Abstract
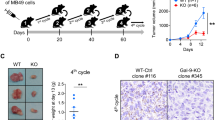
Enhanced green fluorescent protein (EGFP) is a novel marker gene product, which is readily detectable using techniques of fluorescence microscopy, flow cytometry, or macroscopic imaging. In the present studies, we have examined the immunogenicity of EGFP in murine models. A stable transfectant of the transplantable CMS4 sarcoma of BALB/c origin expressing EGFP, CMS4-EGFP-Zeo, was generated. Splenocytes harvested from mice immunized with a recombinant adenovirus expressing EGFP (Ad-EGFP) were restimulated in vitro with CMS4-EGFP-Zeo. Effector lymphocytes displayed strong cytotoxicity against CMS4-EGFP-Zeo, but not against mock-transfected CMS4-Zeo tumor cells. A number of candidate H2-Kd-binding peptides derived from the EGFP protein were chosen according to an epitope prediction program and synthesized. These peptides were tested for their ability to bind to H2-Kd molecules and stimulate IFNγ-production by splenocytes harvested from Ad-EGFP-immunized mice. Using this methodology, the peptide, HYLSTQSAL (corresponding to EGFP200–208) which strongly binds to H2-Kd molecules, was identified as a naturally occurring epitope of EGFP. These results should facilitate the use of EGFP as a model tumor antigen in BALB/c mice.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Donnelly JJ, Ulmer JB, Shiver JW, Liu MA . DNA vaccines Ann Rev Immunol 1997 15: 617–648
Tüting T, Storkus WJ, Falo LD . DNA immunization targeting the skin: molecular control of adaptive immunity J Invest Dermatol 1998 111: 183–188
Restifo NP . The new vaccines: building viruses that elicit antitumor immunity Curr Opin Immunol 1996 8: 658–663
Wang M et al. Active immunotherapy of cancer with a nonreplicating recombinant fowlpoxvirus encoding a model tumor-associated antigen J Immunol 1995 154: 4685–4692
Chen PW et al. Therapeutic antitumor response after immunization with a recombinant adenovirus encoding a model tumor-associated antigen J Immunol 1996 156: 224–231
Rao JB et al. IL-12 is an effective adjuvant to recombinant vaccinia virus-based tumor vaccines J Immunol 1996 156: 3357–3365
Condon C et al. DNA-based immunization by in vivo transfection of dendritic cells Nature Med 1996 2: 1122–1128
Tüting T, Gambotto A, Storkus WJ, De Leo AB . Co-delivery of T helper 1-biasing cytokine genes enhances the efficacy of gene gun immunization of mice: studies with the model tumor antigen β-galactosidase and the BALB/c Meth A p53 tumor-specific antigen Gene Therapy 1999 6: 629–636
Tüting T et al. Induction of tumor antigen-specific immunity using DNA immunization in mice Cancer Gene Ther 1999 6: 73–80
Chalfie M et al. Green fluorescent protein as a marker for gene expression Science 1994 263: 802–805
Zolotukhin S et al. A ‘humanized’ green fluorescent protein cDNA adapted for high-level expression in mammalian cells J Virol 1996 70: 4646–4654
Heim R, Cubitt AB, Tsien RY . Improved green fluorescence Nature 1995 373: 663–664
Stearns T . Green fluorescent protein. The green revolution Curr Biol 1995 5: 262–264
Cheng L, Fu J, Tsukamoto A, Hawley RG . Use of green fluorescent protein variants to monitor gene transfer and expression in mammalian cells Nat Biotechnol 1996 14: 606–609
Cormack BP, Valdivia RH, Falkow S . FACS-optimized mutants of the green fluorescent protein (GFP) Gene 1996 173: 33–38
Stripecke R et al. Immune response to green fluorescent protein: implications for gene therapy Gene Therapy 1999 6: 1305–1312
Parker KC, Bednarek MA, Coligan JE . Scheme for ranking potential HLA-A2 binding peptides based on independent binding of individual peptide side-chains J Immunol 1994 152: 163–175
Salter RD, Howell DN, Cresswell P . Genes regulating HLA class I antigen expression in T-B lymphoblast hybrids Immunogenetics 1985 21: 235–246
Nijman HW et al. Identification of peptide sequences that potentially trigger HLA-A2.1-restricted cytotoxic T lymphocytes Eur J Immunol 1993 23: 1215–1219
Grabbe S, Beissert S, Schwarz T, Granstein RD . Dendritic cells as initiatiors of tumor immune responses: a possibe strategy for tumor immunotherapy? Immunol Today 1995 16: 116–120
Schuler G, Steinman RM . Dendritic cells as adjuvants for immune-mediated resistance to tumors J Exp Med 1997 186: 1183–1187
Tüting T, De Leo AB, Lotze MT, Storkus WJ . Bone marrow-derived dendritic cells genetically modified to express tumor-associated antigens induce antitumor immunity in vivo Eur J Immunol 1997 27: 2702–2707
Tüting T et al. Dendritic cell-based genetic immunization in mice with a recombinant adenovirus encoding murine TRP2 induces effective anti-melanoma immunity J Gene Med 1999 6: 400–406
Hardy S et al. Construction of adenovirus vectors through Cre–lox recombination J Virol 1997 71: 1842–1849
Acknowledgements
This work was supported by National Institute of Health (NIH) Grants CA 64623 (ABD), CA59371 (PDR), a Telethon Grant (AG), and grant Tu 90/2–1 from the Deutsche Forschungsgemeinschaft (TT).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gambotto, A., Dworacki, G., Cicinnati, V. et al. Immunogenicity of enhanced green fluorescent protein (EGFP) in BALB/c mice: identification of an H2-Kd-restricted CTL epitope. Gene Ther 7, 2036–2040 (2000). https://doi.org/10.1038/sj.gt.3301335
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3301335
Keywords
This article is cited by
-
GFP Transfection Alters Protein Expression Patterns in Prostate Cancer Cells: A Proteomic Study
Journal of Fluorescence (2024)
-
Gene expression predicts dormant metastatic breast cancer cell phenotype
Breast Cancer Research (2022)
-
STING-pathway modulation to enhance the immunogenicity of adenoviral-vectored vaccines
Scientific Reports (2022)
-
A mouse model that is immunologically tolerant to reporter and modifier proteins
Communications Biology (2020)
-
Gasdermin E suppresses tumour growth by activating anti-tumour immunity
Nature (2020)