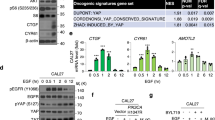
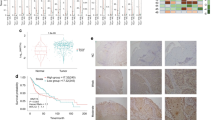
Abstract
Mtss1 is located within chromosomal region 8q23–24, which is one of the three most commonly amplified regions in head and neck squamous cell carcinoma (HNSCC). Mtss1 is lost in metastatic cells, but confusingly is commonly overexpressed in primary tumors. Here we address possible reasons why Mtss1 is positively selected for in primary tumors. We find that Mtss1 enhances the localization of the epidermal growth factor (EGF) receptor to the plasma membrane, prolonging EGF signaling and resulting in enhanced proliferation in HNSCC. Depletion of Mtss1 results in decreased EGF receptor levels and decreased phosphorylation of Erk1/2 and Akt. However, when cells are at high density and adherent to each other, analogous to conditions in a solid tumor, Mtss1 does not confer any growth advantage, either in basal conditions or following EGF stimulation. This could indicate why Mtss1 might be lost in metastases, but preserved in early primary tumors. This is supported by an organotypic assay showing that Mtss1-expressing cells display a less proliferative more epithelial-like morphology on top of a collagen matrix. Furthermore, xenograft tumors expressing Mtss1 initially grow more rapidly, but later show less proliferation and more differentiation. Mtss1 positively modulates EGF signaling at low cell densities to promote proliferation and, therefore, may be beneficial for the early stages of primary HNSCC tumor growth. However, at high cell densities, Mtss1 impacts negatively on EGF signaling and this suggests why it inhibits metastasis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Basu D, Nguyen TT, Montone KT, Zhang G, Wang LP, Diehl JA et al. (2010). Evidence for mesenchymal-like sub-populations within squamous cell carcinomas possessing chemoresistance and phenotypic plasticity. Oncogene 29: 4170–4182.
Bershteyn M, Atwood SX, Woo WM, Li M, Oro AE . (2010). MIM and cortactin antagonism regulates ciliogenesis and hedgehog signaling. Dev Cell 19: 270–283.
Betson M, Lozano E, Zhang J, Braga VM . (2002). Rac activation upon cell–cell contact formation is dependent on signaling from the epidermal growth factor receptor. J Biol Chem 277: 36962–36969.
Boissan M, De Wever O, Lizarraga F, Wendum D, Poincloux R, Chignard N et al. (2010). Implication of metastasis suppressor NM23-H1 in maintaining adherens junctions and limiting the invasive potential of human cancer cells. Cancer Res 70: 7710–7722.
Bompard G, Sharp SJ, Freiss G, Machesky LM . (2005). Involvement of Rac in actin cytoskeleton rearrangements induced by MIM-B. J Cell Sci 118: 5393–5403.
Bremm A, Walch A, Fuchs M, Mages J, Duyster J, Keller G et al. (2008). Enhanced activation of epidermal growth factor receptor caused by tumor-derived E-cadherin mutations. Cancer Res 68: 707–714.
Callahan CA, Ofstad T, Horng L, Wang JK, Zhen HH, Coulombe PA et al. (2004). MIM/BEG4, a Sonic hedgehog-responsive gene that potentiates Gli-dependent transcription. Genes Dev 18: 2724–2729.
Canel M, Secades P, Rodrigo JP, Cabanillas R, Herrero A, Suarez C et al. (2006). Overexpression of focal adhesion kinase in head and neck squamous cell carcinoma is independent of fak gene copy number. Clin Cancer Res 12: 3272–3279.
Cheng GZ, Chan J, Wang Q, Zhang W, Sun CD, Wang LH . (2007). Twist transcriptionally up-regulates AKT2 in breast cancer cells leading to increased migration, invasion, and resistance to paclitaxel. Cancer Res 67: 1979–1987.
Chung CH, Ely K, McGavran L, Varella-Garcia M, Parker J, Parker N et al. (2006). Increased epidermal growth factor receptor gene copy number is associated with poor prognosis in head and neck squamous cell carcinomas. J Clin Oncol 24: 4170–4176.
Cooper JB, Cohen EE . (2009). Mechanisms of resistance to EGFR inhibitors in head and neck cancer. Head Neck 31: 1086–1094.
Danglot L, Chaineau M, Dahan M, Gendron MC, Boggetto N, Perez F et al. (2010). Role of TI-VAMP and CD82 in EGFR cell-surface dynamics and signaling. J Cell Sci 123: 723–735.
Donev IS, Wang W, Yamada T, Li Q, Takeuchi S, Matsumoto K et al. (2011). Transient PI3K inhibition induces apoptosis and overcomes HGF-mediated resistance to EGFR-TKIs in EGFR mutant lung cancer. Clin Cancer Res 17: 2260–2269.
Edward M, Gillan C, Micha D, Tammi RH . (2005). Tumour regulation of fibroblast hyaluronan expression: a mechanism to facilitate tumour growth and invasion. Carcinogenesis 26: 1215–1223.
Gan Y, Shi C, Inge L, Hibner M, Balducci J, Huang Y . (2010). Differential roles of ERK and Akt pathways in regulation of EGFR-mediated signaling and motility in prostate cancer cells. Oncogene 29: 4947–4958.
Goh LK, Huang F, Kim W, Gygi S, Sorkin A . (2010). Multiple mechanisms collectively regulate clathrin-mediated endocytosis of the epidermal growth factor receptor. J Cell Biol 189: 871–883.
Gonzalez-Quevedo R, Shoffer M, Horng L, Oro AE . (2005). Receptor tyrosine phosphatase-dependent cytoskeletal remodeling by the hedgehog-responsive gene MIM/BEG4. J Cell Biol 168: 453–463.
Hsia DA, Mitra SK, Hauck CR, Streblow DN, Nelson JA, Ilic D et al. (2003). Differential regulation of cell motility and invasion by FAK. J Cell Biol 160: 753–767.
Irie HY, Pearline RV, Grueneberg D, Hsia M, Ravichandran P, Kothari N et al. (2005). Distinct roles of Akt1 and Akt2 in regulating cell migration and epithelial–mesenchymal transition. J Cell Biol 171: 1023–1034.
Kelley LC, Shahab S, Weed SA . (2008). Actin cytoskeletal mediators of motility and invasion amplified and overexpressed in head and neck cancer. Clin Exp Metast 25: 289–304.
Lee SH, Kerff F, Chereau D, Ferron F, Klug A, Dominguez R . (2007). Structural basis for the actin-binding function of missing-in-metastasis. Structure 15: 145–155.
Lee YG, Macoska JA, Korenchuk S, Pienta KJ . (2002). MIM, a potential metastasis suppressor gene in bladder cancer. Neoplasia 4: 291–294.
Lin J, Liu J, Wang Y, Zhu J, Zhou K, Smith N et al. (2005). Differential regulation of cortactin and N-WASP-mediated actin polymerization by missing in metastasis (MIM) protein. Oncogene 24: 2059–2066.
Lin M, Smith LT, Smiraglia DJ, Kazhiyur-Mannar R, Lang JC, Schuller DE et al. (2006). DNA copy number gains in head and neck squamous cell carcinoma. Oncogene 25: 1424–1433.
Liu K, Wang G, Ding H, Chen Y, Yu G, Wang J . (2010). Downregulation of metastasis suppressor 1(MTSS1) is associated with nodal metastasis and poor outcome in Chinese patients with gastric cancer. BMC Cancer 10: 428.
Loberg RD, Neeley CK, Adam-Day LL, Fridman Y, St John LN, Nixdorf S et al. (2005). Differential expression analysis of MIM (MTSS1) splice variants and a functional role of MIM in prostate cancer cell biology. Int J Oncol 26: 1699–1705.
Ma S, Guan XY, Lee TK, Chan KW . (2007). Clinicopathological significance of missing in metastasis B expression in hepatocellular carcinoma. Hum Pathol 38: 1201–1206.
Machesky LM, Johnston SA . (2007). MIM: a multifunctional scaffold protein. J Mol Med 85: 569–576.
Mattila PK, Pykalainen A, Saarikangas J, Paavilainen VO, Vihinen H, Jokitalo E et al. (2007). Missing-in-metastasis and IRSp53 deform PI(4,5)P2-rich membranes by an inverse BAR domain-like mechanism. J Cell Biol 176: 953–964.
Mattila PK, Salminen M, Yamashiro T, Lappalainen P . (2003). Mouse MIM, a tissue-specific regulator of cytoskeletal dynamics, interacts with ATP-actin monomers through its C-terminal WH2 domain. J Biol Chem 278: 8452–8459.
Miranti CK . (2009). Controlling cell surface dynamics and signaling: how CD82/KAI1 suppresses metastasis. Cell Signal 21: 196–211.
Mosesson Y, Mills GB, Yarden Y . (2008). Derailed endocytosis: an emerging feature of cancer. Nat Rev Cancer 8: 835–850.
Parr C, Jiang WG . (2009). Metastasis suppressor 1 (MTSS1) demonstrates prognostic value and anti-metastatic properties in breast cancer. Eur J Cancer 45: 1673–1683.
Qian X, Karpova T, Sheppard AM, McNally J, Lowy DR . (2004). E-cadherin-mediated adhesion inhibits ligand-dependent activation of diverse receptor tyrosine kinases. EMBO J 23: 1739–1748.
Quinones GA, Jin J, Oro AE . (2010). I-BAR protein antagonism of endocytosis mediates directional sensing during guided cell migration. J Cell Biol 189: 353–367.
Sigismund S, Argenzio E, Tosoni D, Cavallaro E, Polo S, Di Fiore PP . (2008). Clathrin-mediated internalization is essential for sustained EGFR signaling but dispensable for degradation. Dev Cell 15: 209–219.
Sorkin A, Goh LK . (2009). Endocytosis and intracellular trafficking of ErbBs. Exp Cell Res 315: 683–696.
Thurlow JK, Pena Murillo CL, Hunter KD, Buffa FM, Patiar S, Betts G et al. (2010). Spectral clustering of microarray data elucidates the roles of microenvironment remodeling and immune responses in survival of head and neck squamous cell carcinoma. J Clin Oncol 28: 2881–2888.
Timpson P, Lynch DK, Schramek D, Walker F, Daly RJ . (2005). Cortactin overexpression inhibits ligand-induced down-regulation of the epidermal growth factor receptor. Cancer Res 65: 3273–3280.
Timpson P, Wilson AS, Lehrbach GM, Sutherland RL, Musgrove EA, Daly RJ . (2007). Aberrant expression of cortactin in head and neck squamous cell carcinoma cells is associated with enhanced cell proliferation and resistance to the epidermal growth factor receptor inhibitor gefitinib. Cancer Res 67: 9304–9314.
Uhlenbrock K, Eberth A, Herbrand U, Daryab N, Stege P, Meier F et al. (2004). The RacGEF Tiam1 inhibits migration and invasion of metastatic melanoma via a novel adhesive mechanism. J Cell Sci 117: 4863–4871.
Woodcock SA, Rooney C, Liontos M, Connolly Y, Zoumpourlis V, Whetton AD et al. (2009). SRC-induced disassembly of adherens junctions requires localized phosphorylation and degradation of the rac activator tiam1. Mol Cell 33: 639–653.
Woodings JA, Sharp SJ, Machesky LM . (2003). MIM-B, a putative metastasis suppressor protein, binds to actin and to protein tyrosine phosphatase delta. Biochem J 371: 463–471.
Yang MH, Wu MZ, Chiou SH, Chen PM, Chang SY, Liu CJ et al. (2008). Direct regulation of TWIST by HIF-1alpha promotes metastasis. Nat Cell Biol 10: 295–305.
Acknowledgements
JD was funded by an AICR Grant to LMM (07-0438). LMM received funding from an MRC Senior Research fellowship (G117/569) and a CRUK core grant. We thank Roger Daly for HNSCC cell lines and Heather Spence for technical help.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Dawson, J., Timpson, P., Kalna, G. et al. Mtss1 regulates epidermal growth factor signaling in head and neck squamous carcinoma cells. Oncogene 31, 1781–1793 (2012). https://doi.org/10.1038/onc.2011.376
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2011.376
Keywords
This article is cited by
-
Endocytosis: a pivotal pathway for regulating metastasis
British Journal of Cancer (2021)
-
Computational analysis of the evolutionarily conserved Missing In Metastasis/Metastasis Suppressor 1 gene predicts novel interactions, regulatory regions and transcriptional control
Scientific Reports (2019)
-
Epidermal growth factor-induced ANGPTL4 enhances anoikis resistance and tumour metastasis in head and neck squamous cell carcinoma
Oncogene (2017)
-
Mtss1 promotes maturation and maintenance of cerebellar neurons via splice variant-specific effects
Brain Structure and Function (2017)
-
Elevated MTSS1 expression associated with metastasis and poor prognosis of residual hepatitis B-related hepatocellular carcinoma
Journal of Experimental & Clinical Cancer Research (2016)