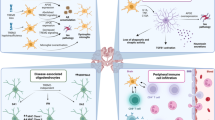
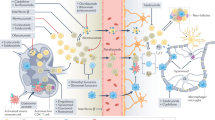
Abstract
Gene therapy for neurological, and in particular neurodegenerative, disease is now a reality. A number of early phase clinical trials have been completed and several are currently in progress. In view of this, it is critically important to evaluate the immunological risk associated with neurological gene therapy, which has clear implications for trial safety and efficacy. Moreover, it is imperative in particular to identify factors indicating potential high risk. In the light of recent advances in understanding immune regulation in the central nervous system (CNS) and with the continued development of new gene delivery vectors, this review critically assesses the current knowledge of immunobiology within the CNS in terms of likely immunological risk pertaining to viral vectors and gene therapy applications for neurodegenerative disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Galea I, Bechmann I, Perry VH . What is immune privilege (not)? Trends Immunol 2007; 28: 12–18.
Bechmann I, Galea I, Perry VH . What is the blood-brain barrier (not)? Trends Immunol 2007; 28: 5–11.
Glezer I, Simard AR, Rivest S . Neuroprotective role of the innate immune system by microglia. Neuroscience 2007; 147: 867–883.
Perry VH . Stress primes microglia to the presence of systemic inflammation: implications for environmental influences on the brain. Brain Behav Immun 2007; 21: 45–46.
Ransohoff RM, Perry VH . Microglial physiology: unique stimuli, specialized responses. Annu Rev Immunol 2009; 27: 119–145.
Barcia C, Thomas CE, Curtin JF, King GD, Wawrowsky K, Candolfi M et al. In vivo mature immunological synapses forming SMACs mediate clearance of virally infected astrocytes from the brain. J Exp Med 2006; 203: 2095–2107.
Kielian T . Overview of toll-like receptors in the CNS. Curr Top Microbiol Immunol 2009; 336: 1–14.
Aravalli RN, Hu S, Lokensgard JR . Toll-like receptor 2 signaling is a mediator of apoptosis in herpes simplex virus-infected microglia. J Neuroinflammation 2007; 4: 11.
Tang SC, Arumugam TV, Xu X, Cheng A, Mughal MR, Jo DG et al. Pivotal role for neuronal Toll-like receptors in ischemic brain injury and functional deficits. Proc Natl Acad Sci USA 2007; 104: 13798–13803.
Vercammen E, Staal J, Beyaert R . Sensing of viral infection and activation of innate immunity by toll-like receptor 3. Clin Microbiol Rev 2008; 21: 13–25.
Pedras-Vasconcelos J, Puig M, Verthelyi D . TLRs as therapeutic targets in CNS inflammation and infection. Front Biosci (Elite Ed) 2009; 1: 476–487.
van Noort JM, Bsibsi M . Toll-like receptors in the CNS: implications for neurodegeneration and repair. Prog Brain Res 2009; 175: 139–148.
Falsig J, van Beek J, Hermann C, Leist M . Molecular basis for detection of invading pathogens in the brain. J Neurosci Res 2008; 86: 1434–1447.
Barcia C, Sanderson NS, Barrett RJ, Wawrowsky K, Kroeger KM, Puntel M et al. T cells’ immunological synapses induce polarization of brain astrocytes in vivo and in vitro: a novel astrocyte response mechanism to cellular injury. PLoS One 2008; 3: e2977.
Barcia Jr C, Gomez A, Gallego-Sanchez JM, Perez-Valles A, Castro MG, Lowenstein PR et al. Infiltrating CTLs in human glioblastoma establish immunological synapses with tumorigenic cells. Am J Pathol 2009; 175: 786–798.
Weiss HA, Millward JM, Owens T . CD8+ T cells in inflammatory demyelinating disease. J Neuroimmunol 2007; 191: 79–85.
Frischer JM, Bramow S, Dal-Bianco A, Lucchinetti CF, Rauschka H, Schmidbauer M et al. The relation between inflammation and neurodegeneration in multiple sclerosis brains. Brain 2009; 132: 1175–1189.
Koedel U . Toll-like receptors in bacterial meningitis. Curr Top Microbiol Immunol 2009; 336: 15–40.
Suh HS, Brosnan CF, Lee SC . Toll-like receptors in CNS viral infections. Curr Top Microbiol Immunol 2009; 336: 63–81.
Iacobelli-Martinez M, Nemerow GR . Preferential activation of Toll-like receptor nine by CD46-utilizing adenoviruses. J Virol 2007; 81: 1305–1312.
Zhu J, Huang X, Yang Y . Innate immune response to adenoviral vectors is mediated by both Toll-like receptor-dependent and -independent pathways. J Virol 2007; 81: 3170–3180.
Cerullo V, Seiler MP, Mane V, Brunetti-Pierri N, Clarke C, Bertin TK et al. Toll-like receptor 9 triggers an innate immune response to helper-dependent adenoviral vectors. Mol Ther 2007; 15: 378–385.
Muruve DA, Petrilli V, Zaiss AK, White LR, Clark SA, Ross PJ et al. The inflammasome recognizes cytosolic microbial and host DNA and triggers an innate immune response. Nature 2008; 452: 103–107.
Saito T, Owen DM, Jiang F, Marcotrigiano J, Gale Jr M . Innate immunity induced by composition-dependent RIG-I recognition of hepatitis C virus RNA. Nature 2008; 454: 523–527.
Sabbah A, Chang TH, Harnack R, Frohlich V, Tominaga K, Dube PH et al. Activation of innate immune antiviral responses by Nod2. Nat Immunol 2009; 10: 1073–1080.
Argyris EG, Acheampong E, Wang F, Huang J, Chen K, Mukhtar M et al. The interferon-induced expression of APOBEC3G in human blood-brain barrier exerts a potent intrinsic immunity to block HIV-1 entry to central nervous system. Virology 2007; 367: 440–451.
Sullivan CS . New roles for large and small viral RNAs in evading host defences. Nat Rev Genet 2008; 9: 503–507.
Gottwein E, Cullen BR . Viral and cellular microRNAs as determinants of viral pathogenesis and immunity. Cell Host Microbe 2008; 3: 375–387.
Moens U . Silencing viral microRNA as a novel antiviral therapy? J Biomed Biotechnol 2009; 2009: 419539.
Ghosh Z, Mallick B, Chakrabarti J . Cellular versus viral microRNAs in host-virus interaction. Nucleic Acids Res 2009; 37: 1035–1048.
Wahner AD, Sinsheimer JS, Bronstein JM, Ritz B . Inflammatory cytokine gene polymorphisms and increased risk of Parkinson disease. Arch Neurol 2007; 64: 836–840.
Heneka MT, O’Banion MK . Inflammatory processes in Alzheimer's disease. J Neuroimmunol 2007; 184: 69–91.
Infante J, Garcia-Gorostiaga I, Sanchez-Juan P, Sanchez-Quintana C, Gurpegui JL, Rodriguez-Rodriguez E et al. Inflammation-related genes and the risk of Parkinson's disease: a multilocus approach. Eur J Neurol 2008; 15: 431–433.
Lassmann H . Mechanisms of inflammation induced tissue injury in multiple sclerosis. J Neurol Sci 2008; 274: 45–47.
O’Brien K, Fitzgerald DC, Naiken K, Alugupalli KR, Rostami AM, Gran B . Role of the innate immune system in autoimmune inflammatory demyelination. Curr Med Chem 2008; 15: 1105–1115.
Rezai-Zadeh K, Gate D, Town T . CNS infiltration of peripheral immune cells: D-day for neurodegenerative disease? J Neuroimmune Pharmacol 2009; 4: 462–475.
Owens T . Toll-like receptors in neurodegeneration. Curr Top Microbiol Immunol 2009; 336: 105–120.
Nimmo AJ, Vink R . Recent patents in CNS drug discovery: the management of inflammation in the central nervous system. Recent Pat CNS Drug Discov 2009; 4: 86–95.
Rivest S . Regulation of innate immune responses in the brain. Nat Rev Immunol 2009; 9: 429–439.
Frank-Cannon TC, Alto LT, McAlpine FE, Tansey MG . Does neuroinflammation fan the flame in neurodegenerative diseases? Mol Neurodegener 2009; 4: 47.
Abraham J, Johnson RW . Central inhibition of interleukin-1beta ameliorates sickness behavior in aged mice. Brain Behav Immun 2009; 23: 396–401.
Hirsch EC, Hunot S . Neuroinflammation in Parkinson's disease: a target for neuroprotection? Lancet Neurol 2009; 8: 382–397.
Reale M, Iarlori C, Thomas A, Gambi D, Perfetti B, Di Nicola M et al. Peripheral cytokines profile in Parkinson's disease. Brain Behav Immun 2009; 23: 55–63.
Banks WA, Farr SA, Morley JE, Wolf KM, Geylis V, Steinitz M . Anti-amyloid beta protein antibody passage across the blood-brain barrier in the SAMP8 mouse model of Alzheimer's disease: an age-related selective uptake with reversal of learning impairment. Exp Neurol 2007; 206: 248–256.
Del Valle J, Duran-Vilaregut J, Manich G, Camins A, Pallas M, Vilaplana J et al. Time-course of blood-brain barrier disruption in senescence-accelerated mouse prone 8 (SAMP8) mice. Int J Dev Neurosci 2009; 27: 47–52.
Landreth GE, Reed-Geaghan EG . Toll-like receptors in Alzheimer's disease. Curr Top Microbiol Immunol 2009; 336: 137–153.
Avagyan H, Goldenson B, Tse E, Masoumi A, Porter V, Wiedau-Pazos M et al. Immune blood biomarkers of Alzheimer disease patients. J Neuroimmunol 2009; 210: 67–72.
Cashman JR, Ghirmai S, Abel KJ, Fiala M . Immune defects in Alzheimer's disease: new medications development. BMC Neurosci 2008; 9 (Suppl 2): S13.
Reale M, Iarlori C, Feliciani C, Gambi D . Peripheral chemokine receptors, their ligands, cytokines and Alzheimer's disease. J Alzheimers Dis 2008; 14: 147–159.
Pelegri C, Canudas AM, Del Valle J, Casadesus G, Smith MA, Camins A et al. Increased permeability of blood-brain barrier on the hippocampus of a murine model of senescence. Mech Ageing Dev 2007; 128: 522–528.
Hafezi-Moghadam A, Thomas KL, Wagner DD . ApoE deficiency leads to a progressive age-dependent blood-brain barrier leakage. Am J Physiol Cell Physiol 2007; 292: C1256–C1262.
Godbout JP, Johnson RW . Age and neuroinflammation: a lifetime of psychoneuroimmune consequences. Immunol Allergy Clin North Am 2009; 29: 321–337.
Urbach-Ross D, Crowell B, Kusnecov AW . Relationship of varying patterns of cytokine production to the anorexic and neuroendocrine effects of repeated Staphylococcal enterotoxin A exposure. J Neuroimmunol 2008; 196: 49–59.
Kohman RA, Crowell B, Urbach-Ross D, Kusnecov AW . Influence of age on behavioral, immune and endocrine responses to the T-cell superantigen staphylococcal enterotoxin A. Eur J Neurosci 2009; 30: 1329–1338.
Henry CJ, Huang Y, Wynne A, Hanke M, Himler J, Bailey MT et al. Minocycline attenuates lipopolysaccharide (LPS)-induced neuroinflammation, sickness behavior, and anhedonia. J Neuroinflammation 2008; 5: 15.
Henry CJ, Huang Y, Wynne AM, Godbout JP . Peripheral lipopolysaccharide (LPS) challenge promotes microglial hyperactivity in aged mice that is associated with exaggerated induction of both pro-inflammatory IL-1beta and anti-inflammatory IL-10 cytokines. Brain Behav Immun 2009; 23: 309–317.
Gonzalez SC, McMenamin MM, Charlton HM, Goodman J, Lantos T, Simpson C et al. Readministration of adenoviral gene delivery to dopamine neurons. Neuroreport 2007; 18: 1609–1614.
Lee MB, McMenamin MM, Byrnes AP, Charlton HM, Wood MJ . Th1 cytokines are upregulated by adenoviral vectors in the brains of primed mice. Neuroreport 2008; 19: 1187–1192.
Lowenstein PR, Mandel RJ, Xiong WD, Kroeger K, Castro MG . Immune responses to adenovirus and adeno-associated vectors used for gene therapy of brain diseases: the role of immunological synapses in understanding the cell biology of neuroimmune interactions. Curr Gene Ther 2007; 7: 347–360.
Mandel RJ, Burger C, Snyder RO . Viral vectors for in vivo gene transfer in Parkinson's disease: properties and clinical grade production. Exp Neurol 2007; 209: 58–71.
Hadaczek P, Forsayeth J, Mirek H, Munson K, Bringas J, Pivirotto P et al. Transduction of non-human primate brain with adeno-associated virus serotype 1: vector trafficking and immune response. Hum Gene Ther 2008; 20: 225–237.
Eberling JL, Jagust WJ, Christine CW, Starr P, Larson P, Bankiewicz KS et al. Results from a phase I safety trial of hAADC gene therapy for Parkinson disease. Neurology 2008; 70: 1980–1983.
Eberling JL, Kells AP, Pivirotto P, Beyer J, Bringas J, Federoff HJ et al. Functional effects of AAV2-GDNF on the dopaminergic nigrostriatal pathway in parkinsonian rhesus monkeys. Hum Gene Ther 2009; 20: 511–518.
Marks Jr WJ, Ostrem JL, Verhagen L, Starr PA, Larson PS, Bakay RA et al. Safety and tolerability of intraputaminal delivery of CERE-120 (adeno-associated virus serotype 2-neurturin) to patients with idiopathic Parkinson's disease: an open-label, phase I trial. Lancet Neurol 2008; 7: 400–408.
Kaplitt MG, Feigin A, Tang C, Fitzsimons HL, Mattis P, Lawlor PA et al. Safety and tolerability of gene therapy with an adeno-associated virus (AAV) borne GAD gene for Parkinson's disease: an open label, phase I trial. Lancet 2007; 369: 2097–2105.
Feigin A, Kaplitt MG, Tang C, Lin T, Mattis P, Dhawan V et al. Modulation of metabolic brain networks after subthalamic gene therapy for Parkinson's disease. Proc Natl Acad Sci USA 2007; 104: 19559–19564.
Worgall S, Sondhi D, Hackett NR, Kosofsky B, Kekatpure MV, Neyzi N et al. Treatment of late infantile neuronal ceroid lipofuscinosis by CNS administration of a serotype 2 adeno-associated virus expressing CLN2 cDNA. Hum Gene Ther 2008; 19: 463–474.
Valori CF, Ning K, Wyles M, Azzouz M . Development and applications of non-HIV-based lentiviral vectors in neurological disorders. Curr Gene Ther 2008; 8: 406–418.
Dissen GA, Lomniczi A, Neff TL, Hobbs TR, Kohama SG, Kroenke CD et al. In vivo manipulation of gene expression in non-human primates using lentiviral vectors as delivery vehicles. Methods 2009; 49: 70–77.
Lundberg C, Bjorklund T, Carlsson T, Jakobsson J, Hantraye P, Deglon N et al. Applications of lentiviral vectors for biology and gene therapy of neurological disorders. Curr Gene Ther 2008; 8: 461–473.
Pluta K, Kacprzak MM . Use of HIV as a gene transfer vector. Acta Biochim Pol 2009; 56: 531–595.
Limberis MP, Bell CL, Heath J, Wilson JM . Activation of transgene-specific T cells following lentivirus-mediated gene delivery to mouse lung. Mol Ther 2009; 18: 143–150.
He Y, Falo Jr LD . Lentivirus as a potent and mechanistically distinct vector for genetic immunization. Curr Opin Mol Ther 2007; 9: 439–446.
Lopes L, Dewannieux M, Gileadi U, Bailey R, Ikeda Y, Whittaker C et al. Immunization with a lentivector that targets tumor antigen expression to dendritic cells induces potent CD8+ and CD4+ T-cell responses. J Virol 2008; 82: 86–95.
Liu Y, Peng Y, Mi M, Guevara-Patino J, Munn DH, Fu N et al. Lentivector immunization stimulates potent CD8 T cell responses against melanoma self-antigen tyrosinase-related protein 1 and generates antitumor immunity in mice. J Immunol 2009; 182: 5960–5969.
Barcia C, Jimenez-Dalmaroni M, Kroeger KM, Puntel M, Rapaport AJ, Larocque D et al. One-year expression from high-capacity adenoviral vectors in the brains of animals with pre-existing anti-adenoviral immunity: clinical implications. Mol Ther 2007; 15: 2154–2163.
Peden CS, Manfredsson FP, Reimsnider SK, Poirier AE, Burger C, Muzyczka N et al. Striatal readministration of rAAV vectors reveals an immune response against AAV2 capsids that can be circumvented. Mol Ther 2009; 17: 524–537.
Nagahara AH, Bernot T, Moseanko R, Brignolo L, Blesch A, Conner JM et al. Long-term reversal of cholinergic neuronal decline in aged non-human primates by lentiviral NGF gene delivery. Exp Neurol 2009; 215: 153–159.
Xiong W, Candolfi M, Kroeger KM, Puntel M, Mondkar S, Larocque D et al. Immunization against the transgene but not the TetON switch reduces expression from gutless adenoviral vectors in the brain. Mol Ther 2008; 16: 343–351.
Madsen D, Cantwell ER, O’Brien T, Johnson PA, Mahon BP . AAV-2 induces cell mediated immune responses directed against multiple epitopes of the capsid protein VP1. J Gen Virol 2009; 90 (Pt 11): 2622–2633.
Seiler MP, Cerullo V, Lee B . Immune response to helper dependent adenoviral mediated liver gene therapy: challenges and prospects. Curr Gene Ther 2007; 7: 297–305.
Malecki M, Wozniak A, Janik P . [Adeno-associated viruses (AAV)]. Postepy Biochem 2008; 54: 57–63.
Brunetti-Pierri N, Ng P . Progress and prospects: gene therapy for genetic diseases with helper-dependent adenoviral vectors. Gene Therapy 2008; 15: 553–560.
Segura MM, Alba R, Bosch A, Chillon M . Advances in helper-dependent adenoviral vector research. Curr Gene Ther 2008; 8: 222–235.
Zaiss AK, Machado HB, Herschman HR . The influence of innate and pre-existing immunity on adenovirus therapy. J Cell Biochem 2009; 108: 778–790.
Taymans JM, Vandenberghe LH, Haute CV, Thiry I, Deroose CM, Mortelmans L et al. Comparative analysis of adeno-associated viral vector serotypes 1, 2, 5, 7, and 8 in mouse brain. Hum Gene Ther 2007; 18: 195–206.
Howard DB, Powers K, Wang Y, Harvey BK . Tropism and toxicity of adeno-associated viral vector serotypes 1, 2, 5, 6, 7, 8, and 9 in rat neurons and glia in vitro. Virology 2008; 372: 24–34.
Lawlor PA, Bland RJ, Mouravlev A, Young D, During MJ . Efficient gene delivery and selective transduction of glial cells in the mammalian brain by AAV serotypes isolated from nonhuman primates. Mol Ther 2009; 17: 1692–1702.
Klein RL, Dayton RD, Tatom JB, Henderson KM, Henning PP . AAV8, 9, Rh10, Rh43 vector gene transfer in the rat brain: effects of serotype, promoter and purification method. Mol Ther 2008; 16: 89–96.
Koerber JT, Klimczak R, Jang JH, Dalkara D, Flannery JG, Schaffer DV . Molecular evolution of adeno-associated virus for enhanced glial gene delivery. Mol Ther 2009; 17: 2088–2095.
Jacquet BV, Patel M, Iyengar M, Liang H, Therit B, Salinas-Mondragon R et al. Analysis of neuronal proliferation, migration and differentiation in the postnatal brain using equine infectious anemia virus-based lentiviral vectors. Gene Therapy 2009; 16: 1021–1033.
Kelly EJ, Russell SJ . MicroRNAs and the regulation of vector tropism. Mol Ther 2009; 17: 409–416.
Stack J, Bowie AG . Characterisation of viral proteins that inhibit Toll-like receptor signal transduction. Methods Mol Biol 2009; 517: 217–235.
Brown BD, Naldini L . Exploiting and antagonizing microRNA regulation for therapeutic and experimental applications. Nat Rev Genet 2009; 10: 578–585.
Benlahrech A, Harris J, Meiser A, Papagatsias T, Hornig J, Hayes P et al. Adenovirus vector vaccination induces expansion of memory CD4 T cells with a mucosal homing phenotype that are readily susceptible to HIV-1. Proc Natl Acad Sci USA 2009; 106: 19940–19945.
Janson C, McPhee S, Bilaniuk L, Haselgrove J, Testaiuti M, Freese A et al. Clinical protocol. Gene therapy of Canavan disease: AAV-2 vector for neurosurgical delivery of aspartoacylase gene (ASPA) to the human brain. Hum Gene Ther 2002; 13: 1391–1412.
McPhee SW, Janson CG, Li C, Samulski RJ, Camp AS, Francis J et al. Immune responses to AAV in a phase I study for Canavan disease. J Gene Med 2006; 8: 577–588
Luo J, Kaplitt MG, Fitzsimons HL, Zuzga DS, Liu Y, Oshinsky ML et al. Subthalamic GAD gene therapy in a Parkinson's disease rat model. Science 2002; 298: 425–429.
Acknowledgements
We thank members of the Wood laboratory for critical input and in particular Yiqi Seow for help in the preparation of Figure 1. MJAW receives funding from the Medical Research Council UK and the Parkinson's Disease Society.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
McMenamin, M., Wood, M. Progress and prospects: Immunobiology of gene therapy for neurodegenerative disease: prospects and risks. Gene Ther 17, 448–458 (2010). https://doi.org/10.1038/gt.2010.2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2010.2
Keywords
This article is cited by
-
Direct reprogramming of mouse fibroblasts into neural cells via Porphyra yezoensis polysaccharide based high efficient gene co-delivery
Journal of Nanobiotechnology (2017)
-
Quantification of HSV-1-mediated expression of the ferritin MRI reporter in the mouse brain
Gene Therapy (2013)
-
Gene Transfer to the CNS Is Efficacious in Immune-primed Mice Harboring Physiologically Relevant Titers of Anti-AAV Antibodies
Molecular Therapy (2012)