Abstract
Objective:
To evaluate point-of-care (POC) measurement of total serum bilirubin (TSB) in the management of neonatal jaundice.
Study design:
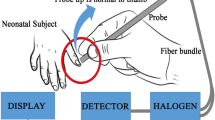
TSB was measured by a POC direct spectrophotometric bilirubin method (Unistat™ (U/TSB)) and a standard diazo clinical laboratory method (Olympus AU640E analyzer (diazo/TSB)). Agreement between U/TSB and diazo/TSB was assessed by correlation coefficient and Bland–Altman analysis. Transcutaneous bilirubin (TcB) was measured using JM-103™ (JM).
Results:
Correlation between U/TSB and diazo/TSB was 0.99 (n=120). Maximum difference (U/TSB minus diazo/TSB) was −2.9 mg/dl, and 79% were ±1 mg/dl; the average difference was −0.37±0.70 mg/dl and the average absolute difference was 0.60±0.52 mg/dl. Median time to determine U/TSB was 5 min. Correlation between U/TSB and JM was 0.92 (n=113). Maximum difference (U/TSB minus JM) was 6.3 mg/dl, and 45% were ±1 mg/dl; the average difference was 0.7±1.8 mg/dl and the average absolute difference was 1.4±1.2 mg/dl.
Conclusion:
Measurement of TSB using Unistat™ provides excellent agreement with diazo/TSB and rapid turnaround time. This technique may provide reliable POC confirmation of TcB results that are above a screening cutoff value.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
American Academy of Pediatrics Subcommittee on Hyperbilirubinemia. Management of hyperbilirubinemia in the newborn infant 35 or more weeks gestation. Pediatrics 2004; 114: 297–316.
Bhutani VK, Gourley GR, Adler S, Kreamer B, Dalin C, Johnson LH . Noninvasive measurement of total serum bilirubin in a multiracial predischarge newborn population to assess the risk of severe hyperbilirubinemia. Pediatrics 2000; 106, 2/e17.
Bhutani VK, Johnson L, Sivieri EM . Predictive ability of a pre-discharge hour-specific serum bilirubin for subsequent significant hyperbilirubinemia in healthy term and near-term newborns. Pediatrics 1999; 103: 6–14.
Ebbesen F . Determination of serum bilirubin concentration during phototherapy of newborns and in vitro: results compared by the direct spectrophotometric method and the diazo method. Clin Chem 1977; 23: 695–699.
Rolinski B, Küster H, Ugele B, Gruber R, Horn K . Total bilirubin measurement by photometry on a blood gas analyzer: potential for use in neonatal testing at the point of care. Clin Chem 2001; 47: 1845–1847.
Kazmierczak SC, Robertson AF, Catrou PG, Briley KP, Kreamer BL, Gourley GR . Direct spectrophotometric method for measurement of bilirubin in newborns: comparison with HPLC and an automated diazo method. Clin Chem 2002; 48: 1096–1097.
Blumenfeld T, Steinbrink C, Cheskin H, George J, Vogl T . Manual micromethods for bilirubin determination in sera of adults and children and investigation of reasons for observed differences. Am J Clin Path 1978; 69: 388–397.
Reichert Unistat™ Operations Manual. Depew, NY; 2003: pp 1–24.
Lo SF, Doumas BT, Ashwood ER . Bilirubin proficiency testing using specimens containing unconjugated bilirubin and human serum. Arch Pathol Lab Med 2004; 128: 1219–1223.
Bhutani VK, Johnson LH . Urgent clinical need for accurate and precise bilirubin measurements in the United States to prevent kernicterus. Clin Chem 2004; 50: 477–480.
Vreman HJ, Verter J, Oh W, Fanaroff AA, Wright LL, Lemons JA et al. Interlaboratory variability of bilirubin measurements. Clin Chem 1996; 42: 869–873.
Stevenson DK, Fanaroff AA, Maisels MJ, Young BW, Wong RJ, Vreman HJ et al. Prediction of hyperbilirubinemia in near-term and term infants. Pediatrics 2001; 108: 31–39.
Harkness RA, Lawrence CR, Renshaw A, Barr IC, Brown SS, Rinsler MG . Assessment of the performance and clinical utility of a ward side-room bilirubinometer. Ann Clin Biochem 1983; 20: 149–152.
Maisels MJ, Ostrea EM, Touch S, Clune SE, Cepeda E, Kring E et al. Evaluation of a new transcutaneous bilirubinometer. Pediatrics 2004; 113: 1628–1635.
Engle WD, Jackson GL, Sendelbach D, Manning D, Frawley WH . Assessment of a transcutaneous device in the evaluation of neonatal hyperbilirubinemia in a primarily Hispanic population. Pediatrics 2002; 110: 61–67.
Engle WD, Jackson GL, Stehel EK, Sendelbach DM, Manning MD . Evaluation of a transcutaneous jaundice meter following hospital discharge in term and near-term neonates. J Perinatol 2005; 25: 486–490.
Bland JM, Altman DG . Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 1986; 1: 307–310.
Wong CM, van Dijk PJE, Laing IA . A comparison of transcutaneous bilirubinometers: SpectRx BiliCheck versus Minolta AirShields. Arch Dis Child Fetal Neonatal Ed 2002; 87: F137–F140.
Nanjundaswamy S, Petrova A, Mehta R, Bernstein W, Hegyi T . The accuracy of transcutaneous bilirubin measurements in neonates: a correlation study. Biol Neonate 2004; 85: 21–25.
Ebbesen F, Rasmussen LM, Wimberley PD . A new transcutaneous bilirubinometer, BiliCheck, used in the neonatal intensive care unit and the maternity ward. Acta Paediatr 2002; 91: 203–211.
Dai J, Parry DM, Krahn J . Transcutaneous bilirubinometry: its role in the assessment of neonatal jaundice. Clin Biochem 1997; 30: 1–9.
Sykes E, Epstein E . Laboratory measurement of bilirubin. Clin Perinatol 1990; 17: 397–416.
Doumas B, Wu T . The measurement of bilirubin fractions in serum. Crit Rev Clin Lab Sci 1991; 28: 415–445.
Meites S, Hogg C . Direct spectrophotometry of total serum bilirubin in the newborn. Clin Chem 1960; 6: 421–428.
http://www.hematek.com/index.html, and personal communication with Ian Hunter, General Manager of Hematechnologies Ltd, February 2005.
Ou C, Frawley V . Investigation in the use of the Du Pont neonatal bilirubin (NBIL) method for the determination of bilirubin in neonates. Clin Chem 1983; 29: 1221.
Acknowledgements
We appreciate the participation of the Parkland Health and Hospital System's Clinical Pathology Laboratory for providing financial assistance for total serum bilirubin levels, Reichert, Inc., for providing the Unistat™, centrifuge, and pipette at no charge, and Minolta/Hill-Rom Air-Shields who provided the JM-103™ at no charge.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Barko, H., Jackson, G. & Engle, W. Evaluation of a point-of-care direct spectrophotometric method for measurement of total serum bilirubin in term and near-term neonates. J Perinatol 26, 100–105 (2006). https://doi.org/10.1038/sj.jp.7211436
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.jp.7211436
Keywords
This article is cited by
-
Modified Kramer’s versus Kramer’s Method for Clinical Assessment of Jaundice in Term and Near-Term Neonates
Indian Journal of Pediatrics (2024)
-
Diagnostic methods for neonatal hyperbilirubinemia: benefits, limitations, requirements, and novel developments
Pediatric Research (2021)