Abstract
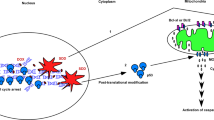
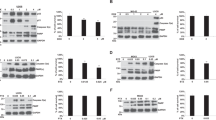
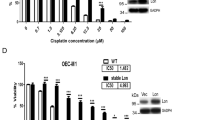
In this study, we employed a panel of cell lines to determine whether p53-dependent cell death in neuroblastoma (NB) cells is caused by apoptotic cellular function, and we further studied the molecular mechanism of apoptosis induced via the p53-dependent pathway. We obtained evidence that a type of p53-dependent stress, doxorubicin (Doxo) administration, causes accumulation of p53 in the nucleus of NB cells and phosphorylation of several serine residues in both Doxo-sensitive and -resistant cell lines. Upregulation of p53-downstream molecules in cells and upregulation of Noxa in the mitochondrial fraction were observed only in Doxo-sensitive NB cells. Significance of Noxa in the Doxo-induced NB cell death was confirmed by Noxa-knockdown experiments. Mitochondrial dysfunction, including cytochrome-c release and membrane potential disregulation, occurred and resulted in the activation of the intrinsic caspase pathway. However, in the Doxo-resistant cells, the accumulation in the nucleus and phosphorylation of p53 did not induce p53-downstream p21Cip1/Waf1 expression and the Noxa upregulation, resulting in the retention of the mitochondrial homeostasis. Taken together, these findings indicate that the p53 pathway seems to play a crucial role in NB cell death by Noxa regulation in mitochondria, and inhibition of the induction of p53-downstream effectors may regulate drug resistance of NB cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Aleyasin H, Cregan SP, Iyirhiaro G, O’Hare MJ, Callaghan SM, Slack RS et al. (2004). Nuclear factor-(kappa)B modulates the p53 response in neurons exposed to DNA damage. J Neurosci 24: 2963–2973.
Bell E, Premkumar R, Carr J, Lu X, Lovat PE, Kees UR et al. (2006). The role of MYCN in the failure of MYCN amplified neuroblastoma cell lines to G1 arrest after DNA damage. Cell Cycle 5: 2639–2647.
Danial NN, Korsmeyer SJ . (2004). Cell death: critical control points. Cell 116: 205–219.
Green DR . (2000). Apoptotic pathways: paper wraps stone blunts scissors. Cell 102: 1–4.
Hempel G, Flege S, Wurthwein G, Boos J . (2002). Peak plasma concentrations of doxorubicin in children with acute lymphoblastic leukemia or non-Hodgkin lymphoma. Cancer Chemother Pharmacol 49: 133–141.
Hijikata M, Kato N, Sato T, Kagami Y, Shimotohno K . (1990). Molecular cloning and characterization of a cDNA for a novel phorbol-12-myristate-13-acetate-responsive gene that is highly expressed in an adult T-cell leukemia cell line. J Virol 64: 4632–4639.
Hopkins-Donaldson S, Yan P, Bourloud KB, Muhlethaler A, Bodmer JL, Gross N . (2002). Doxorubicin-induced death in neuroblastoma does not involve death receptors in S-type cells and is caspase-independent in N-type cells. Oncogene 21: 6132–6137.
Hudson CD, Morris PJ, Latchman DS, Budhram-Mahadeo VS . (2005). Brn-3a transcription factor blocks p53-mediated activation of proapoptotic target genes Noxa and Bax in vitro and in vivo to determine cell fate. J Biol Chem 280: 11851–11948.
Isaacs JS, Saito S, Neckers LM . (2001). Requirement for HDM2 activity in the rapid degradation of p53 in neuroblastoma. J Biol Chem 276: 18497–18506.
Kamijo T, Aoyama T, Miyazaki J, Hashimoto T . (1993). Molecular cloning of the cDNAs for the subunits of rat mitochondrial fatty acid beta-oxidation multienzyme complex. Structural and functional relationships to other mitochondrial and peroxisomal beta-oxidation enzymes. J Biol Chem 268: 26452–26460.
Keshelava N, Zuo JJ, Chen P, Waidyaratne SN, Luna MC, Gomer CJ et al. (2001). Loss of p53 function confers high-level multidrug resistance in neuroblastoma cell lines. Cancer Res 61: 6185–6193.
Kiryu-Seo S, Hirayama T, Kato R, Kiyama H . (2005). Noxa is a critical mediator of p53-dependent motor neuron death after nerve injury in adult mouse. J Neurosci 25: 1442–1447.
Komarova EA, Chernov MV, Franks R, Wang K, Armin G, Zelnick CR et al. (1997). Transgenic mice with p53-responsive lacZ: p53 activity varies dramatically during normal development and determines radiation and drug sensitivity in vivo. EMBO J 16: 1391–1400.
Letai A, Bassik MC, Walensky LD, Sorcinelli MD, Weiler S, Korsmeyer SJ . (2002). Distinct BH3 domains either sensitize or activate mitochondrial apoptosis, serving as prototype cancer therapeutics. Cancer Cell 2: 183–192.
Lee SJ, Kim KM, Namkoong S, Kim CK, Kang YC, Lee H et al. (2005). Nitric oxide inhibition of homocysteine-induced human endothelial cell apoptosis by down-regulation of p53-dependent Noxa expression through the formation of S-nitrosohomocysteine. J Biol Chem 280: 5781–5788.
Lowe SW, Bodis S, McClatchey A, Remington L, Ruley HE, Fisher DE et al. (1994). p53 status and the efficacy of cancer therapy in vivo. Science 266: 807–810.
Machida T, Fujita T, Ooo ML, Ohira M, Isogai E, Mihara M et al. (2006). Increased expression of proapoptotic BMCC1, a novel gene with the BNIP2 and Cdc42GAP homology (BCH) domain, is associated with favorable prognosis in human neuroblastomas. Oncogene 25: 1931–1942.
Matthay KK, Perez C, Seeger RC, Brodeur GM, Shimada H, Atkinson JB et al. (1998). Successful treatment of stage III neuroblastoma based on prospective biologic staging: a Children's Cancer Group study. J Clin Oncol 16: 1256–1264.
Moll UM, LaQuaglia M, Bénard J, Riou G . (1995). Wild-type p53 protein undergoes cytoplasmic sequestration in undifferentiated neuroblastomas but not in differentiated tumors. Proc Natl Acad Sci USA 92: 4407–4411.
Moll UM, Ostermeyer AG, Haladay R, Winkfield B, Frazier M, Zambetti G . (1996). Cytoplasmic sequestration of wild-type p53 protein impairs the G1 checkpoint after DNA damage. Mol Cell Biol 16: 1126–1137.
Nakazawa Y, Kamijo T, Koike K, Noda T . (2003). ARF tumor suppressor induces mitochondria-dependent apoptosis by modulation of mitochondrial Bcl-2 family proteins. J Biol Chem 278: 27888–27895.
Obexer P, Geiger K, Ambros PF, Meister B, Ausserlechner MJ . (2007). FKHRL1-mediated expression of Noxa and Bim induces apoptosis via the mitochondria in neuroblastoma cells. Cell Death Diff 14: 534–547.
Oda E, Ohki R, Murasawa H, Nemoto J, Shibue T, Yamashita T et al. (2003). Noxa, a BH3-only member of the Bcl-2 family and candidate mediator of p53-induced apoptosis. Science 17: 1053–1058.
Oda K, Arakawa H, Tanaka T, Matsuda K, Tanikawa C, Mori T et al. (2000). p53AIP1, a potential mediator of p53-dependent apoptosis, and its regulation by Ser-46-phosphorylated p53. Cell 102: 849–862.
Ohtani S, Kagawa S, Tango Y, Umeoka T, Tokunaga N, Tsunemitsu Y et al. (2004). Quantitative analysis of p53-targeted gene expression and visualization of p53 transcriptional activity following intratumoral administration of adenoviral p53 in vivo. Mol Cancer Ther 3: 93–100.
Oren M . (1999). Regulation of the p53 tumor suppressor protein. J Biol Chem 274: 36031–36034.
Paull AC, Whikehart DR . (2005). Regulation of the p53 tumor suppressor protein. Mol Vis 11: 328–334.
Qin JZ, Stennett L, Bacon P, Bodner B, Hendrix MJ, Seftor RE et al. (2004). p53-independent NOXA induction overcomes apoptotic resistance of malignant melanomas. Mol Cancer Ther 3: 895–902.
Shen Y, White E . (2001). p53-dependent apoptosis pathways. Adv Cancer Res 82: 55–84.
Shibue T, Takeda K, Oda E, Tanaka H, Murasawa H, Takaoka A et al. (2003). Integral role of Noxa in p53-mediated apoptotic response. Genes Dev 17: 2233–2238.
Shieh SY, Ikeda M, Taya Y, Prives C . (1997). DNA damage-induced phosphorylation of p53 alleviates inhibition by MDM2. Cell 91: 325–334.
Tweddle DA, Malcolm AJ, Bown N, Pearson AD, Lunec J . (2001). Evidence for the development of p53 mutations after cytotoxic therapy in a neuroblastoma cell line. Cancer Res 61: 8–13.
Tweddle DA, Pearson AD, Haber M, Norris MD, Xue C, Flemming C et al. (2003). The p53 pathway and its inactivation in neuroblastoma. Cancer Lett 197: 93–98.
Wong HK, Fricker M, Wyttenbach A, Villunger A, Michalak EM, Strasser A et al. (2005). Mutually exclusive subsets of BH3-only proteins are activated by the p53 and c-Jun N-terminal kinase/c-Jun signaling pathways during cortical neuron apoptosis induced by arsenite. Mol Cell Biol 25: 8732–8747.
Wei MC, Zong WX, Cheng EH, Lindsten T, Panoutsakopoulou V, Ross AJ et al. (2001). Proapoptotic BAX and BAK: a requisite gateway to mitochondrial dysfunction and death. Science 292: 727–730.
Wolff A, Technau A, Ihling C, Technau-Ihling K, Erber R, Bosch FX et al. (2001). Evidence that wild-type p53 in neuroblastoma cells is in a conformation refractory to integration into the transcriptional complex. Oncogene 20: 1307–1317.
Yakovlev AG, Di Giovanni S, Wang G, Liu W, Stoica B, Faden AI . (2004). BOK and NOXA are essential mediators of p53-dependent apoptosis. J Biol Chem 279: 28367–28374.
Acknowledgements
We are deeply indebted to Professor Kenichi Koike (Department of Pediatrics, Shinshu University School of Medicine) for his excellent advice. We thank Kumiko Sakurai, Yozo Nakazawa, and Jun Miki for their technical assistance, and Daniel Mrozek, Medical English Service Inc, for editorial assistance. This work was supported by grants from the Japanese Ministry of Education, Science, Sports and Culture, Grant-in-Aid for Scientific Research (C) (contract nos: 15591098 and 17591077).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kurata, K., Yanagisawa, R., Ohira, M. et al. Stress via p53 pathway causes apoptosis by mitochondrial Noxa upregulation in doxorubicin-treated neuroblastoma cells. Oncogene 27, 741–754 (2008). https://doi.org/10.1038/sj.onc.1210672
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1210672
Keywords
This article is cited by
-
EZH2 regulates neuroblastoma cell differentiation via NTRK1 promoter epigenetic modifications
Oncogene (2018)
-
Mechanistic clues to the protective effect of chrysin against doxorubicin-induced cardiomyopathy: Plausible roles of p53, MAPK and AKT pathways
Scientific Reports (2017)
-
Low Dose of Doxorubicin Potentiates the Effect of Temozolomide in Glioblastoma Cells
Molecular Neurobiology (2017)
-
Receptor-type protein tyrosine phosphatase κ directly dephosphorylates CD133 and regulates downstream AKT activation
Oncogene (2015)
-
Discrepant NOXA (PMAIP1) transcript and NOXA protein levels: a potential Achilles’ heel in mantle cell lymphoma
Cell Death & Disease (2014)