speizer FE . Body weight and mortality among women N Engl J Med 1995 333: 677–685.
Harris TB, Savage PJ, Tell GS, Haan M, Kumanyika S, Lynch JC . Carrying the burden of cardiovascular risk in old age: associations of weight and weight change with prevalent cardiovascular disease, risk factors, and health status inthe Cardiovascular Health Study Am J Clin Nutr 1997 66: 837–844.
Tayback M, Kumanyika S, Chee E . Body weight as a risk factor in the elderly Arch Intern Med 1990 150: 1065–1072.
Bouchard C . Current understanding of the etiology of obesity: genetic and nongenetic factors Am J Clin Nutr 1991 53: 1561S–1565S.
Stunkard AJ, Harris JR, Pedersen NL, McClearn GE . The body mass index of twins who have been reared apart N Engl J Med 1990 322: 1483–1487.
Seidell JC, Flegal KM . Assessing obesity: classification and epidemiology Br Med Bull 1997 53: 238–252.
French SA, Jeffery RW, Forster JL, McGovern PG, Kelder SH, Baxter JE . Predictors of weight change over two years among a population of working adults: the Healthy Worker Project Int J Obes Relat Metab Disord 1994 18: 145–154.
Stevens J, Knapp RG, Keil JE, Verdugo RR . Changes in body weight and girths in black and while adults studied over a 25 year interval Int J Obes 1991 15: 803–808.
Colditz GA, Willett WC, Stampfer MJ, London SJ, Segal MR, Speizer FE . Patterns of weight change and their relation to diet in a cohort of healthy women Am J Clin Nutr 1990 51: 1100–1105.
Rissanen A, Heliövaara M, Aromaa A . Overweight and anthropometric changes in adulthood: a prospective study of 17 000 Finns Int J Obes 1988 12: 391–401.
Tchernof A, Poehlman ET . Effects of the menopause transition on body fatness and body fat distribution Obes Res 1998 6: 246–254.
Rissanen AM, Heliövaara M, Knekt P, Reunanen A, Aromaa A . Determinants of weight gain and overweight in adult Finns Eur J Clin Nutr 1991 45: 419–430.
Björkelund C, Lissner L, Andersson S, Lapidus L, Bengtsson C . Reproductive history in relation to relative weight and fat distribution Int J Obes Relat Metab Disord 1996 20: 213–219.
Troisi RJ, Wolf AM, Manson JE, Klingler KM, Colditz GA . Relation of body fat distribution to reproductive factors in pre- and postmenopausal women Obes Res 1995 3:: 143–151.
Klesges RC, Klesges LM, Haddocck CK, Eck LH . A longitudinal analysis of the impact of dietary intake and physical activity on weight change in adults Am J Clin Nutr 1992 55: 818–822.
Seidell JC . Dietary fat and obesity: an epidemiologic perspective Am J Clin Nutr 1998 67: 546S–550S.
Lissner L, Heitmann B . Dietary fat and obesity: evidence from epidemiology Eur J Clin Nutr 1995 49: 79–90.
Korkeila M, Kaprio J, Rissanen A, Koskenvuo M, Sörensen TIA . Predictors of major weight gain in adult Finns: stress, life satisfaction and personality traits Int J Obes Relat Metab Disord 1998 22: 949–957.
Riboli E, Elmståhl S, Saracci R, Gullberg B, Lindgärde F . The Malmö Food Study: Validity of two dietary assessment methods for measuring nutrient intake Int Journal of Epidemiology 1997 26: S161–S173.
Capella D . Descriptive tools and analysis. In: Dukes MNG (ed) Drug utilisation studies. Methods and uses, 45thedn WHO Regional Office for Europe: Copenhagen 1993 pp 55–78.
FAO, WHO and UNU . Report of joint expert consultation: Energy and protein requirements. Technical Report Series 724. WHO Geneva 1985.
Taylor HL, Jacobs Jr DR, Schucker B, Knudsen J, Leon AS, Debacker G . A questionnaire for the assessment of leisure time physical activities J Chronic Dis 1978 31: 741–755.
Statistics Sweden . Occupations in Population and Housing Census 1985. Statistics Sweden: Stockholm 1985.
Stevens J, Keil JE, Waid R, Gazes PC . Accuracy of current, 4-year, and 28-year self-reported body weight in an elderly population Am J Epidemiol 1990 132: 1156–1163.
Casey VA, Dwyer JT, Berkey CS, Coleman KA, Gardner J, Valadian I . Long-term memory of body weight and past weight satisfaction: a longitudinal follow-up study Am J Clin Nutr 1991 53: 1493–1498.
Kaye S, Folsom AR, Prineas R, Potter JD, Gabstur S . The association of body fat distribution with lifestyle and reproductive factors in a population study of postmenopausal women Int J Obes 1990 14: 583–591.
Goldberg GR, Black AE . Assessment of the validity of reported energy intakes—review and recent developments Scand J Nutr 1998 42: 6–9.
Tonkelaar den I, Seidell JC, Noord van PAH, Baanders-van Halewijn EA, Ouwehand IJ . Fat distribution in relation to age, degree of obesity, smoking habits, parity and estrogen use: a cross-sectional study in 11825 Dutch women participating in the DOM-Project Int J Obes 1990 14: 753–761.
World Health Organization . Physical status: the use and interpretation of anthropometry. WHO Technical Report Series 854. WHO: Geneva 1995, pp 312–344.
Lissner L, Bengtsson C, Lapidus L, Björkelund C . Smoking initiation and cessation in relation to body fat distribution based on data from a study of Swedish women Am J Public Health 1992 82: 273–275.
Sobal J, Stunkard AJ . Socioeconomic status and obesity: a review of the literature Psychol Bull 1989 105: 260–275.
Goldblatt PB, Moore ME, Stunkard AJ . Social factors in obesity JAMA 1965 192: 1039.
van de Mheen H, Stronks K, Looman CWN, Mackenbach JP . Does childhood socioeconomic status influence adult health through behavioral factors? Int J Epidemiol 1998 27:: 431–437.
Braddon FEM, Rodgers B, Wadsworth MEJ, Davies JMC . Onset of obesity in a 36-year birth cohort study Br Med J 1986 293: 299–303.
Willett WC, Manson JE, Stampfer MJ, Colditz GA, Rosner B, Speizer FE, Hennekens CH . Weight, Weight change, and coronary heart disease in women JAMA 1995 273: 461–465.
Acknowledgements
The authors would like to thank Elisabet Wirfält and Irene Mattisson for their helpful input and Sivert Carlsson and Ghassan Salameh for technical assistance. This work was supported by grants from the Swedish Cancer Society (2684-B93-05XAA) and the Swedish Medical Research Council (B93-39X-09534-03C).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lahmann, P., Lissner, L., Gullberg, B. et al. Sociodemographic factors associated with long-term weight gain, current body fatness and central adiposity in Swedish women. Int J Obes 24, 685–694 (2000). https://doi.org/10.1038/sj.ijo.0801219
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.ijo.0801219
Keywords
This article is cited by
-
Examining negative affect, sleep duration, and using food to cope as predictors of weight in midlife women
Journal of Behavioral Medicine (2022)
-
Life course socioeconomic position and body composition in adulthood: a systematic review and narrative synthesis
International Journal of Obesity (2021)
-
Ovarian estradiol supports sexual behavior but not energy homeostasis in female marmoset monkeys
International Journal of Obesity (2019)
-
Socioeconomic Position and Reproduction: Findings from the Australian Longitudinal Study on Women’s Health
Maternal and Child Health Journal (2018)
-
Body mass index, waist-hip ratio and risk of chronic medical condition in the elderly population: results from the Well-being of the Singapore Elderly (WiSE) Study
BMC Geriatrics (2016)