Abstract
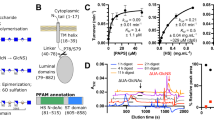
Heparan sulfate (HS) is a glycosaminoglycan that forms a key component of the extracellular matrix (ECM). Breakdown of HS is carried out by heparanase (HPSE), an endo-β-glucuronidase of the glycoside hydrolase 79 (GH79) family. Overexpression of HPSE results in breakdown of extracellular HS and release of stored growth factors and hence is strongly linked to cancer metastasis. Here we present crystal structures of human HPSE at 1.6-Å to 1.9-Å resolution that reveal how an endo-acting binding cleft is exposed by proteolytic activation of latent proHPSE. We used oligosaccharide complexes to map the substrate-binding and sulfate-recognition motifs. These data shed light on the structure and interactions of a key enzyme involved in ECM maintenance and provide a starting point for the design of HPSE inhibitors for use as biochemical tools and anticancer therapeutics.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
08 December 2015
In the version of this article initially published, there were errors in two figures. The schematic in Figure 4a was mistakenly drawn with a β1→3 linkage between the –1 GlcUA and the +1 GlcNS(6S), rather than the correct β1→4 linkage. Supplementary Figure 1 incorrectly gave the names of the GlcNX monomers as N-acetyl-α-D-glucuronic acid and N-sulfo-α-D-glucuronic acid, rather than N-acetyl-α-D-glucosamine and N-sulfo- α-D-glucosamine. These errors have been corrected in the HTML and PDF versions of the article.
References
Lawrence, R. et al. Evolutionary differences in glycosaminoglycan fine structure detected by quantitative glycan reductive isotope labeling. J. Biol. Chem. 283, 33674–33684 (2008).
Yamada, S., Morimoto, H., Fujisawa, T. & Sugahara, K. Glycosaminoglycans in Hydra magnipapillata (Hydrozoa, Cnidaria): demonstration of chondroitin in the developing nematocyst, the sting organelle, and structural characterization of glycosaminoglycans. Glycobiology 17, 886–894 (2007).
Rabenstein, D.L. Heparin and heparan sulfate: structure and function. Nat. Prod. Rep. 19, 312–331 (2002).
Sarrazin, S., Lamanna, W.C. & Esko, J.D. Heparan sulfate proteoglycans. Cold Spring Harb. Perspect. Biol. 3, a004952 (2011).
Iozzo, R.V. Turnover of heparan sulfate proteoglycan in human colon carcinoma cells: a quantitative biochemical and autoradiographic study. J. Biol. Chem. 262, 1888–1900 (1987).
Owens, R.T. & Wagner, W.D. Metabolism and turnover of cell surface-associated heparan sulfate proteoglycan and chondroitin sulfate proteoglycan in normal and cholesterol-enriched macrophages. Arterioscler. Thromb. 11, 1752–1758 (1991).
Cantarel, B.L. et al. The Carbohydrate-Active EnZymes database (CAZy): an expert resource for glycogenomics. Nucleic Acids Res. 37, D233–D238 (2009).
Wilson, J.C., Laloo, A.E., Singh, S. & Ferro, V. 1H NMR spectroscopic studies establish that heparanase is a retaining glycosidase. Biochem. Biophys. Res. Commun. 443, 185–188 (2014).
Okada, Y. et al. Structural recognition by recombinant human heparanase that plays critical roles in tumor metastasis: hierarchical sulfate groups with different effects and the essential target disulfated trisaccharide sequence. J. Biol. Chem. 277, 42488–42495 (2002).
Pikas, D.S., Li, J.P., Vlodavsky, I. & Lindahl, U. Substrate specificity of heparanases from human hepatoma and platelets. J. Biol. Chem. 273, 18770–18777 (1998).
Peterson, S.B. & Liu, J. Multi-faceted substrate specificity of heparanase. Matrix Biol. 32, 223–227 (2013).
Goldshmidt, O. et al. Human heparanase is localized within lysosomes in a stable form. Exp. Cell Res. 281, 50–62 (2002).
Nadav, L. et al. Activation, processing and trafficking of extracellular heparanase by primary human fibroblasts. J. Cell Sci. 115, 2179–2187 (2002).
Sasaki, N., Higashi, N., Taka, T., Nakajima, M. & Irimura, T. Cell surface localization of heparanase on macrophages regulates degradation of extracellular matrix heparan sulfate. J. Immunol. 172, 3830–3835 (2004).
Elkin, M. et al. Heparanase as mediator of angiogenesis: mode of action. FASEB J. 15, 1661–1663 (2001).
Ishai-Michaeli, R., Eldor, A. & Vlodavsky, I. Heparanase activity expressed by platelets, neutrophils, and lymphoma cells releases active fibroblast growth factor from extracellular matrix. Cell Regul. 1, 833–842 (1990).
Goodall, K.J., Poon, I.K., Phipps, S. & Hulett, M.D. Soluble heparan sulfate fragments generated by heparanase trigger the release of pro-inflammatory cytokines through TLR-4. PLoS One 9, e109596 (2014).
Goldshmidt, O. et al. Cell surface expression and secretion of heparanase markedly promote tumor angiogenesis and metastasis. Proc. Natl. Acad. Sci. USA 99, 10031–10036 (2002).
Vlodavsky, I. et al. The impact of heparanese and heparin on cancer metastasis and angiogenesis. Pathophysiol. Haemost. Thromb. 35, 116–127 (2006).
Vlodavsky, I. et al. Mammalian heparanase: gene cloning, expression and function in tumor progression and metastasis. Nat. Med. 5, 793–802 (1999).
Ilan, N., Elkin, M. & Vlodavsky, I. Regulation, function and clinical significance of heparanase in cancer metastasis and angiogenesis. Int. J. Biochem. Cell Biol. 38, 2018–2039 (2006).
Xu, Y.J. et al. N-(4-[4-(1H-Benzoimidazol-2-yl)-arylamino]-methyl-phenyl)-benzamide derivatives as small molecule heparanase inhibitors. Bioorg. Med. Chem. Lett. 16, 404–408 (2006).
Courtney, S.M. et al. 2,3-Dihydro-1,3-dioxo-1H-isoindole-5-carboxylic acid derivatives: a novel class of small molecule heparanase inhibitors. Bioorg. Med. Chem. Lett. 14, 3269–3273 (2004).
Cassinelli, G. et al. Antitumor efficacy of the heparanase inhibitor SST0001 alone and in combination with antiangiogenic agents in the treatment of human pediatric sarcoma models. Biochem. Pharmacol. 85, 1424–1432 (2013).
Hammond, E., Handley, P., Dredge, K. & Bytheway, I. Mechanisms of heparanase inhibition by the heparan sulfate mimetic PG545 and three structural analogues. FEBS Open Bio 3, 346–351 (2013).
Lewis, K.D. et al. A phase II study of the heparanase inhibitor PI-88 in patients with advanced melanoma. Invest. New Drugs 26, 89–94 (2008).
Michikawa, M. et al. Structural and biochemical characterization of glycoside hydrolase family 79 β-glucuronidase from Acidobacterium capsulatum. J. Biol. Chem. 287, 14069–14077 (2012).
Bohlmann, L. et al. Functional and structural characterization of a heparanase. Nat. Chem. Biol. doi:10.1038/nchembio.1956 (2 November 2015).
Fairbanks, M.B. et al. Processing of the human heparanase precursor and evidence that the active enzyme is a heterodimer. J. Biol. Chem. 274, 29587–29590 (1999).
McKenzie, E. et al. Biochemical characterization of the active heterodimer form of human heparanase (Hpa1) protein expressed in insect cells. Biochem. J. 373, 423–435 (2003).
Hulett, M.D. et al. Identification of active-site residues of the pro-metastatic endoglycosidase heparanase. Biochemistry 39, 15659–15667 (2000).
Davies, G. & Henrissat, B. Structures and mechanisms of glycosyl hydrolases. Structure 3, 853–859 (1995).
Nardella, C. et al. Mechanism of activation of human heparanase investigated by protein engineering. Biochemistry 43, 1862–1873 (2004).
Peterson, S. & Liu, J. Deciphering mode of action of heparanase using structurally defined oligosaccharides. J. Biol. Chem. 287, 34836–34843 (2012).
Peterson, S.B. & Liu, J. Unraveling the specificity of heparanase utilizing synthetic substrates. J. Biol. Chem. 285, 14504–14513 (2010).
Davies, G.J., Wilson, K.S. & Henrissat, B. Nomenclature for sugar-binding subsites in glycosyl hydrolases. Biochem. J. 321, 557–559 (1997).
Fairweather, J.K., Hammond, E., Johnstone, K.D. & Ferro, V. Synthesis and heparanase inhibitory activity of sulfated mannooligosaccharides related to the antiangiogenic agent PI-88. Bioorg. Med. Chem. 16, 699–709 (2008).
Hammond, E., Li, C.P. & Ferro, V. Development of a colorimetric assay for heparanase activity suitable for kinetic analysis and inhibitor screening. Anal. Biochem. 396, 112–116 (2010).
Torri, G. et al. Mono- and bidimensional 500 MHz 1H-NMR spectra of a synthetic pentasaccharide corresponding to the binding sequence of heparin to antithrombin-III: evidence for conformational peculiarity of the sulfated iduronate residue. Biochem. Biophys. Res. Commun. 128, 134–140 (1985).
Speciale, G., Thompson, A.J., Davies, G.J. & Williams, S.J. Dissecting conformational contributions to glycosidase catalysis and inhibition. Curr. Opin. Struct. Biol. 28, 1–13 (2014).
Atkins, E.D. & Nieduszynski, I.A. Crystalline structure of heparin. Adv. Exp. Med. Biol. 52, 19–37 (1975).
Mulloy, B., Forster, M.J., Jones, C. & Davies, D.B. N.m.r. and molecular-modelling studies of the solution conformation of heparin. Biochem. J. 293, 849–858 (1993).
Mikhailov, D., Linhardt, R.J. & Mayo, K.H. NMR solution conformation of heparin-derived hexasaccharide. Biochem. J. 328, 51–61 (1997).
Faham, S., Hileman, R.E., Fromm, J.R., Linhardt, R.J. & Rees, D.C. Heparin structure and interactions with basic fibroblast growth factor. Science 271, 1116–1120 (1996).
Schlessinger, J. et al. Crystal structure of a ternary FGF-FGFR-heparin complex reveals a dual role for heparin in FGFR binding and dimerization. Mol. Cell 6, 743–750 (2000).
Dementiev, A., Petitou, M., Herbert, J.M. & Gettins, P.G. The ternary complex of antithrombin–anhydrothrombin–heparin reveals the basis of inhibitor specificity. Nat. Struct. Mol. Biol. 11, 863–867 (2004).
Neurath, H. & Walsh, K.A. Role of proteolytic enzymes in biological regulation (a review). Proc. Natl. Acad. Sci. USA 73, 3825–3832 (1976).
Bieniossek, C., Imasaki, T., Takagi, Y. & Berger, I. MultiBac: expanding the research toolbox for multiprotein complexes. Trends Biochem. Sci. 37, 49–57 (2012).
Dauter, Z., Dauter, M. & Rajashankar, K.R. Novel approach to phasing proteins: derivatization by short cryo-soaking with halides. Acta Crystallogr. D Biol. Crystallogr. 56, 232–237 (2000).
Kabsch, W. Xds. Acta Crystallogr. D Biol. Crystallogr. 66, 125–132 (2010).
Evans, P.R. & Murshudov, G.N. How good are my data and what is the resolution? Acta Crystallogr. D Biol. Crystallogr. 69, 1204–1214 (2013).
Sheldrick, G.M. Experimental phasing with SHELXC/D/E: combining chain tracing with density modification. Acta Crystallogr. D Biol. Crystallogr. 66, 479–485 (2010).
Cowtan, K. The Buccaneer software for automated model building. 1. Tracing protein chains. Acta Crystallogr. D Biol. Crystallogr. 62, 1002–1011 (2006).
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D Biol. Crystallogr. 60, 2126–2132 (2004).
Murshudov, G.N., Vagin, A.A. & Dodson, E.J. Refinement of macromolecular structures by the maximum-likelihood method. Acta Crystallogr. D Biol. Crystallogr. 53, 240–255 (1997).
Chen, V.B. et al. MolProbity: all-atom structure validation for macromolecular crystallography. Acta Crystallogr. D Biol. Crystallogr. 66, 12–21 (2010).
Agirre, J. et al. Privateer: software for the conformational validation of carbohydrate structures. Nat. Struct. Mol. Biol. 22, 833–834 (2015).
McNicholas, S., Potterton, E., Wilson, K.S. & Noble, M.E.M. Presenting your structures: the CCP4mg molecular-graphics software. Acta Crystallogr. D Biol. Crystallogr. 67, 386–394 (2011).
Lebedev, A.A. et al. JLigand: a graphical tool for the CCP4 template-restraint library. Acta Crystallogr. D Biol. Crystallogr. 68, 431–440 (2012).
Acknowledgements
We thank Diamond Light Source for access to beamlines I02, I03 and I04 (proposal mx-9948), which contributed to the results presented here. DH10EMBacY cells used to generate recombinant bacmids were a gift from I. Berger (University of Bristol). The full HPSE gene was obtained from a baculoviral construct from D. Jarvis (University of Wyoming; US National Institutes of Health grant RR005351). G.J.D. and L.W. acknowledge support from the European Research Council through Advanced Grant Glycopoise (AdG 322942 (G.J.D.)). A.M.B. and C.M.V. thank the UK Medical Research Council for funding (MR/K000179/1 (A.M.B.)).
Author information
Authors and Affiliations
Contributions
L.W. and G.J.D. designed and interpreted the experiments. L.W. and C.M.V. cloned, expressed and purified proteins with help from A.M.B. in eukaryotic protein expression facilities. L.W. carried out kinetics experiments and protein crystallizations, and solved the structures of protein and ligand complexes. L.W. and G.J.D. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Molecular and domain composition of HS.
HS is a structurally heterogeneous linear polysaccharide composed of repeating HexUA-(1,4)-GlcNX-(1,4) disaccharide units. (a) Chemical structures of the constituent HS sugars. HexUA is either GlcUA or IdoUA, which differ only in stereochemistry of the 5 position. GlcNX is either GlcNAc or its N-deacetylated-N-sulfated congener GlcNS. IdoUA, GlcNAc and GlcNS are subject to varying degrees of O-sulfation. Also shown is the structure of ΔHexUA, an unsaturated uronic acid formed at the non-reducing end of heparins depolymerized by bacterial heparin lyases, such as the dp4 tetrasaccharide used in this study. (b) Macromolecular 'domain' like organization of mature HS. NA domains are rich in GlcUA and GlcNAc, and show low levels of O-sulfation. NS domains are enriched in IdoUA and GlcNS, with a higher degree of O-sulfation. NA and NS domains are bounded by mixed NA/NS domains, which show intermediate characteristics.
Supplementary Figure 2 Supplemental crystal-structure illustrations.
(a) Ribbon representations of HPSE (blue and gold) superposed against the bacterial exoglucuronidase AcaGH79 (dark red; PDB accession code 3VNY), and bacterial endoglucuronidase BpHPSE (coral; PDB accession code 5BWI). The three GH79 proteins possess a high degree of similarity in their overall fold topology. (b) M09 S05a −2 subsite GlcNS(6S) with density contoured to 1σ (0.25 electrons/Å3) and 2σ (0.51 electrons/Å3). The relative weakness of 6O sulfate density compared to N-sulfate can be seen. Electron densities are REFMAC maximum-likelihood/σÅ weighted 2Fo−Fc syntheses. (c) View of HPSE (colored by secondary structure) along the active site cleft towards the 'positive' end, showing the presence of a symmetry molecule (ice blue) at the opening. This symmetry molecule prevents HPSE interactions in crystallo which involve substrates protruding too far out of the 'positive' end of the cleft.
Supplementary Figure 3 Michaelis-Menten kinetics for HPSE hydrolysis of the HepMers.
Reaction was measured using the reducing end detection dye WST1. Baseline subtractions using a no enzyme control were carried out for all reactions, to control for non-enzymatic paranitrophenol autohydrolysis. Error bars are standard errors of the mean for technical replicates (n=3 for all points). N.d. stands for not determinable.
Supplementary Figure 4 Relationship of HPSE active site residues to those of other GH79 enzymes.
(a) Clustalω (Sievers, F. et al. Mol Syst Biol 7 (2011)) alignment of 4 eukaryotic GH79 sequences, AcaGH79, and BpHPSE. Residues corresponding to those which interact with substrates in human HPSE (as outlined in Figure 4) are highlighted: green where identical with the human sequence, or orange where a non-conserved residue can interact in a similar fashion. Residues of the −1 subsite are hyperconserved, illustrating their requirement for interacting with GlcUA. In contrast, residues of the −2 and +1 subsites are only well conserved amongst the mammalian heparanases. (b) Active site view of the dp4-HPSE complex, with residues colored by conservation to bacterial enzymes AcaGH79 and BpHPSE (green – identical, yellow – partially conserved, red – not conserved). Dp4 ligand is shown in grey.
Supplementary Figure 5 Relationship of HSPE proenzyme linker sequence to those of other GH79 enzymes.
Alignment of 4 eukaryotic GH79 sequences, AcaGH79, and BpHPSE, across the region corresponding to the proenzyme linker of human HPSE (dashed box). Eukaryotic GH79s show an extended sequence at this position, likely corresponding to a proteolytically cleavable linker as in human HPSE. The corresponding AcaGH79 sequence is shorter, and forms a loop which creates part of its exo- binding pocket. The corresponding BpHPSE sequence is effectively absent, and corresponds to a very short loop which reveals the endo- acting binding cleft of this enzyme.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5 and Supplementary Table 1 (PDF 11289 kb)
Rights and permissions
About this article
Cite this article
Wu, L., Viola, C., Brzozowski, A. et al. Structural characterization of human heparanase reveals insights into substrate recognition. Nat Struct Mol Biol 22, 1016–1022 (2015). https://doi.org/10.1038/nsmb.3136
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.3136
This article is cited by
-
Histone H4 induces heparan sulfate degradation by activating heparanase in chlorine gas-induced acute respiratory distress syndrome
Respiratory Research (2022)
-
Chemoenzymatic synthesis of sulfur-linked sugar polymers as heparanase inhibitors
Nature Communications (2022)