Abstract
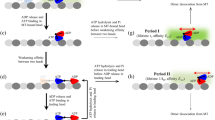
Kinesin is an ATP-driven molecular motor that moves processively along a microtubule. Processivity has been explained as a mechanism that involves alternating single- and double-headed binding of kinesin to microtubules coupled to the ATPase cycle of the motor. The internal load imposed between the two bound heads has been proposed to be a key factor regulating the ATPase cycle in each head. Here we show that external load imposed along the direction of motility on a single kinesin molecule enhances the binding affinity of ADP for kinesin, whereas an external load imposed against the direction of motility decreases it. This coupling between loading direction and enzymatic activity is in accord with the idea that the internal load plays a key role in the unidirectional and cooperative movement of processive motors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Vale, R.D., Reese, T.S. & Sheetz, M.P. Identification of a novel force-generating protein, kinesin, involved in microtubule-based motility. Cell 42, 39–50 (1985).
Brady, S.T. A novel brain ATPase with properties expected for the fast axonal transport motor. Nature 317, 73–75 (1985).
Scholey, J.M., Porter, M.E., Grissom, P.M. & McIntosh, J.R. Identification of kinesin in sea urchin eggs and evidence for its localization in the mitotic spindle. Nature 318, 483–486 (1985).
Hirokawa, N. Kinesin and dynein superfamily proteins and the mechanism of organelle transport. Science 279, 519–526 (1998).
Svoboda, K. & Block, S.M. Force and velocity measured for single kinesin molecules. Cell 77, 773–784 (1994).
Hackney, D.D. Evidence for alternating head catalysis by kinesin during microtubule-stimulated ATP hydrolysis. Proc. Natl. Acad. Sci. USA 91, 6865–6869 (1994).
Rice, S. et al. A structural change in the kinesin motor protein that drives motility. Nature 402, 778–784 (1999).
Vale, R.D. & Milligan, R.A. The way things move: looking under the hood of molecular motor proteins. Science 288, 88–95 (2000).
Mandelkow, E. & Johnson, K.A. The structural and mechanochemical cycle of kinesin. Trends Biochem. Sci. 23, 429–433 (1998).
Uemura, S. et al. Kinesin-microtubule binding depends on both nucleotide state and loading direction. Proc. Natl. Acad. Sci. USA 99, 5977–5981 (2002).
Kawaguchi, K. & Ishiwata, S. Nucleotide-dependent single- to double-headed binding of kinesin. Science 291, 667–669 (2001).
Kawaguchi, K., Uemura, S. & Ishiwata, S. Equilibrium and transition between single- and double-headed binding of kinesin as revealed by single-molecule mechanics. Biophys. J. 84, 1103–1113 (2003).
Hancock, W.O. & Howard, J. Kinesin's processivity results from mechanical and chemical coordination between the ATP hydrolysis cycles of the two motor domains. Proc. Natl. Acad. Sci. USA 96, 13147–13152 (1999).
Derenyi, I. & Vicsek, T. The kinesin walk: a dynamic model with elastically coupled head. Proc. Natl. Acad. Sci. USA 93, 6775–6779 (1996).
Cross, R.A. et al. The conformational cycle of kinesin. Philos. Trans. R. Soc. Lond. B Biol. Sci. 355, 459–464 (2000).
Crevel, I., Carter, N., Schliwa, M. & Cross, R. Coupled chemical and mechanical reaction steps in a processive Neurospora kinesin. EMBO J. 18, 5863–5872 (1999).
Xing, J. et al. Kinesin has three nucleotide-dependent conformations. Implications for strain-dependent release. J. Biol. Chem. 275 35413–35423 (2000).
Ma, Y.Z. & Taylor, E.W. Kinetic mechanism of a monomeric kinesin construct. J. Biol. Chem. 272, 717–723 (1997).
Ma, Y.Z. & Taylor, E.W. Interacting head mechanism of microtubule-kinesin ATPase. J. Biol. Chem. 272, 724–730 (1997).
Askenasy, N. & Koretsky, A.P. Transgenic livers expressing mitochondrial and cytosolic CK: mitochondrial CK modulates free ADP levels. Am. J. Physiol. Cell Physiol. 282, C338–346 (2002).
Visscher, K., Schnitzer, M.J & Block, S.M. Single kinesin molecules studied with a molecular force clamp. Nature 400, 184–189 (1999).
Nishiyama, M., Higuchi, H. & Yanagida, T. Chemomechanical coupling of the forward and backward steps of single kinesin molecules. Nat. Cell Biol. 4, 790–797 (2002).
Rief, M. et al. Myosin-V stepping kinetics: a molecular model for processivity. Proc. Natl. Acad. Sci. USA 97, 9482–9486 (2000).
Schnitzer, M.J., Visscher, K. & Block, S.M. Force production by single kinesin motors. Nat. Cell Biol. 2, 718–723 (2000).
Veigel, C., Wang, F., Bartoo, M.C., Sellers, J.R. & Molloy, J.E. The gated gait of the processive molecular motor, myosin V. Nat. Cell Biol. 4, 59–65 (2002).
Spudich, J.A. & Rock, R.S. A crossbridge too far. Nat. Cell Biol. 4, E8–E10 (2002).
Kojima, H., Muto, E., Higuchi, H. & Yanagida, T. Mechanics of single kinesin molecules measured by optical trapping nanometry. Biophys. J. 73, 2012–2022 (1997).
Weingarten, M.D., Lockwood, A.H., Hwo, S.Y. & Kirschner, M.W. A protein factor essential for microtubule assembly. Proc. Natl. Acad. Sci. USA 72, 1858–1862 (1975).
Hyman, A.A. Preparation of marked microtubules for the assay of the polarity of microtubule-based motors by fluorescence. J. Cell Sci. s14, 125–127 (1991).
Nishizaka, T., Miyata, H., Yoshikawa, H., Ishiwata, S. & Kinosita, K. Jr. Unbinding force of a single motor molecule of muscle measured using optical tweezers. Nature 377, 251–254 (1995).
Acknowledgements
We thank S.S. Rosenfeld and M. Chee for critical reading of the manuscript. We also thank D.D. Hackney and K. Kinosita Jr. for valuable discussion. This research was partly supported by Grants-in-Aid for Specially Promoted Research and for the Bio-venture Project from the Ministry of Education, Sports, Culture, Science and Technology of Japan. S.U. is a recipient of predoctoral fellowship of the Japan Society for the Promotion of Science.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Uemura, S., Ishiwata, S. Loading direction regulates the affinity of ADP for kinesin. Nat Struct Mol Biol 10, 308–311 (2003). https://doi.org/10.1038/nsb911
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsb911
This article is cited by
-
Structural basis of mechano-chemical coupling by the mitotic kinesin KIF14
Nature Communications (2021)
-
Delineating elastic properties of kinesin linker and their sensitivity to point mutations
Scientific Reports (2020)
-
Molecular dynamics simulation of the nanosecond pulsed electric field effect on kinesin nanomotor
Scientific Reports (2019)
-
Embedding dual function into molecular motors through collective motion
Scientific Reports (2017)
-
Design principles governing chemomechanical coupling of kinesin
Scientific Reports (2017)