Key Points
-
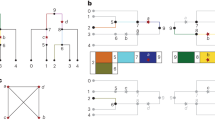
When alleles from different loci are found together in a population, at higher than expected frequencies, they are said to be in linkage disequilibrium (LD). Levels of LD are highest for loci that are physically close in the genome and are reduced by recombination and recurrent mutations.
-
If a disease locus is in LD with linked markers, geneticists can detect the presence of the disease locus by assessing the association between the markers and the disease. This has proved to be a useful strategy for mapping monogenic disease genes, and it is now important to determine whether the same strategy could be applied to complex diseases.
-
One of the issues that needs to be resolved is how best to measure LD. At present, several measures are in use (such as D′, r2 and 4Ner), which means that it is difficult to compare studies.
-
Extensive studies of LD have been conducted in natural populations of Drosophila species, and have shown that LD is variable in different regions of the genome, and is influenced by selective forces, in addition to variation in rates of recombination. Hitchhiking effects can increase LD whilst reducing levels of nucleotide diversity in a region.
-
In humans, simulation studies have provided some useful insights into the factors that are likely to shape patterns of diversity and LD in the human genome. However, it is clear from several studies that simple models of human demography cannot account for the observed levels and variation in nucleotide diversity.
-
These studies emphasize the interplay of many factors in shaping patterns of LD. Factors include genetic drift, selection, demographic factors, variable rates of mutation, recombination and gene conversion.
-
The recent expansion of human genomic resources has provided the impetus for many empirical studies of human LD. Such studies highlight the variation that is seen in different parts of the genome. It is not clear, at present, whether there will also be variation between populations with different demographic histories.
-
The main practical implication of these findings for the study of complex disease is that pilot studies of a genomic region in the population of interest will be essential before a suitable strategy can be devised to look for an association between markers and disease susceptibility.
Abstract
Particular alleles at neighbouring loci tend to be co-inherited. For tightly linked loci, this might lead to associations between alleles in the population — a property known as linkage disequilibrium (LD). LD has recently become the focus of intense study in the hope that it might facilitate the mapping of complex disease loci through whole-genome association studies. This approach depends crucially on the patterns of LD in the human genome. In this review, we draw on empirical studies in humans and Drosophila, as well as simulation studies, to assess the current state of knowledge about patterns of LD, and consider the implications for the use of LD as a mapping tool.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Boehnke, M. Limits of resolution of genetic linkage studies: implications for the positional cloning of human disease genes. Am. J. Hum. Genet. 55, 379–390 (1994).
Kruglyak, L. & Lander, E. S. High-resolution genetic mapping of complex traits. Am. J. Hum. Genet. 56, 1212–1223 (1995).
Kerem, B. et al. Identification of the cystic fibrosis gene: genetic analysis. Science 245, 1073–1080 (1989).
Hastbacka, J. et al. Linkage disequilibrium mapping in isolated founder populations: diastrophic dysplasia in Finland. Nature Genet. 2, 204–211 (1992).
Jorde, L. B. Linkage disequilibrium as a gene mapping tool. Am. J. Hum. Genet. 56, 11–14 (1995).
Jorde, L. B. et al. Linkage disequilibrium in the neurofibromatosis 1 (NF1) region: implications for gene mapping. Am. J. Hum. Genet. 53, 1038–1050 (1993).
Jorde, L. B. et al. Linkage disequilibrium predicts distance in the adenomatous polyposis coli region. Am. J. Hum. Genet. 54, 884–898 (1994).
Jorde, L. B. Linkage disequilibrium and the search for complex disease genes. Genome Res. 10, 1435–1444 (2000).
Watkins, W. S. Linkage disequilibrium patterns vary with chromosomal location: a case study from the von Willebrand factor region. Am. J. Hum. Genet. 55, 348–355 (1994).
Sachidanandam, R. et al. A map of the human genome sequence variation containing 1.42 million single nucleotide polymorphisms. Nature 409, 928–933 (2001).
Risch, N. & Merikangas, K. The future of genetic studies of complex human diseases. Science 273, 1516–1517 (1996).
Kruglyak, L. & Nickerson, D. A. Variation is the spice of life. Nature Genet. 27, 234–236 (2001).
Syvanen, A. C. Accessing genetic variation: genotyping single nucleotide polymorphisms. Nature Rev. Genet. 2, 930–942 (2001).
Lewontin, R. C. The interaction of selection and linkage. I. General considerations; heterotic models. Genetics 49, 49–67 (1964).
Clark, A. G. et al. Haplotype structure and population genetic inferences from nucleotide-sequence variation in human lipoprotein lipase. Am. J. Hum. Genet. 63, 595–612 (1998).
Dunning, A. M. et al. The extent of linkage disequilibrium in four populations with distinct demographic histories. Am. J. Hum. Genet. 67, 1544–1554 (2000).
Moffatt, M. F., Trahern, J. A., Abecasis, G. R. & Cookson, W. O. Single nucleotide polymorphism and linkage disequilibrium within the TCRα/δ locus. Hum. Mol. Genet. 9, 1011–1019 (2000).
Ardlie, K. et al. Lower-than-expected linkage disequilibrium between tightly linked markers in humans suggests a role for gene conversion. Am. J. Hum. Genet. 69, 582–589 (2001).
Collins, A., Lonjou, C. & Morton, N. E. Genetic epidemiology of single-nucleotide polymorphisms. Proc. Natl Acad. Sci. USA 96, 15173–15177 (1999).
Abecasis, G. R. et al. Extent and distribution of linkage disequilibrium in three genomic regions. Am. J. Hum. Genet. 68, 191–197 (2001).
Reich, D. E. et al. Linkage disequilibrium in the human genome. Nature 411, 199–204 (2001).
Stephens, J. C. et al. Haplotype variation and linkage disequilibrium in 313 human genes. Science 293, 489–493 (2001).
Devlin, B. & Risch, N. A comparison of linkage disequilibrium measures for fine-scale mapping. Genomics 29, 311–322 (1995).
Pritchard, J. K. & Przeworski, M. Linkage disequilibrium in humans: models and data. Am. J. Hum. Genet. 69, 1–14 (2001).A comprehensive review that compares empirical data with population models, from a population genetic perspective. Discusses different measures of LD.
Weiss, K. M. & Clark, A. G. Linkage disequilibrium and the mapping of complex human traits. Trends Genet. 18, 19–24 (2002).An excellent critical survey of the issues that surround LD mapping and SNP map design.
Kruglyak, L. Prospects for whole-genome linkage disequilibrium mapping of common disease genes. Nature Genet. 22, 139–144 (1999).
Fearnhead, P. & Donnelly, P. Estimating recombination rates from population genetic data. Genetics 159, 1299–1318 (2001).
Frisse, L. et al. Gene conversion and different population histories may explain the contrast between polymorphism and linkage disequilibrium levels. Am. J. Hum. Genet. 69, 831–843 (2001).
Hudson, R. R. Properties of a neutral allele model with intragenic recombination. Theor. Popul. Biol. 23, 183–201 (1983).
Kuhner, M. K., Yamato, J. & Felsenstein, J. Maximum likelihood estimation of recombination rates from population data. Genetics 156, 1393–1401 (2000).
Long, A. D. & Langley C. H. The power of association studies to detect the contribution of candidate genetic loci to variation in complex traits. Genome Res. 8, 720–731 (1999).A good introduction to the population genetic parameter 4 N e c as a measurement by which to compare LD across different regions.
Nielsen, R. Estimation of population parameters and recombination rates from single nucleotide polymorphisms. Genetics 154, 931–942 (2000).
Wall, J. D. A comparison of estimators of the population recombination rate. Mol. Biol. Evol. 17, 156–163 (2000).
Zapata, C. & Alvarez, C. On the detection of nonrandom associations between DNA polymorphisms in natural populations of Drosophila. Mol. Biol. Evol. 10, 823–841 (1983).
Schaeffer, S. W. & Miller, E. L. Estimates of linkage disequilibrium and the recombination parameter determined from segregating nucleotide sites in the achohol dehydrogenase region of Drosophila pseudoobscura. Genetics 135, 541–542 (1993).
Langley, C. H. et al. Linkage disequilibrium and the site frequency spectra in the su(s) and su(wa) regions of the Drosophila melanogaster X chromosome. Genetics 156, 1837–1852 (2000).
Schaeffer, S. W. et al. Protein variation in ADH and ADH-RELATED in Drosophila pseudoobscura: linkage disequilibrium between single nucleotide polymorphisms and protein alleles. Genetics 159, 673–687 (2001).
Wang, W., Thornton, K., Berry, A. & Long, M. Nucelotide variation along the Drosophila melanogaster fourth chromosome. Science 295, 134–137 (2002).
Begun, D. J. & Aquadro, C. F. Levels of naturally occurring DNA polymorphism correlate with recombination rates in D. melanogaster. Nature 356, 519–520 (1992).
Begun, D. J. & Aquadro, C. F. Evolutionary inferences from DNA variation at the 6-phosphogluconate dehydrogenase locus in natural populations of Drosophila: selection and geographic differentiation. Genetics 136, 155–171 (1994).
Parsch, J., Meiklejohn, C. D. & Hartl, D. L. Patterns of DNA sequence variation suggest the recent action of positive selection in the janus-ocnus region of Drosophila simulans. Genetics 159, 647–657 (2001).
Verrelli, B. C. & Eanes, W. F. Clinal variation for amino acid polymorphisms at the Pgm locus in Drosophila melanogaster. Genetics 157, 1649–1663 (2001).
Charlesworth, B., Morgan, M. T. & Charlesworth, D. The effect of deleterious mutations on neutral molecular variation. Genetics 134, 1289–1303 (1983).
Andolfatto, P. & Przeworsk, M. A genome-wide departure from the standard neutral model in natural populations of Drosophila. Genetics 156, 257–268 (2000).
Payseur, B. A. & Nachman, M. W. Microsatellite variation and recombination rate in the human genome. Genetics 156, 1285–1298 (2000).
Nachman, M. W. Single nucleotide polymorphisms and recombination rate in humans. Trends Genet. 17, 481–485 (2001).
Cargill, M. et al. Characterization of single-nucleotide polymorphisms in coding regions of human genes. Nature Genet. 22, 231–237 (1999).
Wakeley, J., Nielsen, R., Liu-Cordero, S. N. & Ardlie, K. The discovery of single nucleotide polymorphisms and inferences about human demographic history. Am. J. Hum. Genet. 69, 1332–1347 (2001).
Peterson, A. C. et al. The distribution of linkage disequilibrium over anonymous genome regions. Hum. Mol. Genet. 4, 887–894 (1995).
Laan, M. & Paabo, S. Demographic history and linkage disequilibrium in human populations. Nature Genet. 4, 435–438 (1997).
Huttley, G. A., Smith, M. W., Carrington, M. & O'Brien, S. J. A scan for linkage disequilibrium across the human genome. Genetics 152, 1711–1722 (1999).
Patil, N. et al. Blocks of limited haplotype diversity revealed by high resolution scanning of human chromosome 21. Science 294, 1719–1722 (2001).Describes an innovative and exciting experimental approach to whole-chromosome/genome studies of sequence variation and LD, and finds surprisingly limited haplotype diversity over much of chromosome 21 in a global sample.
Goddard, K. A., Hopkins, P. J., Hall, J. M. & Witte, J. S. Linkage disequilibrium and allele frequency distributions for 114 single-nucleotide polymorphisms in five populations. Am. J. Hum. Genet. 66, 216–234 (2000).
Kidd, J. R. et al. Haplotypes and linkage disequilibrium at the phenylalanine hydroxylase locus, PAH, in a global representation of populations. Am. J. Hum. Genet. 66, 1882–1899 (2000).
Lonjou, C., Collins, A. & Morton, N. E. Allelic association between marker loci. Proc. Natl Acad. Sci. USA 96, 1621–1626 (1999).
Bonnen, P. E. et al. Haplotypes at ATM identify coding-sequence variation and indicate a region of extensive linkage disequilibrium. Am. J. Hum. Genet. 67, 1437–1451 (2000).
Taillon-Miller, P. et al. Juxtaposed regions of extensive and minimal linkage disequilibrium in human Xq25 and Xq28. Nature Genet. 25, 324–328 (2000).
Rieder, M. J., Taylor, S. L., Clark, A. G. & Nickerson, D. A. Sequence variation in the human angiotensin converting enzyme. Nature Genet. 22, 59–62 (1999).
Templeton, A. R. et al. Cladistic structure within the human lipoprotein lipase gene and it's implications for phenotypic association studies. Genetics 156, 1259–1275 (2000).
Mateu, E. et al. Worldwide genetic analysis of the CFTR region. Am. J. Hum. Genet. 68, 103–117 (2001).
Nakajima, T. et al. Nucleotide diversity and haplotype structure of the human angiotensinogen gene in two populations. Am. J. Hum. Genet. 70, 109–123 (2002).
Subrahmanyan, L. et al. Sequence variation and linkage disequilibrium in the human T-cell receptor β (TCRB) locus. Am. J. Hum. Genet. 69, 381–395 (2001).
Broman, K. W. & Weber, J. L. Long homozygous chromosomal segments in reference families from the center d'Étude du Polymorphisme Humain. Am. J. Hum. Genet. 65, 1493–1500 (1999).
Goldstein, D. B. Islands of linkage disequilibrium. Nature Genet. 29, 109–111 (2001).
Daly, M. J. et al. High resolution haplotype structure in the human genome. Nature Genet. 29, 229–232 (2001).
Jeffreys, A. J., Kauppi, L. & Neumann, R. Intensely punctate meiotic recombination in the class II region of the major histocompatability complex. Nature Genet. 29, 217–222 (2001).
Johnson, G. C. L. et al. Haplotype tagging for the identification of common disease genes. Nature Genet. 29, 233–237 (2001).
Rioux, J. D. et al. Genetic variation in the 5q31 cytokine gene cluster confers susceptibility to Crohns disease. Nature Genet. 29, 223–228 (2001).
Martin, E. R. et al. SNPing away at complex diseases: analysis of single-nucleotide polymorphisms around APOE in Alzheimer disease. Am. J. Hum. Genet. 67, 383–394 (2000).
Pritchard, J. K. Are rare variants responsible for susceptibility to complex diseases? Am. J. Hum. Genet. 69, 124–137 (2001).
Weiss, K. M. & Terwilliger, J. D. How many SNPs does it take to map a gene with SNPs? Nature Genet. 26, 151–157 (2000).A clear and critical discussion of the very real difficulties that are inherent in identifying genes that contribute to complex traits.
Reich, D. E. & Lander, E. S. On the allelic spectrum of human disease. Trends Genet. 17, 502–510 (2001).
Przeworski, M. & Wall, J. D. Why is there so little intragenic linkage disequilibrium in humans? Genet. Res. 77, 143–151 (2001).
Quintana, P. J. E., Neuwirth, E. A. H. & Grosovsky, A. J. Interchromosomal gene conversion at an endogenous human cell locus. Genetics 158, 757–767 (2001).
Terwilliger, J. D., Zollner, S., Laan, M. & Paabo, S. Mapping genes through the use of linkage disequilibrium generated by genetic drift: 'drift mapping' in small populations with no demographic expansion. Hum. Hered. 48, 138–154 (1998).
Chakraborty, R. & Weiss, K. M. Admixture as a tool for finding linked genes and detecting that difference from allelic association between loci. Proc. Natl Acad. Sci. USA 85, 9119–9123 (1988).
Stephens, J. C., Briscoe, D. & O'Brien, S. J. Mapping by admixture linkage disequilibrium in human populations: limits and guidelines. Am. J. Hum. Genet. 55, 809–824 (1994).
Wilson, J. F. & Goldstein, D. B. Consistent long-range linkage disequilibrium generated by admixture in a Bantu–Semitic hybrid population. Am. J. Hum. Genet. 67, 926–935 (2000).
Pfaff, C. L. et al. Population structure in admixed populations: effect of admixture dynamics on the pattern of linkage disequilibrium. Am. J. Hum. Genet. 68, 198–207 (2001).
Nordborg, M. et al. The extent of linkage disequilibrium in Arabidopsis thaliana. Nature Genet. 30, 190–193 (2002).
Cannon, G. B. The effects of natural selection on linkage disequilibrium and relative fitness in experimental populations of Drosophila melanogaster. Genetics 48, 1201–1216 (1963).
Acknowledgements
We are grateful to M. Eberle for carrying out the simulations presented in Box 2. The manuscript was greatly improved by suggestions from three anonymous referees. Supported in part by a grant from the National Institutes of Mental Health (to L.K.). L.K. is a James S. McDonnell Centennial Fellow. M.S. is supported by the National Human Genome Research Institute.
Author information
Authors and Affiliations
Corresponding authors
Related links
Glossary
- TEST OF ASSOCIATION
-
A study that searches for a population association between a phenotype and a particular allele. In one of the simplest such tests — the case–control study — an excess (or deficiency) of a particular allele in a sample of individuals with the phenotype is looked for, as compared with a sample without the phenotype drawn from the same population.
- HAPLOTYPE
-
The combination of alleles found at neighbouring loci on a single chromosome or haploid DNA molecule.
- EFFECTIVE POPULATION SIZE
-
(Ne). An abstraction, equivalent to the breeding population size of an ideal, randomly mating population, which maintains the same level of variation as observed in the actual population. Ne often bears little relation to the actual number of living or reproducing individuals (the census population size), and is generally much smaller.
- ADMIXTURE
-
The introduction of mates (more properly, their genes) from one previously distinct population (the gene frequencies of which might differ) into another.
- CLINE
-
The exhibition of regular and directional variation in phenotype, or genotype, across a geographical region — for example, steadily increasing pigmentation from temperate to tropical latitudes.
- PANMIXIS
-
The process in a population by which mates are chosen at random with equal probability regardless of geographical location or genotype.
- NUCLEOTIDE DIVERSITY
-
A measure of DNA sequence variation that is influenced both by the number of variable sites and their population allele frequencies in a given stretch of DNA.
- MINOR ALLELE
-
The less frequent of two alleles at a locus.
- BOTTLENECK
-
A period when the size of a population is reduced, generally by a large factor.
Rights and permissions
About this article
Cite this article
Ardlie, K., Kruglyak, L. & Seielstad, M. Patterns of linkage disequilibrium in the human genome. Nat Rev Genet 3, 299–309 (2002). https://doi.org/10.1038/nrg777
Issue Date:
DOI: https://doi.org/10.1038/nrg777
This article is cited by
-
Restriction site-associated DNA sequencing technologies as an alternative to low-density SNP chips for genomic selection: a simulation study in layer chickens
BMC Genomics (2023)
-
Genetic insights into crossbred dairy cattle of Pakistan: exploring allele frequency, linkage disequilibrium, and effective population size at a genome-wide scale
Mammalian Genome (2023)
-
Association analysis of germination level cold stress tolerance and candidate gene identification in Upland cotton (Gossypium hirsutum L.)
Physiology and Molecular Biology of Plants (2022)
-
Fine human genetic map based on UK10K data set
Human Genetics (2022)
-
Further insight into the global variability of the OCA2-HERC2 locus for human pigmentation from multiallelic markers
Scientific Reports (2021)