Abstract
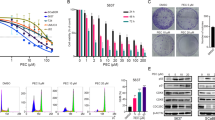
To improve conventional chemotherapeutic efficacy, a combination use of traditional medicines is effective but detailed mechanisms have been rarely elucidated. In the this study, we attempted to clarify how triptolide (PG490), an oxygenated diterpene derived from a Chinese herb, enhances the cisplatin (CDDP)-induced cytotoxicity in urothelial cancer cells. Our results showed that a combined CDDP/triptolide therapy induced apoptosis in urothelial cancer cell lines with wild-type p53, but not in those with mutant-type p53 or normal human urothelium. As the mechanism, triptolide suppressed CDDP-induced p53 transcriptional activity, leading to p21 attenuation, which promoted apoptosis via the activation of c-Jun N-terminal kinase (JNK) and Bax. We further demonstrated that the functional regulation of p53 by triptolide was mediated by an intranuclear association of p53 with glycogen synthase kinase-3β (GSK3β), which was inactivated by protein kinase C (PKC). This modulation of the PKC-GSK3β axis by triptolide was observed in a cancer-specific manner. A mouse xenograft model also showed that a combined CDDP/triptolide therapy completely suppressed tumor growth without any side effects. We expect that cancer-specific enhancement of CDDP-induced cytotoxicity with triptolide may effectively overcome the resistance to a CDDP-based conventional chemotherapy as a treatment for urothelial cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Aaltonen V, Koivunen J, Laato M, Peltonen J . (2006). Heterogeneity of cellular proliferation within transitional cell carcinoma: correlation of protein kinase C alpha/betaI expression and activity. J Histochem Cytochem 54: 795–806.
Asada M, Yamada T, Ichijo H, Delia D, Miyazono K, Fukumuro K et al. (1999). Apoptosis inhibitory activity of cytoplasmic p21(Cipl/WAFl) in monocytic differentiation. EMBO J 18: 1223–1234.
Bode AM, Dong Z . (2004). Post-translational modification of p53 in tumorigenesis. Nat Rev Cancer 4: 793–805.
Camacho-Carvajal MM, Wollscheid B, Aebersold R, Steimle V, Schamel WW . (2004). Two-dimensional Blue native/SDS gel electrophoresis of multi-protein complexes from whole cellular lysates: a proteomics approach. Mol Cell Proteomics 3: 176–182.
Carter BZ, Mak DH, Schober WD, McQueen T, Harris D, Estrov Z et al. (2006). Triptolide induces caspase-dependent cell death mediated via the mitochondrial pathway in leukemic cells. Blood 108: 630–637.
Chang WT, Kang JJ, Lee KY, Wei K, Anderson E, Gotmare S et al. (2001). Triptolide and chemotherapy cooperate in tumor cell apoptosis. A role for the p53 pathway. J Biol Chem 276: 2221–2227.
Chen BJ, Liu C, Cui X, Fidler JM, Chao NJ . (2000). Prevention of graft-versus-host disease by a novel immunosuppressant, PG490-88, through inhibition of alloreactive T cell expansion. Transplantation 70: 1442–1447.
Cibere J, Deng Z, Lin Y, Ou R, He Y, Wang Z et al. (2003). A randomized double blind, placebo controlled trial of topical Tripterygium wilfordii in rheumatoid arthritis: reanalysis using logistic regression analysis. J Rheumatol 30: 465–467.
Cote RJ, Esrig D, Groshen S, Jones PA, Skinner DG . (1997). p53 and treatment of bladder cancer. Nature 385: 123–125.
Coutts AS, La Thangue NB . (2005). The p53 response: emerging levels of co-factor complexity. Biochem Biophys Res Commun 331: 778–785.
Demidenko ZN, Blagosklonny MV . (2004). Flavopiridol induces p53 via initial inhibition of Mdm2 and p21 and, independently of p53, sensitizes apoptosis-reluctant cells to tumor necrosis factor. Cancer Res 64: 3653–3660.
Erster S, Mihara M, Kim RH, Petrenko O, Moll UM . (2004). In vivo mitochondrial p53 translocation triggers a rapid first wave of cell death in response to DNA damage that can precede p53 target gene activation. Mol Cell Biol 24: 6728–6741.
Fidler JM, Li K, Chung C, Wei K, Ross JA, Gao M et al. (2003). PG490-88, a derivative of triptolide, causes tumor regression and sensitizes tumors to chemotherapy. Mol Cancer Ther 2: 855–862.
Gartel AL, Tyner AL . (2002). The role of the cyclin-dependent kinase inhibitor p21 in apoptosis. Mol Cancer Ther 1: 639–649.
Huang S, Shu L, Dilling MB, Easton J, Harwood FC, Ichijo H et al. (2003). Sustained activation of the JNK cascade and rapamycin-induced apoptosis are suppressed by p53/p21(Cipl). Mol Cell 11: 1491–1501.
Jiang XH, Tu SP, Cui JT, Lin MC, Xia HH, Wong WM et al. (2004). Antisense targeting protein kinase C alpha and betal inhibits gastric carcinogenesis. Cancer Res 64: 5787–5794.
Jiang XH, Wong BC, Lin MC, Zhu GH, Kung HF, Jiang SH et al. (2001). Functional p53 is required for triptolide-induced apoptosis and AP-1 and nuclear factor-kappaB activation in gastric cancer cells. Oncogene 20: 8009–8018.
Koivunen J, Aaltonen V, Koskela S, Lehenkari P, Laato M, Peltonen J . (2004). Protein kinase C alpha/beta inhibitor Go6976 promotes formation of cell junctions and inhibits invasion of urinary bladder carcinoma cells. Cancer Res 64: 5693–5701.
Kulikov R, Boehme KA, Blattner C . (2005). Glycogen synthase kinase 3-dependent phosphorylation of Mdm2 regulates p53 abundance. Mol Cell Biol 25: 7170–7180.
Kupchan SM, Court WA, Dailey Jr RG, Gilmore CJ, Bryan RF . (1972). Triptolide and tripdiolide, novel antileukemic diterpenoid triepoxides from Tripterygium wilfordii. J Am Chem Soc 94: 7194–7195.
Lam KS, Liu R, Miyamoto S, Lehman AL, Tuscano JM . (2003). Applications of one-bead one-compound combinatorial libraries and chemical microarrays in signal transduction research. Acc Chem Res 36: 370–377.
Lee KY, Chang W, Qiu D, Kao PN, Rosen GD . (1999). PG490 (triptolide) cooperates with tumor necrosis factor-alpha to induce apoptosis in tumor cells. J Biol Chem 274: 13451–13455.
Mandil R, Ashkenazi E, Blass M, Kronfeld I, Kazimirsky G, Rosenthal G et al. (2001). Protein kinase Calpha and protein kinase Cdelta play opposite roles in the proliferation and apoptosis of glioma cells. Cancer Res 61: 4612–4619.
Mansouri A, Ridgway LD, Korapati AL, Zhang Q, Tian L, Wang Y et al. (2003). Sustained activation of JNK/p38 MAPK pathways in response to cisplatin leads to Fas ligand induction and cell death in ovarian carcinoma cells. J Biol Chem 278: 19245–19256.
Matsui Y, Ueda S, Watanabe J, Kuwabara I, Ogawa O, Nishiyama H . (2007). Sensitizing effect of galectin-7 in urothelial cancer to cisplatin through the accumulation of intracellular reactive oxygen species. Cancer Res 67: 1212–1220.
Miyata Y, Sato T, Ito A . (2005). Triptolide, a diterpenoid triepoxide, induces antitumor proliferation via activation of c-Jun NH2-terminal kinase 1 by decreasing phosphatidylinositol 3-kinase activity in human tumor cells. Biochem Biophys Res Commun 336: 1081–1086.
Nishiyama H, Habuchi T, Watanabe J, Teramukai S, Tada H, Ono Y et al. (2004). Clinical outcome of a large-scale multi-institutional retrospective study for locally advanced bladder cancer: a survey including 1131 patients treated during 1990–2000 in Japan. Eur Urol 45: 176–181.
Niwa H, Yamamura K, Miyazaki J . (1991). Efficient selection for high-expression transfectants with a novel eukaryotic vector. Gene 108: 193–199.
Nogawa M, Yuasa T, Kimura S, Kuroda J, Sato K, Segawa H et al. (2005). Monitoring luciferase-labeled cancer cell growth and metastasis in different in vivo models. Cancer Lett 217: 243–253.
Qiu D, Zhao G, Aoki Y, Shi L, Uyei A, Nazarian S et al. (1999). Immunosuppressant PG490 (triptolide) inhibits T-cell interleukin-2 expression at the level of purine-box/nuclear factor of activated T-cells and NF-kappaB transcriptional activation. J Biol Chem 274: 13443–13450.
Sarkis AS, Bajorin DF, Reuter VE, Herr HW, Netto G, Zhang ZF et al. (1995). Prognostic value of p53 nuclear overexpression in patients with invasive bladder cancer treated with neoadjuvant MVAC. J Clin Oncol 13: 1384–1390.
Shamon LA, Pezzuto JM, Graves JM, Mehta RR, Wangcharoentrakul S, Sangsuwan R et al. (1997). Evaluation of the mutagenic, cytotoxic, and antitumor potential of triptolide, a highly oxygenated diterpene isolated from Tripterygium wilfordii. Cancer Lett 112: 113–117.
Southgate J, Hutton KA, Thomas DF, Trejdosiewicz LK . (1994). Normal human urothelial cells in vitro: proliferation and induction of stratification. Lab Invest 71: 583–594.
Tan J, Zhuang L, Leong HS, Iyer NG, Liu ET, Yu Q . (2005). Pharmacologic modulation of glycogen synthase kinase-3beta promotes p53-dependent apoptosis through a direct Bax-mediated mitochondrial pathway in colorectal cancer cells. Cancer Res 65: 9012–9020.
Wan CK, Wang C, Cheung HY, Yang M, Fong WF . (2006). Triptolide induces Bcl-2 cleavage and mitochondria dependent apoptosis in p53-deficient HL-60 cells. Cancer Lett 241: 31–41.
Watanabe J, Nishiyama H, Matsui Y, Ito M, Kawanishi H, Kamoto T et al. (2006). Dicoumarol potentiates cisplatin-induced apoptosis mediated by c-Jun N-terminal kinase in p53 wild-type urogenital cancer cell lines. Oncogene 25: 2500–2508.
Watcharasit P, Bijur GN, Song L, Zhu J, Chen X, Jope RS . (2003). Glycogen synthase kinase-3beta (GSK3beta) binds to and promotes the actions of p53. J Biol Chem 278: 48872–48879.
Watcharasit P, Bijur GN, Zmijewski JW, Song L, Zmijewska A, Chen X et al. (2002). Direct, activating interaction between glycogen synthase kinase-3beta and p53 after DNA damage. Proc Natl Acad Sci USA 99: 7951–7955.
Weiss RH, Marshall D, Howard L, Corbacho AM, Cheung AT, Sawai ET . (2003). Suppression of breast cancer growth and angiogenesis by an antisense oligodeoxynucleotide to p21(Wafl/Cipl). Cancer Lett 189: 39–48.
Weiss RH . (2003). p21Wafl/Cipl as a therapeutic target in breast and other cancers. Cancer Cell 4: 425–429.
Yang S, Chen J, Guo Z, Xu XM, Wang L, Pei XF et al. (2003). Triptolide inhibits the growth and metastasis of solid tumors. Mol Cancer Ther 2: 65–72.
Yinjun L, Jie J, Yungui W . (2005). Triptolide inhibits transcription factor NF-kappaB and induces apoptosis of multiple myeloma cells. Leuk Res 29: 99–105.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc).
Rights and permissions
About this article
Cite this article
Matsui, Y., Watanabe, J., Ikegawa, M. et al. Cancer-specific enhancement of cisplatin-induced cytotoxicity with triptolide through an interaction of inactivated glycogen synthase kinase-3β with p53. Oncogene 27, 4603–4614 (2008). https://doi.org/10.1038/onc.2008.89
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2008.89
Keywords
This article is cited by
-
Triptolide potentiates lung cancer cells to cisplatin-induced apoptosis by selectively inhibiting the NER activity
Biomarker Research (2015)
-
XPB, a subunit of TFIIH, is a target of the natural product triptolide
Nature Chemical Biology (2011)
-
p53 and chemosensitivity in bladder cancer
International Journal of Clinical Oncology (2008)