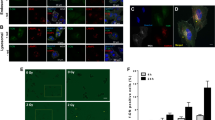
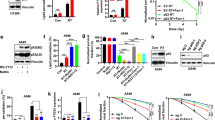
Abstract
The use of tumor-suppressor gene p53 as an anticancer therapeutic has been vigorously investigated. However, progress has met with limited success to date. Some major drawbacks are the difficulty in achieving controllable and efficient gene transfer as well as in analyzing the transferred gene expression in real time and the treatment response in a timely manner. Thus, development of novel gene transfer vector with a regulative gene expression system coupled with the reporter gene, by which transgene can be monitored simultaneously, is critical. Moreover, noninvasive imaging-based assessment of the therapeutic response to exogenous wild-type p53 gene transfer is crucial for refining treatment protocols. In this study, as a simple preclinical model, we constructed a doxycycline-regulated bidirectional vector harboring a reporter gene encoding red fluorescence protein and p53. Then, we determined the controllable and simultaneously coordinated expression of both proteins and the p53-mediated anticancer effects in vitro and in vivo. Next, we observed that cells or tumors with induced p53 overexpression exhibited decreased uptake of [14C]FDG in cellular assay and [18F]FDG in positron emission tomography (PET) imaging. Thus, by coupling with bidirectional vector, controllable p53 transfer was achieved and the capability of fluoro-2-deoxy-D-glucose (FDG)–PET to assess the therapeutic response to p53 gene therapy was evidently confirmed, which may have an impact on the improvement of p53 gene therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Mendoza-Rodriguez CA, Cerbon MA . Tumor suppressor gene p53: mechanisms of action in cell proliferation and death. Rev Invest Clin 2001; 53: 266–273.
Vogelstein B, Lane D, Levine AJ . Surfing the p53 network. Nature 2000; 408: 307–310.
Vousden KH . Outcomes of p53 activation—spoilt for choice. J Cell Sci 2006; 119: 5015–5020.
Almazov VP, Kochetkov DV, Chumakov PM . The use of p53 as a tool for human cancer therapy. Mol Biol (Mosk) 2007; 41: 947–963.
Fang B, Roth JA . Tumor-suppressing gene therapy. Cancer Biol Ther 2003; 2: S115–S121.
Nielsen LL, Maneval DC . P53 tumor suppressor gene therapy for cancer. Cancer Gene Ther 1998; 5: 52–63.
Senzer N, Nemunaitis J . A review of contusugene ladenovec (Advexin) p53 therapy. Curr Opin Mol Ther 2009; 11: 54–61.
Taghavi MH, Davoodi J . Restoration of p53 functions suppresses tumor growth of pancreatic cells with different p53 status. Cancer Biother Radiopharm 2007; 22: 322–332.
Bouvet M, Bold RJ, Lee J, Evans DB, Abbruzzese JL, Chiao PJ et al. Adenovirus-mediated wild-type p53 tumor suppressor gene therapy induces apoptosis and suppresses growth of human pancreatic cancer. Ann Surg Oncol 1998; 5: 681–688.
Jacobs A, Dubrovin M, Hewett J, Sena-Esteves M, Tan CW, Slack M et al. Functional coexpression of HSV-1 thymidine kinase and green fluorescent protein: implications for noninvasive imaging of transgene expression. Neoplasia 1999; 1: 154–161.
Gambhir SS, Barrio JR, Phelps ME, Iyer M, Namavari M, Satyamurthy N et al. Imaging adenoviral-directed reporter gene expression in living animals with positron emission tomography. Proc Natl Acad Sci USA 1999; 96: 2333–2338.
Senzer N, Nemunaitis J, Nemunaitis M, Lamont J, Gore M, Gabra H et al. p53 therapy in a patient with Li-Fraumeni syndrome. Mol Cancer Ther 2007; 6: 1478–1482.
Chung HK, Cheon GJ, Choi CW, Kim MJ, Lee SJ, Lim SM . Comparison of cellular metabolic responses of (18)F-FDG according to the effect of beta-irradiation in p53 wild and deleted cell lines. Cancer Biother Radiopharm 2007; 22: 636–643.
Wang S, Mintz A, Mochizuki K, Dorsey JF, Ackermann JM, Alavi A et al. Multimodality optical imaging and 18F-FDG uptake in wild-type p53-containing and p53-null human colon tumor xenografts. Cancer Biol Ther 2007; 6: 1649–1653.
Smith TA, Sharma RI, Thompson AM, Paulin FE . Tumor 18F-FDG incorporation is enhanced by attenuation of P53 function in breast cancer cells in vitro. J Nucl Med 2006; 47: 1525–1530.
Taylor MD, Smith PW, Brix WK, Wick MR, Theodosakis N, Swenson BR et al. Fluorodeoxyglucose positron emission tomography and tumor marker expression in non-small cell lung cancer. J Thorac Cardiovasc Surg 2009; 137: 43–48.
Taylor MD, Smith PW, Brix WK, Wick MR, Theodosakis N, Swenson BR et al. Correlations between selected tumor markers and fluorodeoxyglucose maximal standardized uptake values in esophageal cancer. Eur J Cardiothorac Surg 2009; 35: 699–705.
Gil-Rendo A, Martinez-Regueira F, Zornoza G, Garcia-Velloso MJ, Beorlegui C, Rodriguez-Spiteri N . Association between [18F]fluorodeoxyglucose uptake and prognostic parameters in breast cancer. Br J Surg 2009; 96: 166–170.
Sasaki M, Sugio K, Kuwabara Y, Koga H, Nakagawa M, Chen T et al. Alterations of tumor suppressor genes (Rb, p16, p27 and p53) and an increased FDG uptake in lung cancer. Ann Nucl Med 2003; 17: 189–196.
Aung W, Okauchi T, Sato M, Saito T, Nakagawa H, Ishihara H et al. In-vivo PET imaging of inducible D2R reporter transgene expression using [11C]FLB 457 as reporter probe in living rats. Nucl Med Commun 2005; 26: 259–268.
Sun X, Annala AJ, Yaghoubi SS, Barrio JR, Nguyen KN, Toyokuni T et al. Quantitative imaging of gene induction in living animals. Gene Therapy 2001; 8: 1572–1579.
Lowe SW, Ruley HE . Stabilization of the p53 tumor suppressor is induced by adenovirus 5 E1A and accompanies apoptosis. Genes Dev 1993; 7: 535–545.
Barak Y, Juven T, Haffner R, Oren M . mdm2 expression is induced by wild type p53 activity. EMBO J 1993; 12: 461–468.
Momand J, Zambetti GP, Olson DC, George D, Levine AJ . The mdm-2 oncogene product forms a complex with the p53 protein and inhibits p53-mediated transactivation. Cell 1992; 69: 1237–1245.
Honda R, Tanaka H, Yasuda H . Oncoprotein MDM2 is a ubiquitin ligase E3 for tumor suppressor p53. FEBS Lett 1997; 420: 25–27.
Bottger A, Bottger V, Garcia-Echeverria C, Chene P, Hochkeppel HK, Sampson W et al. Molecular characterization of the hdm2-p53 interaction. J Mol Biol 1997; 269: 744–756.
Shieh SY, Ikeda M, Taya Y, Prives C . DNA damage-induced phosphorylation of p53 alleviates inhibition by MDM2. Cell 1997; 91: 325–334.
Unger T, Sionov RV, Moallem E, Yee CL, Howley PM, Oren M et al. Mutations in serines 15 and 20 of human p53 impair its apoptotic activity. Oncogene 1999; 18: 3205–3212.
Avril NE, Weber WA . Monitoring response to treatment in patients utilizing PET. Radiol Clin North Am 2005; 43: 189–204.
Buerkle A, Weber WA . Imaging of tumor glucose utilization with positron emission tomography. Cancer Metastasis Rev 2008; 27: 545–554.
Czernin J, Phelps ME . Positron emission tomography scanning: current and future applications. Annu Rev Med 2002; 53: 89–112.
Czernin J . Clinical applications of FDG-PET in oncology. Acta Med Austriaca 2002; 29: 162–170.
Schwartzenberg-Bar-Yoseph F, Armoni M, Karnieli E . The tumor suppressor p53 down-regulates glucose transporters GLUT1 and GLUT4 gene expression. Cancer Res 2004; 64: 2627–2633.
Acknowledgements
We thank all members in our group for helpful discussions and suggestions, and the staff of the Laboratory of Animal Sciences section for animal management. This study was supported in part by a grant from the National Institute of Radiological Sciences.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Aung, W., Hasegawa, S., Koshikawa-Yano, M. et al. Noninvasive assessment of regulable transferred-p53 gene expression and evaluation of therapeutic response with FDG–PET in tumor model. Gene Ther 17, 1142–1151 (2010). https://doi.org/10.1038/gt.2010.70
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2010.70
Keywords
This article is cited by
-
Molecular Mechanism Underlying the Detection of Colorectal Cancer by 18F-2-Fluoro-2-Deoxy-d-Glucose Positron Emission Tomography
Journal of Gastrointestinal Surgery (2012)