Abstract
Data sources
PubMed Medline, Embase, LILACS, and reference lists of potential eligible studies.
Study selection
Prospective control trials, cohort and case series analysing results on at least 20 patients treated with ARD therapy (IV or orally). Studies reporting a protocol used for dental extraction in patients on ARD; studies reporting data on medically related osteonecrosis of the jaw after dental extraction.
Data extraction and synthesis
Two review authors independently assessed titles and abstracts for each article identified by the searches in order to decide whether the article was likely to be relevant. After the final selection and before the analyses, authors rated the quality of 13 studies according to a specific study-design-related checklist for each type of study.
Results
Data from 2,566 participants (2098 women and 468 men) were available from 13 selected studies [nine case series, two cohort studies, two randomised clinical trials]. Regarding the mode of administration of bisphosphonates, 21.2% and 79.9% were intravenous (IV) or oral (PO) respectively. The occurrence of MRONJ is statistically higher (P<0.0001) among oncologic patients treated with IV ADR (3.2%, CI=1,7-4,7%) than osteoporotic patients treated with PO ADR (0.15%, CI=0,0-0,36). Alveolectomy compared to non-alveolectomy procedure (P=0.028) and the use of biologic membranes (P=0.015) seems to attenuate the risk of MRONJ after dental extractions. Type of intention was not associated with MRONJ (P=0.32) in both adult IV and PO.
Conclusions
The authors recommend considering an adjusted protocol for dental extractions in oncological patients taking ARDs to reduce the occurrence of medication-related osteonecrosis of the jaw (MRONJ).
Similar content being viewed by others
Commentary
Treatment of bone loss in cancer and non-cancer patients has improved over the years because of the use of ARD. The first cases of jaw osteonecrosis in patients taking these medications were reported in 2003. Since then multiple approaches have been developed following the evolving understanding of the condition and risk factors. Numerous studies helped us learn of the incidence and prevalence also indicate that patients are at higher risk of developing MRONJ when using IV or oral ARD for more than five years. Some patients go on to develop MRONJ without dental treatment while some will it develop after a surgical procedure.
This review focused on MRONJ as one of the complications of the use of bisphosphonates in oncologic and osteoporotic patients after dental extractions. The main question of the study was clearly described and targeted the occurrence rate and the risk indicators of MRONJ in patients treated with Antiresorptive Drugs - ARD.
The systematic review was well organised and guided by PRISMA guidelines. The authors searched potential papers in three online databases followed by hand searching of the references. All steps of the review were clearly described. Two investigators independently screened and assessed for inclusion. Ultimately, 13 studies were included in the review: two RCTs, nine case series and two cohorts. Quality of the included studies was evaluated with a checklist that, according to the authors, is designed for each study design. A score of ≥7/11 was required from inclusion.
The authors separately performed a meta-analysis on the proportion of MRONJ for oncologic patients treated intravenously, and for osteoporotic patients treated orally. In addition, they compared the surgical techniques and protocols for preventing MRONJ.
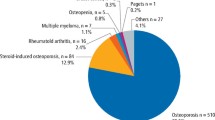
The review included 6331 surgical procedures in 2662 patients. Of those patients, 80% were treated for osteoporosis, using alendronate in 72.2% of the cases. Regarding the type of extraction eight studies reported surgical and four performed simple extractions. The proportion of MRONJ cases among oncologic patients and osteoporotic patients was 2.9% and 0.15% respectively. Regarding the relation of surgical procedure and MRONJ it seems that alveolectomy performed after extraction reduces the chance of osteonecrosis in patients treated with ARD for cancer by 6%. Additionally, the use of a biological membrane (PRGF) combined with alveolectomy causes a modest reduction of the cases of MRONJ compared to alveolectomy alone.
Caution should be used when looking at the results because different study designs were combined and a tool with unclear ability to determine the quality of observational studies besides cohorts was employed.
There is insufficient evidence to conclude the optimal surgical protocol for patients under ARD to prevent MRONJ. It seems reasonable to continue reinforcing the importance of preventive care for this target group of patients from the very beginning of treatment with ARD. The latest position finds that papers from national and international organisations are useful tools for case evaluation and treatment together with the University of Connecticut Osteonecrosis Numerical Scale1 for risk evaluation.
References
Landesberg R, Woo V, Cremers S, et al. Potential pathophysiological mechanisms in osteonecrosis of the jaw. Ann NY Acad Sci 2011; 1218: 62–79.
Author information
Authors and Affiliations
Additional information
Address for correspondence: France Lambert, Head of Clinic Department of Periodontology and Oral Surgery, Faculty of Medicine, University of Liège. Domaine du Sart Tilman Bat B-35CHU de Liège B-4000 Liège, Belgium. E-mail: france.lambert@chu.ulg.ac.be
Gaudin E, Seidel L, Bacevic M, Rompen E, Lambert F. Occurrence and risk indicators of medication-related osteonecrosis of the jaw after dental extraction: a systematic review and meta-analysis. J Clin Periodontol 2015; 42: 922–932. doi: 10.1111/jcpe.12455. Epub 2015 Nov 3.
Rights and permissions
About this article
Cite this article
Borges, C., Spivakovsky, S. Adjusted protocol for dental extractions in oncology patients taking anti-resorptive drugs may reduce occurrence of medication-related osteonecrosis of the jaw. Evid Based Dent 17, 14–15 (2016). https://doi.org/10.1038/sj.ebd.6401149
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.ebd.6401149