Abstract
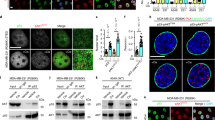
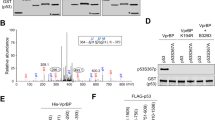
The tumor-suppressor function of p53 relies on its transcriptional activity, which is modulated by post-translational modifications and interactions with regulatory proteins. The prolyl isomerase Pin1 has a central role in transducing phosphorylation of p53 into conformational changes that affect p53 stability and function. We found that Pin1 is required for efficient loading of p53 on target promoters upon stress. In addition, Pin1 is recruited to chromatin by p53 and stimulates binding of the p300 acetyltransferase and consequent p53 acetylation. Accordingly, tumor-associated mutations at Pin1-binding residues within the p53 proline-rich domain hamper acetylation of p53 by p300. After phosphorylation of p53 at Ser46 triggered by cytotoxic stimuli, Pin1 also mediates p53's dissociation from the apoptosis inhibitor iASPP, promoting cell death. In tumors bearing wild-type p53, expression of Pin1 and iASPP are inversely correlated, supporting the clinical relevance of these interactions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Appella, E. & Anderson, C.W. Post-translational modifications and activation of p53 by genotoxic stresses. Eur. J. Biochem. 268, 2764–2772 (2001).
Lavin, M.F. & Gueven, N. The complexity of p53 stabilization and activation. Cell Death Differ. 13, 941–950 (2006).
Gu, W. & Roeder, R.G. Activation of p53 sequence-specific DNA binding by acetylation of the p53 C-terminal domain. Cell 90, 595–606 (1997).
Luo, J. et al. Acetylation of p53 augments its site-specific DNA binding both in vitro and in vivo. Proc. Natl. Acad. Sci. USA 101, 2259–2264 (2004).
Knights, C.D. et al. Distinct p53 acetylation cassettes differentially influence gene-expression patterns and cell fate. J. Cell Biol. 173, 533–544 (2006).
Tang, Y., Luo, J., Zhang, W. & Gu, W. Tip60-dependent acetylation of p53 modulates the decision between cell-cycle arrest and apoptosis. Mol. Cell 24, 827–839 (2006).
Sykes, S.M. et al. Acetylation of the p53 DNA-binding domain regulates apoptosis induction. Mol. Cell 24, 841–851 (2006).
Ceskova, P., Chichger, H., Wallace, M., Vojtesek, B. & Hupp, T.R. On the mechanism of sequence-specific DNA-dependent acetylation of p53: the acetylation motif is exposed upon DNA binding. J. Mol. Biol. 357, 442–456 (2006).
Yaffe, M.B. et al. Sequence-specific and phosphorylation-dependent proline isomerization: a potential mitotic regulatory mechanism. Science 278, 1957–1960 (1997).
Yeh, E.S. & Means, A.R. PIN1, the cell cycle and cancer. Nat. Rev. Cancer 7, 381–388 (2007).
Bulavin, D.V. et al. Phosphorylation of human p53 by p38 kinase coordinates N-terminal phosphorylation and apoptosis in response to UV radiation. EMBO J. 18, 6845–6854 (1999).
Buschmann, T. et al. Jun NH2-terminal kinase phosphorylation of p53 on Thr-81 is important for p53 stabilization and transcriptional activities in response to stress. Mol. Cell. Biol. 21, 2743–2754 (2001).
Hofmann, T.G. et al. Regulation of p53 activity by its interaction with homeodomain-interacting protein kinase-2. Nat. Cell Biol. 4, 1–10 (2002).
D'Orazi, G. et al. Homeodomain-interacting protein kinase-2 phosphorylates p53 at Ser 46 and mediates apoptosis. Nat. Cell Biol. 4, 11–19 (2002).
Radhakrishnan, S.K. & Gartel, A.L. CDK9 phosphorylates p53 on serine residues 33, 315 and 392. Cell Cycle 5, 519–521 (2006).
Wang, Y. & Prives, C. Increased and altered DNA binding of human p53 by S and G2/M but not G1 cyclin-dependent kinases. Nature 376, 88–91 (1995).
Zacchi, P. et al. The prolyl isomerase Pin1 reveals a mechanism to control p53 functions after genotoxic insults. Nature 419, 853–857 (2002).
Zheng, H. et al. The prolyl isomerase Pin1 is a regulator of p53 in genotoxic response. Nature 419, 849–853 (2002).
Wulf, G.M., Liou, Y.C., Ryo, A., Lee, S.W. & Lu, K.P. Role of Pin1 in the regulation of p53 stability and p21 transactivation, and cell cycle checkpoints in response to DNA damage. J. Biol. Chem. 277, 47976–47979 (2002).
Berger, M., Stahl, N., Del Sal, G. & Haupt, Y. Mutations in proline 82 of p53 impair its activation by Pin1 and Chk2 in response to DNA damage. Mol. Cell. Biol. 25, 5380–5388 (2005).
Walker, K.K. & Levine, A.J. Identification of a novel p53 functional domain that is necessary for efficient growth suppression. Proc. Natl. Acad. Sci. USA 93, 15335–15340 (1996).
Berger, M., Vogt Sionov, R., Levine, A.J. & Haupt, Y. A role for the polyproline domain of p53 in its regulation by Mdm2. J. Biol. Chem. 276, 3785–3790 (2001).
Dornan, D., Shimizu, H., Burch, L., Smith, A.J. & Hupp, T.R. The proline repeat domain of p53 binds directly to the transcriptional coactivator p300 and allosterically controls DNA-dependent acetylation of p53. Mol. Cell. Biol. 23, 8846–8861 (2003).
Venot, C. et al. The requirement for the p53 proline-rich functional domain for mediation of apoptosis is correlated with specific PIG3 gene transactivation and with transcriptional repression. EMBO J. 17, 4668–4679 (1998).
Zhu, J., Jiang, J., Zhou, W., Zhu, K. & Chen, X. Differential regulation of cellular target genes by p53 devoid of the PXXP motifs with impaired apoptotic activity. Oncogene 18, 2149–2155 (1999).
Toledo, F. et al. A mouse p53 mutant lacking the proline-rich domain rescues Mdm4 deficiency and provides insight into the Mdm2-Mdm4-p53 regulatory network. Cancer Cell 9, 273–285 (2006).
Chipuk, J.E. et al. Direct activation of Bax by p53 mediates mitochondrial membrane permeabilization and apoptosis. Science 303, 1010–1014 (2004).
Bergamaschi, D. et al. iASPP oncoprotein is a key inhibitor of p53 conserved from worm to human. Nat. Genet. 33, 162–167 (2003).
Bergamaschi, D. et al. iASPP preferentially binds p53 proline-rich region and modulates apoptotic function of codon 72-polymorphic p53. Nat. Genet. 38, 1133–1141 (2006).
Matlashewski, G.J. et al. Primary structure polymorphism at amino acid residue 72 of human p53. Mol. Cell. Biol. 7, 961–963 (1987).
Thomas, M. et al. Two polymorphic variants of wild-type p53 differ biochemically and biologically. Mol. Cell. Biol. 19, 1092–1100 (1999).
Sullivan, A. et al. Polymorphism in wild-type p53 modulates response to chemotherapy in vitro and in vivo. Oncogene 23, 3328–3337 (2004).
Vassilev, L.T. et al. In vivo activation of the p53 pathway by small-molecule antagonists of MDM2. Science 303, 844–848 (2004).
Mantovani, F. et al. Pin1 links the activities of c-Abl and p300 in regulating p73 function. Mol. Cell 14, 625–636 (2004).
Zhao, Y. et al. Acetylation of p53 at lysine 373/382 by the histone deacetylase inhibitor depsipeptide induces expression of p21(Waf1/Cip1). Mol. Cell. Biol. 26, 2782–2790 (2006).
Barlev, N.A. et al. Acetylation of p53 activates transcription through recruitment of coactivators/histone acetyltransferases. Mol. Cell 8, 1243–1254 (2001).
Zhou, X.Z. et al. Pin1-dependent prolyl isomerization regulates dephosphorylation of cdc25 and tau proteins. Mol. Cell 6, 873–883 (2000).
Ard, P.G. et al. Transcriptional regulation of the mdm2 oncogene by p53 requires TRRAP acetyltransferase complexes. Mol. Cell. Biol. 22, 5650–5661 (2002).
Uchida, T. et al. Pin1 and Par14 peptidyl prolyl isomerase inhibitors block cell proliferation. Chem. Biol. 10, 15–24 (2003).
Kern, S.E. et al. Oncogenic forms of p53 inhibit p53-regulated gene expression. Science 256, 827–830 (1992).
Dornan, D., Shimizu, H., Perkins, N.D. & Hupp, T.R. DNA-dependent acetylation of p53 by the transcription coactivator p300. J. Biol. Chem. 278, 13431–13441 (2003).
Bao, L. et al. Prevalent overexpression of prolyl isomerase Pin1 in human cancers. Am. J. Pathol. 164, 1727–1737 (2004).
Dornan, D. & Hupp, T.R. Inhibition of p53-dependent transcription by BOX-I phospho-peptide mimetics that bind to p300. EMBO Rep. 2, 139–144 (2001).
Sun, X.F. et al. A novel p53 germline alteration identified in a late onset breast cancer kindred. Oncogene 13, 407–411 (1996).
Wulf, G., Finn, G., Suizu, F. & Lu, K.P. Phosphorylation-specific prolyl isomerization: is there an underlying theme? Nat. Cell Biol. 7, 435–441 (2005).
Xu, Y.X., Hirose, Y., Zhou, X.Z., Lu, K.P. & Manley, J.L. Pin1 modulates the structure and function of human RNA polymerase II. Genes Dev. 17, 2765–2776 (2003).
Oda, K. et al. p53AIP1, a potential mediator of p53-dependent apoptosis, and its regulation by Ser-46-phosphorylated p53. Cell 102, 849–862 (2000).
Mayo, L.D. et al. Phosphorylation of human p53 at serine 46 determines promoter selection and whether apoptosis is attenuated or amplified. J. Biol. Chem. 280, 25953–25959 (2005).
Samuels-Lev, Y. et al. ASPP proteins specifically stimulate the apoptotic function of p53. Mol. Cell 8, 781–794 (2001).
Geng, Y. et al. Cyclin E ablation in the mouse. Cell 114, 431–443 (2003).
Monte, M. et al. MAGE-A tumor antigens target p53 transactivation function through histone deacetylase recruitment and confer resistance to chemotherapeutic agents. Proc. Natl. Acad. Sci. USA 103, 11160–11165 (2006).
Kaeser, M.D. & Iggo, R.D. Chromatin immunoprecipitation analysis fails to support the latency model for regulation of p53 DNA binding activity in vivo. Proc. Natl. Acad. Sci. USA 99, 95–100 (2002).
Livak, K.J. & Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔC(T) method. Methods 25, 402–408 (2001).
Acknowledgements
We thank C. Schneider, M. Monte and our colleagues at the Laboratorio Nazionale Consorzio Interuniversitario Biotecnologie for advice and discussion; A. Grison, A. Bisso, M. Mioni and A. Soldano for technical assistance; B. Amati (European Institute of Oncology) for critical reading of the manuscript; M. Murphy (Fox Chase Cancer Center), Y. Haupt (Hadassah University Hospital), M. Oren (Weizmann Institute) and G. Blandino (Regina Elena Cancer Institute) for providing reagents; and M. Giacca, M. Lusic, M. Bestagno, N. Gammoh and L. Banks for access to International Centre for Genetic Engineering and Biotechnology facilities. pGFP-p300 was provided by B. Amati; pcDNA3-p53-Pro72, pcDNA3-p53-Arg72 and pcDNA3-p53-R175H were provided by L. Banks; GFP-specific polyclonal antiserum was provided by C. Brancolini (Udine University); polyclonal antibody to HMGA1b was provided by G. Manfioletti (Trieste University); purified recombinant p300 was provided by R. Mantovani (Milan University). J.G. is an International Centre for Genetic Engineering and Biotechnology Fellow. T.C. is a clinical research fellow of Cancer Research UK. This work was supported by grants from the Associazione Italiana per la Ricerca sul Cancro, Association for International Cancer Research UK and Ministero dell'Università e della Ricerca to G.D.S. and by European Community Sixth Framework Programme funding (contract 503576). This publication reflects the authors' views and not necessarily those of the European Community. The European Community is not liable for any use that may be made of the information contained herein.
Author information
Authors and Affiliations
Contributions
F.M., F.T. and J.G. performed biochemical and cell biology experiments, P.S. and M.G. analyzed tissue samples, X.L. produced constructs and reagents, and T.C. supervised P.S. and M.G. and produced reagents. G.D.S. was responsible for the overall project.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8, Supplementary Table 1, Supplementary Methods (PDF 526 kb)
Rights and permissions
About this article
Cite this article
Mantovani, F., Tocco, F., Girardini, J. et al. The prolyl isomerase Pin1 orchestrates p53 acetylation and dissociation from the apoptosis inhibitor iASPP. Nat Struct Mol Biol 14, 912–920 (2007). https://doi.org/10.1038/nsmb1306
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb1306
This article is cited by
-
Cancer and Alzheimer’s disease inverse relationship: an age-associated diverging derailment of shared pathways
Molecular Psychiatry (2021)
-
The Peptidyl-prolyl Isomerase Pin1 in Neuronal Signaling: from Neurodevelopment to Neurodegeneration
Molecular Neurobiology (2021)
-
Ser46 phosphorylation of p53 is an essential event in prolyl-isomerase Pin1-mediated p53-independent apoptosis in response to heat stress
Cell Death & Disease (2019)
-
PIN1 in breast development and cancer: a clinical perspective
Cell Death & Differentiation (2017)
-
The isomerase PIN1 controls numerous cancer-driving pathways and is a unique drug target
Nature Reviews Cancer (2016)