Abstract
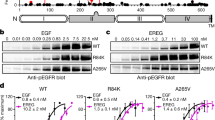
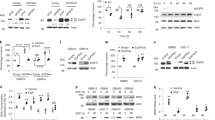
Mutations of the epidermal growth factor receptor (EGFR) gene are found at a relatively high frequency in glioma, with the most common being the de2–7 EGFR (or EGFRvIII). This mutation arises from an in-frame deletion of exons 2–7, which removes 267 amino acids from the extracellular domain of the receptor. Despite being unable to bind ligand, the de2–7 EGFR is constitutively active and imparts a significant in vivo growth advantage to glioma cells. In order to examine the signalling pathways activated by the de2–7 EGFR and its biological effects in an in vitro system, the de2–7 EGFR gene was transfected into the murine IL-3-dependent pro-B-cell line BaF/3. Expression of the de2–7 EGFR enhanced the survival of BaF/3 cells in the absence of IL-3 by reducing apoptosis in a phosphatidylinositol 3-kinase (PI3-K)-dependent manner. Interestingly, while de2–7 EGFR also enhanced proliferation of BaF/3 cells in low levels of IL-3, this effect was independent of PI3-K. Survival and proliferation were further enhanced when BaF/3 cells were cotransfected with the de2–7 and wt EGFR. This was due to heterodimerization between the de2–7 and wt EGFR leading to trans-phosphorylation of the wt EGFR. This observation is directly relevant to glioma where de2–7 and wt EGFR appear to be coexpressed. Thus, expression of de2–7 EGFR in BaF/3 cells provides an in vitro model for evaluating the signalling pathways activated by this receptor.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Batra SK, Castelino-Prabhu S, Wikstrand CJ, Zhu X, Humphrey PA, Friedman HS and Bigner DD . (1995). Cell Growth Differ., 6, 1251–1259.
Burri SH, Kim CN, Fang G, Chang BS, Perkins C, Harris W, Davis LW, Thompson CB and Bhalla KN . (1999). Int. J. Radiat. Oncol. Biol. Phys., 43, 423–430.
Chu CT, Everiss KD, Wikstrand CJ, Batra SK, Kung HJ and Bigner DD . (1997). Biochem. J., 324, 855–861.
Coffer PJ, Jin J and Woodgett JR . (1998). Biochem. J., 335, 1–13.
Datta SR, Brunet A and Greenberg ME . (1999). Genes Dev., 13, 2905–2927.
David M, Wong L, Flavell R, Thompson SA, Wells A, Larner AC and Johnson GR . (1996). J. Biol. Chem., 271, 9185–9188.
Downward J . (1998). Curr. Opin. Cell. Biol., 10, 262–267.
Ekstrand AJ, James CD, Cavenee WK, Seliger B, Pettersson RF and Collins VP . (1991). Cancer Res., 51, 2164–2172.
Garcia de Palazzo IE, Adams GP, Sundareshan P, Wong AJ, Testa JR, Bigner DD and Weiner LM . (1993). Cancer Res., 53, 3217–3220.
Grandis JR, Melhem MF, Gooding WE, Day R, Holst VA, Wagener MM, Drenning SD and Tweardy DJ . (1998). J. Natl. Cancer Inst., 90, 824–832.
Gullick WJ . (1991). Br. Med. Bull., 47, 87–98.
Hidalgo M and Rowinsky EK . (2000). Oncogene, 19, 6680–6686.
Huang HS, Nagane M, Klingbeil CK, Lin H, Nishikawa R, Ji XD, Huang CM, Gill GN, Wiley HS and Cavenee WK . (1997). J. Biol. Chem., 272, 2927–2935.
Klingler-Hoffmann M, Bukczynska P and Tiganis T . (2003). Int. J. Cancer, 105, 331–339.
Korshunov A, Golanov A, Sycheva R and Pronin I . (1999). J. Clin. Pathol., 52, 574–580.
Kuan CT, Wikstrand CJ and Bigner DD . (2001). Endocr. Relat. Cancer, 8, 83–96.
Levitzki A, Gazit A, Osherov N, Posner I and Gilon C . (1991). Methods Enzymol., 201, 347–361.
Lorimer IA and Lavictoire SJ . (2001). Biochim. Biophys. Acta, 1538, 1–9.
Luwor RB, Johns TG, Murone C, Huang HJ, Cavenee WK, Ritter G, Old LJ, Burgess AW and Scott AM . (2001). Cancer Res., 61, 5355–5361.
Masui H, Kawamoto T, Sato JD, Wolf B, Sato G and Mendelsohn J . (1984). Cancer Res., 44, 1002–1007.
Montgomery RB, Moscatello DK, Wong AJ, Cooper JA and Stahl WL . (1995). J. Biol. Chem., 270, 30562–30566.
Moscatello DK, Holgado-Madruga M, Emlet DR, Montgomery RB and Wong AJ . (1998). J. Biol. Chem., 273, 200–206.
Moscatello DK, Holgado-Madruga M, Godwin AK, Ramirez G, Gunn G, Zoltick PW, Biegel JA, Hayes RL and Wong AJ . (1995). Cancer Res., 55, 5536–5539.
Moscatello DK, Montgomery RB, Sundareshan P, McDanel H, Wong MY and Wong AJ . (1996). Oncogene, 13, 85–96.
Nagane M, Coufal F, Lin H, Bogler O, Cavenee WK and Huang HJ . (1996). Cancer Res., 56, 5079–5086.
Narita Y, Nagane M, Mishima K, Huang HJ, Furnari FB and Cavenee WK . (2002). Cancer Res., 62, 6764–6769.
Nishikawa R, Ji XD, Harmon RC, Lazar CS, Gill GN, Cavenee WK and Huang HJ . (1994). Proc. Natl. Acad. Sci. USA, 91, 7727–7731.
O'Rourke DM, Nute EJ, Davis JG, Wu C, Lee A, Murali R, Zhang HT, Qian X, Kao CC and Greene MI . (1998). Oncogene, 16, 1197–1207.
Olapade-Olaopa EO, Moscatello DK, MacKay EH, Horsburgh T, Sandhu DP, Terry TR, Wong AJ and Habib FK . (2000). Br. J. Cancer, 82, 186–194.
Palacios R and Steinmetz M . (1985). Cell, 41, 727–734.
Reddy EP, Korapati A, Chaturvedi P and Rane S . (2000). Oncogene, 19, 2532–2547.
Riese II DJ, Bermingham Y, van Raaij TM, Buckley S, Plowman GD and Stern DF . (1996a). Oncogene, 12, 345–353.
Riese DJ, Kim ED, Elenius K, Buckley S, Klagsbrun M, Plowman GD and Stern DF . (1996b). J. Biol. Chem., 271, 20047–20052.
Riese II DJ, van Raaij TM, Plowman GD, Andrews GC and Stern DF . (1995). Mol. Cell. Biol., 15, 5770–5776.
Sugawa N, Ekstrand AJ, James CD and Collins VP . (1990). Proc. Natl. Acad. Sci. USA, 87, 8602–8606.
Tang CK, Gong XQ, Moscatello DK, Wong AJ and Lippman ME . (2000). Cancer Res., 60, 3081–3087.
Walker F, Hibbs ML, Zhang HH, Gonez LJ and Burgess AW . (1998a). Growth Factors, 16, 53–67.
Walker F, Kato A, Gonez LJ, Hibbs ML, Pouliot N, Levitzki A and Burgess AW . (1998b). Mol. Cell. Biol., 18, 7192–7204.
Waterfield MD, Mayes EL, Stroobant P, Bennet PL, Young S, Goodfellow PN, Banting GS and Ozanne B . (1982). J. Cell Biochem., 20, 149–161.
Weinmann P, Gaehtgens P and Walzog B . (1999). Blood, 93, 3106–3115.
Wikstrand CJ, Hale LP, Batra SK, Hill ML, Humphrey PA, Kurpad SN, McLendon RE, Moscatello D, Pegram CN and Reist CJ . (1995). Cancer Res., 55, 3140–3148.
Wikstrand CJ, Reist CJ, Archer GE, Zalutsky MR and Bigner DD . (1998). J. Neurovirol., 4, 148–158.
Wright JH, Munar E, Jameson DR, Andreassen PR, Margolis RL, Seger R and Krebs EG . (1999). Proc. Natl. Acad. Sci. USA, 96, 11335–11340.
Wu CJ, Qian X and O'Rourke DM . (1999). DNA Cell Biol., 18, 731–741.
Zhu HJ, Iaria J, Orchard S, Walker F and Burgess AW . (2003). Growth Factors, 21, 15–30.
Acknowledgements
This work was supported by the NH&MRC of Australia.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Luwor, R., Zhu, HJ., Walker, F. et al. The tumor-specific de2–7 epidermal growth factor receptor (EGFR) promotes cells survival and heterodimerizes with the wild-type EGFR. Oncogene 23, 6095–6104 (2004). https://doi.org/10.1038/sj.onc.1207870
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1207870
Keywords
This article is cited by
-
EGFRvIII tumorigenicity requires PDGFRA co-signaling and reveals therapeutic vulnerabilities in glioblastoma
Oncogene (2021)
-
Reduced EGFR and increased miR-221 is associated with increased resistance to temozolomide and radiotherapy in glioblastoma
Scientific Reports (2020)
-
Synthetic cytokine receptors transmit biological signals using artificial ligands
Nature Communications (2018)
-
Epidermal growth factor receptor and EGFRvIII in glioblastoma: signaling pathways and targeted therapies
Oncogene (2018)
-
Interleukin-13 receptor alpha 2 cooperates with EGFRvIII signaling to promote glioblastoma multiforme
Nature Communications (2017)