Key Points
-
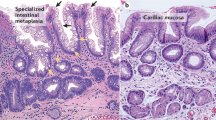
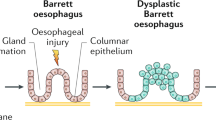
Oesophageal adenocarcinoma (EA) is commonly preceded by replacement of the normal squamous epithelium by a specialized metaplastic columnar-like epithelium, termed Barrett's oesophagus (BE). Individuals with BE have at at least an order of magnitude increased risk of EA.
-
EA is increasing in incidence in Western countries and in Australia, with an approximate doubling in incidence over a ten-year period. However, potential misclassification of adenocarcinomas of the lower oesophagus and gastric cardia complicate interpretations of time trends in incidence. There is much less information concerning trends in the incidence of BE.
-
Gastro-oesophageal reflux disease (GERD) is a strong risk factor for both the development of BE and EA. Other risk factors include obesity; diets low in fruit, vegetables and fibre; and tobacco.
-
The process by which reflux constituents provoke BE and progression to EA are ill-defined. Acid exposure stimulates the formation of a columnar epithelial phenotype and modifies cell proliferation. The role of bile salts might be potentially modulated by the mildly acidic pH conditions that are stimulated by acid-suppressive medication.
-
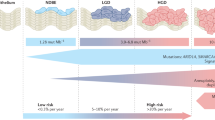
The development and progression of BE is characterized by the acquisition of many molecular changes. These molecular changes could be used as biomarkers to refine surveillance programmes in patients with BE.
-
Evidence indicates that oxidative stress and chronic inflammation promote the development and progression of BE. This affords the opportunity for preventative intervention strategies — potentially involving dietary antioxidants and non-steroidal anti-inflammatory drugs.
Abstract
The incidence of oesophageal adenocarcinoma is increasing rapidly in Western populations. Gastro-oesophageal reflux disease is a strong risk factor for both this tumour and the pre-cancerous lesion Barrett's oesophagus, but the underlying disease mechanisms remain unclear. Developing a better understanding of the aetiology and pathogenesis of Barrett's oesophagus, including the induction of DNA damage and genetic alterations, might provide opportunities for improved management of individuals with this disease. This could include a better rationale for screening and surveillance programmes, as well as targeted intervention strategies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
van Sandick, J. W., van Lanschot, J. J. B., Tytgat, G. N. J., Offerhaus, G. J. A. & Obertop, H. Barrett oesophagus and adenocarcinoma: an overview of epidemiologic, conceptual and clinical issues. Scand. J. Gastroenterol. 36, 51–60 (2001).
Shaheen, N. J., Crosby, M. A., Bozymski, E. M. & Sandler, R. S. Is there publication bias in the reporting of cancer risk in Barrett's esophagus? Gastroenterology 119, 333–338 (2000).
Jankowski, J. A., Provenzale, D. & Moayyedi, P. Esophageal adenocarcinoma arising from Barret's metaplasia has regional variations in the west. Gastroenterology 122, 588–595 (2002).
Devesa, S. S., Blot, W. J. & Fraumeni, J. F. Changing patterns in the incidence of esophageal and gastric carcinoma in the United States. Cancer 83, 2049–2053 (1998).
Botterweck, A. A. M., Schouten, L. J., Volovics, A., Dorant, E. & van den Brandt, P. A. Trends in incidence of adenocarcinoma of the oesophagus and gastric cardia in ten European countries. Int. J. Epidemiol. 29, 645–654 (2000).
Bollschweiler, E., Wolfgarten, E., Gutschow, C. & Holscher, A. H. Demographic variations in the rising incidence of esophageal adenocarcinoma in white males. Cancer 92, 549–555 (2001).
Vizcaino, A. P., Moreno, V., Lambert, R. & Parkin, D. M. Time trends incidence of both major histologic types of esophageal carcinomas in selected countries, 1973–1995. Int. J. Cancer 99, 860–868 (2002). Describes the trends in incidence of EAs in a number of countries and includes a discussion of the difficulties of classification of EAs and the implications for studies of aetiology.
IARC Sci. Publ. no. 155. Cancer in Five Continents, Vol III (Eds Parkin, D. M. et al.) (IARC, Lyon, France, 2002)
Chang, S. S. et al. Unchanging trend of adenocarcinoma of the esophagus and gastric cardia in Taiwan: a 15-year experience in a single center. Dig. Dis. Sci. 47, 735–740 (2002).
Forman, D. Counting cancers at the junction — a problem of routine statistics. Eur. J. Gastroenterol. Hepatol. 14, 1–3 (2002).
Ekstrom, A. M. et al. Evaluating gastric cancer misclassification: a potential explanation for the rise in cardia cancer incidence. J. Natl Cancer Inst. 91, 786–790 (1999).
Mckinney, P. A., Sharp, L., Macfarlane, G. J. & Muir, C. S. Esophageal and gastric cancer in Scotland 1960–90. Br. J. Cancer 71, 411–415 (1995).
Kubo, A. & Corley, D. A. Marked regional variation in adenocarcinomas of the esophagus and the gastric cardia in the United States. Cancer 95, 2096–2102 (2002).
Younes, M., Henson, D. E., Ertan, A. & Miller, C. C. Incidence and survival trends of esophageal carcinoma in the United States: racial and gender differences by histological type. Scand. J. Gastroenterol. 37, 1359–1365 (2002).
El Serag, H. B., Mason, A. C., Petersen, N. & Key, C. R. Epidemiological differences between adenocarcinoma of the oesophagus and adenocarcinoma of the gastric cardia in the USA. Gut 50, 368–372 (2002).
Ormsby, A. H et al. The location and frequency of intestinal metaplasia at the esophagogastric junction in 223 consecutive autopsies: implications for patient treatment and preventative strategies in Barrett's esophagus. Mod. Pathol, 13, 614–620 (2000).
Tytgat, G. N. J. & Hameeteman, W. The neoplastic potential of columnar-lined (Barrett) esophagus. World J. Surg. 16, 308–312 (1992).
Conio, M. et al. Secular trends in the epidemiology and outcome of Barrett's oesophagus in Olmsted County, Minnesota. Gut 48, 304–309 (2001).
Prach, A. T., MacDonald, T. A., Hopwood, D. A. & Johnston, D. A. Increasing incidence of Barrett's oesophagus: education, enthusiasm, or epidemiology? Lancet 350, 933 (1997).
Lagergren, J., Bergstrom, R., Lindgren, A. & Nyren, O. Symptomatic gastroesophageal reflux as a risk factor for esophageal adenocarcinoma. N. Engl. J. Med. 340, 825–831 (1999). Describes the association between reflux symptoms and the risk of developing EAs compared with the risk of developing other tumours of the oesophagus and gastro-oesophageal junction.
Dulai, G. S., Guha, S., Kahn, K. L., Gornbein, J. & Weinstein, W. M. Preoperative prevalence of Barrett's esophagus in esophageal adenocarcinoma: a systematic review. Gastroenterology 122, 26–33 (2002).
Locke, G. R., Talley, N. J., Fett, S. L., Zinsmeister, A. R. & Melton, L. J. Prevalence and clinical spectrum of gastroesophageal reflux: a population-based study in Olmsted County, Minnesota. Gastroenterology 112, 1448–1456 (1997).
Cameron, A. J. et al. Gastroesophageal reflux disease in monozygotic and dizygotic twins. Gastroenterology 122, 55–59 (2002).
Voutilainen, M., Sipponen, P., Mecklin, J. P., Juhola, M. & Farkkila, M. Gastroesophageal reflux disease: prevalence, clinical, endoscopic and histopathological findings in 1,128 consecutive patients referred for endoscopy due to dyspeptic and reflux symptoms. Digestion 61, 6–13 (2000).
Farrow, D. C. et al. Gastroesophageal reflux disease, use of H2 receptor antagonists, and risk of esophageal and gastric cancer. Cancer Causes Control 11, 231–238 (2000).
Coenraad, M. et al. Is Barrett's esophagus characterized by more pronounced acid reflux than severe esophagitis? Am. J. Gastroenterol. 93, 1068–1072 (1998).
Conio, M. et al. Risk factors for Barrett's esophagus: a case-control study. Int. J. Cancer 97, 225–229 (2002). This paper is one of the few to describe risk factors for development of Barrett's oesophagus in a case–control study.
Lieberman, D. A., Oehlke, M. & Helfand, M. Risk factors for Barrett's esophagus in community-based practice. Am. J. Gastroenterol. 92, 1293–1297 (1997).
Campos, G. M. R. et al. Predictive factors of Barrett esophagus: multivariate analysis of 502 patients with gastroesophageal reflux disease. Arch. Surg. 136, 1267–1273 (2001).
Oberg, S. et al. Determinants of intestinal metaplasia within the columnar-lined esophagus. Arch. Surg. 135, 651–655 (2000).
Nebel, O. T., Fornes, M. F. & Castell, D. O. Symptomatic gastroesophageal reflux: incidence and precipitating factors. Am. J. Dig. Dis. 21, 953–956 (1976).
Bashford, J. N. R., Norwood, J. & Chapman, S. R. Why are patients prescribed proton pump inhibitors? Retrospective analysis of link between morbidity and prescribing in the General Practice Research Database. Br. Med. J. 317, 452–456 (1998).
Chow, W. H. et al. Body mass index and risk of adenocarcinomas of the esophagus and gastric cardia. J. Natl Cancer Inst. 90, 150–155 (1998).
Cheng, K. K. et al. A case-control study of oesophageal adenocarcinoma in women: a preventable disease. Br. J. Cancer 83, 127–132 (2000).
Mathus–Vliegen, E. M. H. & Tytgat, G. N. J. Gastro-oesophageal reflux in obese subjects: influence of overweight, weight loss and chronic gastric balloon distension. Scand. J. Gastroenterol. 37, 1246–1252 (2002).
Locke, G. R., Talley, N. J., Fett, S. L., Zinsmeister, A. R. & Melton, L. J. Risk factors associated with symptoms of gastroesophageal reflux. Am. J. Med. 106, 642–649 (1999).
Wilson, L. J., Ma, W. Z. & Hirschowitz, B. I. Association of obesity with hiatal hernia and esophagitis. Am. J. Gastroenterol. 94, 2840–2844 (1999).
Lagergren, J., Bergstrom, R. & Nyren, O. No relation between body mass and gastro-oesophgeal reflux symptoms in a Swedish population based study. Gut 47, 26–29 (2000).
Nilsson, M., Lundegardh, G., Carling, L., Ye, W. & Lagergren, J. Body mass and reflux oesophagitis: an oestrogen-dependent association? Scand. J. Gastroenterol. 37, 626–630 (2002).
Lagergren, J. et al. Association between medications that relax the lower esophageal sphincter and risk for esophageal adenocarcinoma. Ann. Intern. Med. 133, 165–175 (2000)
Terry, P., Lagergren, J., Ye, W. M., Nyren, O. & Wolk, A. Antioxidants and cancers of the esophagus and gastric cardia. Int. J. Cancer 87, 750–754 (2000).
Mayne, S. T. et al. Nutrient intake and risk of subtypes of esophageal and gastric cancer. Cancer Epidemiol. Biomarkers Prev. 10, 1055–1062 (2001).
Zhang, Z. F. et al. Adenocarcinomas of the esophagus and gastric cardia: the role of diet. Nutr. Cancer 27, 298–309 (1997).
Colombo, P., Mangano, M., Bianchi, P. A. & Penagini, R. Effect of calories and fat on postprandial gastro-oesophageal reflux. Scand. J. Gastroenterol. 37, 3–5 (2002).
Gammon, M. D. et al. Tobacco, alcohol, and socioeconomic status and adenocarcinomas of the esophagus and gastric cardia. J. Natl Cancer Inst. 89, 1277–1284 (1997).
Lagergren, J., Bergstrom, R., Lindgren, A. & Nyren, O. The role of tobacco, snuff and alcohol use in the aetiology of cancer of the oesophagus and gastric cardia. Int. J. Cancer 85, 340–346 (2000).
Vaughan, T. L. et al. Risk of esophageal and gastric adenocarcinomas in relation to use of calcium channel blockers, asthma drugs and other medications that promote gastroesophageal reflux. Cancer Epidemiol. Biomarkers Prev. 7, 749–756 (1998).
Romero, Y. et al. Familial aggregation of gastroesophageal reflux in patients with Barrett's esophagus and esophageal adenocarcinoma. Gastroenterology 113, 1449–1456 (1997).
Chak, A. et al. Familial aggregation of Barrett's oesophagus, oesophageal adenocarcinoma, and oesophagogastric junctional adenocarcinoma in Caucasian adults. Gut 51, 323–328 (2002).
Hu, F. Z. et al. Mapping of a gene for severe pediatric gastroesophageal reflux to chromosome 13q14. JAMA 284, 325–334 (2000).
Gutschow, C. A., Schroder, W. & Holscher, A. H. Barrett's esophagus: what is the poison-alkaline, biliary or acidic reflux? Dis. Esophagus 15, 5–9 (2002).
Koak, Y. & Winslet, M. Changing role of in vivo models in columnar-lined lower esophagus. Dis. Esophagus 15, 271–277 (2002).
Tobey, N. A. et al. The role of pepsin in acid injury to esophageal epithelium. Am. J. Gastroenterol. 96, 3062–3070 (2001).
Fitzgerald, R. C., Omary, M. B. & Triadafilopoulos, G. Dynamic effects of acid on Barrett's esophagus: an ex vivo proliferation and differentiation model. J. Clin. Invest. 98, 2120–2128 (1996). Describes the role of acid on biopsies of Barrett's tissue and provides an initial understanding of the possible mechanisms by which reflux could modulate events in the oesophageal mucosa.
Stein, H. J., Kauer, W. K., Feussner, H. & Siewert, J. R. Bile reflux in benign and malignant Barrett's esophagus: effect of medical acid suppression and nissen fundoplication. J. Gastrointest. Surg. 2, 333–341 (1998).
DeMeester, S. R. & DeMeester, T. R. Columnar mucosa and intestinal metaplasia of the esophagus: fifty years of controversy. Ann. Surg. 231, 303–321 (2000).
Nehra, D., Howell, P., Williams, C. P., Pye, J. K. & Beynon, J. Toxic bile acids in gastro-oesophageal reflux disease: influence of gastric acidity. Gut 44, 598–602 (1999).
DeMeester, T. R. Clinical biology of the Barrett's metaplasia, dysplasia to carcinoma sequence. Surg. Oncol. 10, 91–102 (2001).
Kaur, B. S., Ouatu–Lascar, R., Omary, M. B. & Triadafilopoulos, G. Bile salts induce or blunt cell proliferation in Barrett's esophagus in an acid-dependent fashion. Am. J. Physiol. Gastrointest. Liver Physiol. 278, G1000–G1009 (2000).
Menges, M., Muller, M. & Zeitz, M. Increased acid and bile reflux in Barrett's esophagus compared to reflux esophagitis, and effect of proton pump inhibitor therapy. Am. J. Gastroenterol. 96, 331–337 (2001).
Theisen, J. et al. Suppression of gastric acid secretion in patients with gastroesophageal reflux disease results in gastric bacterial overgrowth and deconjugation of bile acids. J. Gastrointest. Surg. 4, 50–54 (2000).
Haigh, C. R. et al. Gastrin induces proliferation in Barrett's metaplasia through activation of the CCK2 receptor. Gastroenterology 124, 615–625 (2003).
Fitzgerald, R. C. et al. Diversity in the oesophageal phenotypic response to gastro- oesophageal reflux: immunological determinants. Gut 50, 451–459 (2002).
Lechner, S. et al. Bile acids mimic oxidative stress induced upregulation of thioredoxin reductase in colon cancer cell lines. Carcinogenesis 23, 1281–1288 (2002).
Bernstein, H. et al. Activation of the promoters of genes associated with DNA damage, oxidative stress, ER stress and protein malfolding by the bile salt, deoxycholate. Toxicol. Lett. 108, 37–46 (1999).
Chen, X. X. et al. Oxidative damage in an esophageal adenocarcinoma model with rats. Carcinogenesis 21, 257–263 (2000). Provides information from an animal model on the possible link between reflux and oxidative damage of cellular macromolecules that could be important in oesophageal carcinogenesis.
Oh, T. Y. et al. Oxidative stress is more important than acid in the pathogenesis of reflux oesophagitis in rats. Gut 49, 364–371 (2001).
Fountoulakis, A. et al. The role of vitamin C in the pathogenesis of Barrett's oesophagus. Br. J. Cancer 86, S51 (2002).
Wetscher, G. J. et al. Reflux esophagitis in humans is a free radical event. Dis. Esophagus 10, 29–32 (1997).
Sihvo, E. I. T. et al. Oxidative stress has a role in malignant transformation in Barrett's oesophagus. Int. J. Cancer 102, 551–555 (2002).
Wilson, K. T., Fu, S. D., Ramanujam, K. S. & Meltzer, S. J. Increased expression of inducible nitric oxide synthase and cyclooxygenase-2 in Barrett's esophagus and associated adenocarcinomas. Cancer Res. 58, 2929–2934 (1998). Reports on alterations in the oesophageal mucosa that are consistent with an effect of oxidative stress in driving the carcinogenic progression from Barrett's oesophagus to adenocarcinoma.
Iijima, K. et al. Dietary nitrate generates potentially mutagenic concentrations of nitric oxide at the gastroesophageal junction. Gastroenterology 122, 1248–1257 (2002).
Olliver, J. R. et al. The role of DNA damage in the development of Barrett's oesophagus and oesophageal adenocarcinoma. Br. J. Cancer 86, S51–S52 (2002).
Carlson, N. et al. Acid suppression therapy may not alter malignant progression in Barrett's metaplasia showing p53 protein accumulation. Am. J. Gastroenterol. 97, 1340–1345 (2002).
Olliver, J., Wild, C. P., Sahay, P., Decter, S. & Hardie, L. J. DNA damage following chromoendoscopy with methylene blue in Barrett's oesophagus. Lancet 362, 373–374 (2003).
Schneider, P. M. et al. Mutations of p53 in Barrett's esophagus and Barrett's cancer: a prospective study of ninety-eight cases. J. Thorac. Cardiovasc. Surg. 111, 323–331 (1996).
Riegman, P. H. J. et al. Genomic alterations in malignant transformation of Barrett's esophagus. Cancer Res. 61, 3164–3170 (2001).
Jenkins, G. J. S. et al. Genetic pathways involved in the progression of Barrett's metaplasia to adenocarcinoma. Br. J. Surg. 89, 824–837 (2002).
Eads, C. A. et al. Epigenetic patterns in the progression of esophageal adenocarcinoma. Cancer Res. 61, 3410–3418 (2001).
Coppola, D., Schreiber, R. H., Mora, L., Dalton, W. & Karl, R. C. Significance of Fas and retinoblastoma protein expression during the progression of Barrett's metaplasia to adenocarcinoma. Ann. Surg. Oncol. 6, 298–304 (1999).
Tselepis, C. et al. Tumour necrosis factor-alpha in Barrett's oesophagus: a potential novel mechanism of action. Oncogene 21, 6071–6081 (2002).
Tselepis, C. et al. Upregulation of the oncogene c-myc in Barrett's adenocarcinoma: induction of c-myc by acidified bile acid in vitro. Gut 52, 174–180 (2003).
Corn, P. G. et al. Frequent hypermethylation of the 5' CpG island of E-cadherin in esophageal adenocarcinoma. Clin. Cancer Res. 7, 2765–2769 (2001).
Bian, Y. S., Osterheld, M. C., Bosman, F. T., Fontolliet, C. & Benhattar, J. Nuclear accumulation of beta-catenin is a common and early event during neoplastic progression of Barrett esophagus. Am. J. Clin. Pathol. 114, 583–590 (2000).
Jankowski, J. A. et al. Molecular evolution of the metaplasia-dysplasia-adenocarcinoma sequence in the esophagus. Am. J. Pathol. 154, 965–973 (1999).
Wijnhoven, B. P. L., Tilanus, H. W. & Dinjens, W. N. M. Molecular biology of Barrett's adenocarcinoma. Ann. Surg. 233, 322–337 (2001).
Souza, R. F., Morales, C. P. & Spechler, S. J. A conceptual approach to understanding the molecular mechanisms of cancer development in Barrett's oesophagus. Aliment. Pharmacol. Ther. 15, 1087–1100 (2001). Provides a detailed review of the molecular changes occurring in the development of oesophageal adenocarcinoma.
Weston, A. P. et al. Long term follow-up of Barrett's high-grade dysplasia. Am. J Gastroenterol. 95, 1888–1893 (2000).
Streitz, J. M. et al. Endoscopic surveillance of Barrett-esophagus: does it help? J. Thorac. Cardiovasc. Surg. 105, 383–388 (1993).
van Sandick, J. W. et al. Impact of endoscopic biopsy surveillance of Barrett's oesophagus on pathological stage and clinical outcome of Barrett's carcinoma. Gut 43, 216–222 (1998).
Corley, D. A., Levin, T. R., Habel, L. A., Weiss, N. S. & Buffler, P. A. Surveillance and survival in Barrett's adenocarcinomas: a population-based study. Gastroenterology 122, 633–640 (2002). Describes a population-based study to address the question of whether surveillance results in decreased mortality in Barrett's patients.
Bani–Hani, K. et al. Barrett's oesophagus: results from a 13-year surveillance programme. Eur. J. Gastroenterol. Hepatol. 12, 649–654 (2000).
Provenzale, D., Kemp, J. A., Arora, S. & Wong, J. B. A guide for surveillance of patients with Barretts-esophagus. Am. J. Gastroenterol. 89, 670–680 (1994).
Streitz, J. M. Jr, Ellis, F. H. Jr, Tilden, R. L. & Erickson, R. V. Endoscopic surveillance of Barrett's esophagus: a cost-effectiveness comparison with mammographic surveillance for breast cancer. Am. J. Gastroenterol. 93, 911–915 (1998).
Inadomi, J. M. et al. Screening and surveillance for Barrett esophagus in high-risk groups: a cost-utility analysis. Ann. Intern. Med. 138, 176–186 (2003).
Ofman, J. J. et al. The economic impact of the diagnosis of dysplasia in Barrett's Esophagus. Am. J. Gastroenterol. 95, 2946–2952 (2000).
Rabinovitch, P. S., Longton, G., Blount, P. L., Levine, D. S. & Reid, B. J. Predictors of progression in Barrett's esophagus III: baseline flow cytometric variables. Am. J. Gastroenterol. 96, 3071–3083 (2001).
Reid, B. J. et al. Predictors of progression in Barrett's esophagus II: baseline 17p (p53) loss of heterozygosity identifies a patient subset at increased risk for neoplastic progression. Am. J. Gastroenterol. 96, 2839–2848 (2001).
Bani–Hani, K. et al. Prospective study of cyclin D1 overexpression in Barrett's esophagus: association with increased risk of adenocarcinoma. J. Natl Cancer Inst. 92, 1316–1321 (2000).
Rudolph, R. E. et al. Serum selenium levels in relation to markers of neoplastic progression among persons with Barrett's Esophagus. J. Natl Cancer Inst. 95, 750–757 (2003).
Ye, W., Chow, W. H., Lagergren, J., Yin, L. & Nyren, O. Risk of adenocarcinomas of the esophagus and gastric cardia in patients with gastroesophageal reflux diseases and after antireflux surgery. Gastroenterology 121, 1286–1293 (2001).
Spechler, S. J. et al. Long-term outcome of medical and surgical therapies for gastroesophageal reflux disease: follow-up of a randomized controlled trial. JAMA 285, 2331–2338 (2001).
Bowers, S. P., Mattar, S. G., Smith, C. D., Waring, J. P. & Hunter, J. G. Clinical and histologic follow-up after antireflux surgery for Barrett's esophagus. J. Gastrointest. Surg. 6, 532–538 (2002).
Ouatu–Lascar, R., Fitzgerald, R. C. & Triadafilopoulos, G. Differentiation and proliferation in Barrett's esophagus and the effects of acid suppression. Gastroenterology 117, 327–335 (1999).
Umansky, M. et al. Proton pump inhibitors reduce cell cycle abnormalities in Barrett's esophagus. Oncogene 20, 7987–7991 (2001).
Cameron, A. J. & Lomboy, C. T. Barrett's esophagus: age, prevalence, and extent of columnar epithelium. Gastroenterology 103, 1241–1245 (1992).
Qualman, S. J., Murray, R. D., Mcclung, H. J. & Lucas, J. Intestinal metaplasia is age related in Barrett's esophagus. Arch. Pathol. Lab. Med. 114, 1236–1240 (1990).
Lee, J. S. et al. Involvement of oxidative stress in experimentally induced reflux esophagitis and Barrett's esophagus: clue for the chemoprevention of esophageal carcinoma by antioxidants. Mutat. Res. 480, 189–200 (2001).
Vaughan, T. L. et al. Nonsteroidal anti-inflammatory drug use, body mass index and anthropometry in relation to genetic and flow cytometric abnormalities in Barrett's esophagus. Cancer Epidemiol. Biomarkers Prev. 11, 745–752 (2002).
Corley, D. A., Kerlikowske, K., Verma, R. & Buffler, P. Protective association of aspirin/NSAIDs and esophageal cancer: a systematic review and meta-analysis. Gastroenterology 124, 47–56 (2003).
Thun, M. J. NSAIDs and esophageal cancer: ready for trials but not yet broad clinical application. Gastroenterology 124, 246–257 (2003).
Gerson, L. B., Shetler, K. & Triadafilopoulos, G. Prevalence of Barrett's Esophagus in asymptomatic individuals. Gastroenterology 123, 461–467 (2002).
Acknowledgements
The authors would like to thank D. Forman for his critical comments on an earlier version of this manuscript and Yorkshire Cancer Research for financial support for research into oesophageal cancer at the Molecular Epidemiology Unit.
Author information
Authors and Affiliations
Corresponding author
Related links
Related links
DATABASES
Cancer.gov
LocusLink
FURTHER INFORMATION
Glossary
- UPPER ENDOSCOPY
-
A procedure whereby a flexible fibre-optic lens is passed via the mouth to visualize the oesophagus, stomach or small intestine.
- GASTRO-OESOPHAGEAL REFLUX DISEASE
-
(GERD). A consequence of chronic reflux, GERD comprises various possible clinical and histological alterations to the lining of the oesophagus. These include erosive oesophagitis, thickening of the oesophageal lining, strictures and ulceration of the oesophagus. GERD can be accompanied by Barrett's oesophagus.
- GASTRO-OESOPHAGEAL REFLUX
-
The passage of contents of the stomach and sometimes the duodenum back in to the oesophagus. It typically occurs after a meal — the lower oesophageal sphincter usually remains closed, but if it relaxes inappropriately, acid, bile and food particles can reflux into the oesophagus. This can irritate the walls of the oesophagus and induce a secondary peristaltic contraction of smooth muscle, causing the symptoms of discomfort or pain that is known as heartburn.
- OESOPHAGITIS
-
Inflammation of the oesophagus provoked by reflux and motility disturbances.
- RECALL BIAS
-
Differences in recollection across individuals or study groups that are the result of altered perception or circumstance because of disease rather than true experience.
- PROTON-PUMP INHIBITORS
-
A class of drugs that specifically bind to the gastric-acid pump (H+/K+-ATPase) of parietal cells and that inhibit gastric-acid secretion.
- BODY-MASS INDEX
-
A measure of obesity as defined by weight (kg)/ height (m2). Individuals with values of 30 and above are defined as clinically obese.
- ANTICHOLINERGICS
-
Agents that inhibit parasympathetic neural activity by blocking the neurotransmitter acetylcholine.
- TH1 RESPONSE
-
A T-helper-1 cell-mediated immune response is mediated by pro-inflammatory cytokines such as interferon-γ, interleukin-1β and tumour necrosis factor-α. It promotes cellular immune responses against intracellular infections and malignancy.
- TH2 RESPONSE
-
A T-helper-2 response involves production of cytokines such as interleukin-4, which stimulate antibody production. TH2 cytokines promote secretory immune responses of mucosal surfaces to extracellular pathogens and allergic reactions.
- ANEUPLOIDY
-
A chromosome complement that is not a simple multiple of the haploid set.
- LOSS OF HETEROZYGOSITY
-
(LOH). Occurs when loss of a particular segment of the genome can be shown by the analysis of a polymorphic marker in that region. If an individual is somatically heterozygous for this marker, but homozygous in the tumour, then there has been 'loss of heterozygosity' in that region. Recurrent LOH of a region indicates the presence of a classical tumour-suppressor gene, although recurrent regional loss is also seen for other reasons (for example, because of the presence of fragile sites).
- ANTI-REFLUX SURGERY
-
A surgical procedure whereby the upper part of the stomach is wrapped around the lower oesophagus to improve gastro-oesophageal competence and to limit reflux.
- META-ANALYSIS
-
A retrospective analysis of the results from different studies, making certain assumptions, to reach a conclusion that is based on the pooled data.
Rights and permissions
About this article
Cite this article
Wild, C., Hardie, L. Reflux, Barrett's oesophagus and adenocarcinoma: burning questions. Nat Rev Cancer 3, 676–684 (2003). https://doi.org/10.1038/nrc1166
Issue Date:
DOI: https://doi.org/10.1038/nrc1166
This article is cited by
-
Downregulation of SODD mediates carnosol-induced reduction in cell proliferation in esophageal adenocarcinoma cells
Scientific Reports (2023)
-
Mouse models of Kras activation in gastric cancer
Experimental & Molecular Medicine (2022)
-
Utility of ancillary studies in the diagnosis and risk assessment of Barrett's esophagus and dysplasia
Modern Pathology (2022)
-
Lipid droplet biogenesis and COX-2 pathway activation are triggered by Barrett’s esophagus and adenocarcinoma, but not esophageal squamous cell carcinoma risk factors
Scientific Reports (2021)
-
Long-term consequences of one anastomosis gastric bypass on esogastric mucosa in a preclinical rat model
Scientific Reports (2020)