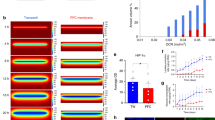
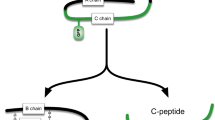
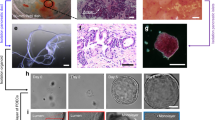
Abstract
Studies on the cellular function of the pancreas are typically performed in vitro on its isolated functional units, the endocrine islets of Langerhans and the exocrine acini. However, these approaches are hampered by preparation-induced changes of cell physiology and the lack of an intact surrounding. We present here a detailed protocol for the preparation of pancreas tissue slices. This procedure is less damaging to the tissue and faster than alternative approaches, and it enables the in situ study of pancreatic endocrine and exocrine cell physiology in a conserved environment. Pancreas tissue slices facilitate the investigation of cellular mechanisms underlying the function, pathology and interaction of the endocrine and exocrine components of the pancreas. We provide examples for several experimental applications of pancreas tissue slices to study various aspects of pancreas cell biology. Furthermore, we describe the preparation of human and porcine pancreas tissue slices for the validation and translation of research findings obtained in the mouse model. Preparation of pancreas tissue slices according to the protocol described here takes less than 45 min from tissue preparation to receipt of the first slices.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
References
Williams, J.A., Korc, M. & Dormer, R.L. Action of secretagogues on a new preparation of functionally intact, isolated pancreatic acini. Am. J. Physiol. 235, 517–524 (1978).
Peikin, S.R., Rottman, A.J., Batzri, S. & Gardner, J.D. Kinetics of amylase release by dispersed acini prepared from guinea pig pancreas. Am. J. Physiol. 235, E743–749 (1978).
Park, M.K., Lee, M. & Petersen, O.H. Morphological and functional changes of dissociated single pancreatic acinar cells: testing the suitability of the single cell as a model for exocytosis and calcium signaling. Cell Calcium 35, 367–379 (2004).
Meda, P., Bruzzone, R., Chanson, M., Bosco, D. & Orci, L. Gap junctional coupling modulates secretion of exocrine pancreas. Proc. Natl. Acad. Sci. USA 84, 4901–4904 (1987).
Logsdon, C.D. & Williams, J.A. Pancreatic acini in short-term culture: regulation by EGF, carbachol, insulin, and corticosterone. Am. J. Physiol. 244, G675–682 (1983).
Halban, P.A. et al. The possible importance of contact between pancreatic islet cells for the control of insulin release. Endocrinology 111, 86–94 (1982).
Jonkers, F.C., Jonas, J.C., Gilon, P. & Henquin, J.C. Influence of cell number on the characteristics and synchrony of Ca2+ oscillations in clusters of mouse pancreatic islet cells. J. Physiol. 520 Part 3: 839–849 (1999).
Zhang, M., Goforth, P., Bertram, R., Sherman, A. & Satin, L. The Ca2+ dynamics of isolated mouse beta cells and islets: implications for mathematical models. Biophys. J. 84, 2852–2870 (2003).
Schuit, F.C., In't Veld, P.A. & Pipeleers, D.G. Glucose stimulates proinsulin biosynthesis by a dose-dependent recruitment of pancreatic beta cells. Proc. Natl. Acad. Sci. USA 85, 3865–3869 (1988).
Ahn, Y.B. et al. Changes in gene expression in beta cells after islet isolation and transplantation using laser-capture microdissection. Diabetologia 50, 334–342 (2007).
Bhagat, L. et al. Heat shock protein 70 prevents secretagogue-induced cell injury in the pancreas by preventing intracellular trypsinogen activation. J. Clin. Invest. 106, 81–89 (2000).
Blinman, T.A. et al. Activation of pancreatic acinar cells on isolation from tissue: cytokine upregulation via p38 MAP kinase. Am. J. Physiol. Cell Physiol. 279, C1993–2003 (2000).
Irving-Rodgers, H.F. et al. Pancreatic islet basement membrane loss and remodeling after mouse islet isolation and transplantation: impact for allograft rejection. Cell Transplant. 23, 59–72 (2012).
Negi, S. et al. Analysis of beta cell gene expression reveals inflammatory signaling and evidence of dedifferentiation following human islet isolation and culture. PLoS ONE 7, e30415 (2012).
Paraskevas, S. et al. Activation and expression of ERK, JNK, and p38 MAP-kinases in isolated islets of Langerhans: implications for cultured islet survival. FEBS Lett. 455, 203–208 (1999).
Raposo do Amaral, A.S. et al. Glutathione ethyl ester supplementation during pancreatic islet isolation improves viability and transplant outcomes in a murine marginal islet mass model. PLoS ONE 8, e55288 (2013).
Speier, S. & Rupnik, M. A novel approach to in situ characterization of pancreatic beta-cells. Pflugers Arch. 446, 553–558 (2003).
Castaneda-Castellanos, D.R., Flint, A.C. & Kriegstein, A.R. Blind patch-clamp recordings in embryonic and adult mammalian brain slices. Nat. Protoc. 1, 532–542 (2006).
de Graaf, I.A. et al. Preparation and incubation of precision-cut liver and intestinal slices for application in drug metabolism and toxicity studies. Nat. Protoc. 5, 1540–1551 (2010).
Edwards, F.A., Konnerth, A., Sakmann, B. & Takahashi, T. A thin slice preparation for patch-clamp recordings from neurones of the mammalian central nervous system. Pflugers Arch. 414, 600–612 (1989).
Lacy, P.E. & Kostianovsky, M. Method for the isolation of intact islets of Langerhans from the rat pancreas. Diabetes 16, 35–39 (1967).
Li, D.S., Yuan, Y.H., Tu, H.J., Liang, Q.L. & Dai, L.J. A protocol for islet isolation from mouse pancreas. Nat. Protoc. 4, 1649–1652 (2009).
Ivanova, A. et al. Age-dependent labeling and imaging of insulin secretory granules. Diabetes 62, 3687–3696 (2013).
Rodriguez-Diaz, R. et al. Noninvasive in vivo model demonstrating the effects of autonomic innervation on pancreatic islet function. Proc. Natl. Acad. Sci. USA 109, 21456–21461 (2012).
Marciniak, A., Selck, C., Friedrich, B. & Speier, S. Mouse pancreas tissue slice culture facilitates long-term studies of exocrine and endocrine cell physiology in situ. PLoS ONE 8, e78706 (2013).
Speier, S., Yang, S.B., Sroka, K., Rose, T. & Rupnik, M. KATP-channels in beta cells in tissue slices are directly modulated by millimolar ATP. Mol. Cell. Endocrinol. 230, 51–58 (2005).
Speier, S., Gjinovci, A., Charollais, A., Meda, P. & Rupnik, M. Cx36-mediated coupling reduces beta cell heterogeneity, confines the stimulating glucose concentration range, and affects insulin release kinetics. Diabetes 56, 1078–1086 (2007).
Dolensek, J., Stozer, A., Skelin Klemen, M., Miller, E.W. & Slak Rupnik, M. The relationship between membrane potential and calcium dynamics in glucose-stimulated beta cell syncytium in acute mouse pancreas tissue slices. PLoS ONE 8, e82374 (2013).
Stozer, A. et al. Functional connectivity in islets of Langerhans from mouse pancreas tissue slices. PLoS Comput. Biol. 9, e1002923 (2013).
Huang, Y.C. et al. In situ electrophysiological examination of pancreatic alpha cells in the streptozotocin-induced diabetes model, revealing the cellular basis of glucagon hypersecretion. Diabetes 62, 519–530 (2013).
Rose, T., Efendic, S. & Rupnik, M. Ca2+-secretion coupling is impaired in diabetic Goto Kakizaki rats. J. Gen. Physiol. 129, 493–508 (2007).
Meneghel-Rozzo, T., Rozzo, A., Poppi, L. & Rupnik, M. In vivo and in vitro development of mouse pancreatic beta cells in organotypic slices. Cell Tissue Res. 316, 295–303 (2004).
Borg, D.J. et al. Mesenchymal stromal cells improve transplanted islet survival and islet function in a syngeneic mouse model. Diabetologia 57, 522–531 (2014).
Karimian, N. et al. Somatostatin receptor type 2 antagonism improves glucagon counterregulation in biobreeding diabetic rats. Diabetes 62, 2968–2977 (2013).
Huang, Y.C., Rupnik, M. & Gaisano, H.Y. Unperturbed islet alpha cell function examined in mouse pancreas tissue slices. J. Physiol. 589, 395–408 (2011).
van der Windt, D.J. et al. Clinical islet xenotransplantation: how close are we? Diabetes 61, 3046–3055 (2012).
Kilkenny, C., Browne, W.J., Cuthill, I.C., Emerson, M. & Altman, D.G. Improving bioscience research reporting: the ARRIVE guidelines for reporting animal research. PLoS Biol. 8, e1000412 (2010).
Gu, G., Dubauskaite, J. & Melton, D.A. Direct evidence for the pancreatic lineage: NGN3+ cells are islet progenitors and are distinct from duct progenitors. Development 129, 2447–2457 (2002).
Muzumdar, M.D., Tasic, B., Miyamichi, K., Li, L. & Luo, L. A global double-fluorescent Cre reporter mouse. Genesis 45, 593–605 (2007).
Zariwala, H.A. et al. A Cre-dependent GCaMP3 reporter mouse for neuronal imaging in vivo. J. Neurosci. 32, 3131–3141 (2012).
Conget, J.I. et al. Deleterious effect of dithizone-DMSO staining on insulin secretion in rat and human pancreatic islets. Pancreas 9, 157–160 (1994).
Botticher, G. et al. Isolation of human islets from partially pancreatectomized patients. J. Vis. Exp. 53, e2962 (2011).
Sturm, D. et al. Improved protocol for laser microdissection of human pancreatic islets from surgical specimens. J. Vis. Exp. 71, e50231 (2013).
Acknowledgements
This work was supported with funds from the Emmy Noether Program of the German Research Foundation (DFG), the CRTD—DFG Research Center for Regenerative Therapies Dresden, Cluster of Excellence, the DFG-SFB/Transregio 127 and the German Ministry for Education and Research (BMBF) to the German Centre for Diabetes Research (DZD) and to the Network of Competence for Diabetes (KKNDm). The work leading to this publication has received support from the Innovative Medicines Initiative Joint Undertaking under grant agreement no. 155005 ('improving beta cell function and identification of diagnostic biomarkers for treatment monitoring in diabetes'; IMIDIA), resources of which are composed of financial contribution from the European Union's Seventh Framework Programme (FP7/2007-2013) and the European Federation of Pharmaceutical Industries and Associations (EFPIA) companies' in-kind contributions. We thank K. Hüttner, C. Kühn, A. Gröbe and K. Pfützner for their excellent technical assistance, and D. Richter, M. Roessler, M. Distler, R. Grutzmann and G. Baretton for providing human pancreatic tissues.
Author information
Authors and Affiliations
Contributions
M.S.R. and S.S. developed the original tissue slice preparation for mouse pancreas; A.M., C.M.C., V.T., J.A.C., C.S., J.S., S.R. and S.S. developed the organotypic culture of adult mouse pancreas slices, the perifusion of slices for calcium imaging, the secretion assays and immunohistochemistry protocols for slices, and the slicing procedure for human and porcine pancreas; T.R. and M.S.R. adapted the tissue slice preparation for rat pancreas; F.E., J.W. and M.S. provided living human pancreas tissue; and A.M., C.M.C., M.S.R. and S.S. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Marciniak, A., Cohrs, C., Tsata, V. et al. Using pancreas tissue slices for in situ studies of islet of Langerhans and acinar cell biology. Nat Protoc 9, 2809–2822 (2014). https://doi.org/10.1038/nprot.2014.195
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2014.195
This article is cited by
-
Improved in vivo imaging method for individual islets across the mouse pancreas reveals a heterogeneous insulin secretion response to glucose
Scientific Reports (2021)
-
Structural and functional polarisation of human pancreatic beta cells in islets from organ donors with and without type 2 diabetes
Diabetologia (2021)
-
Novel metabolic role for BDNF in pancreatic β-cell insulin secretion
Nature Communications (2020)
-
Long-term culture of human pancreatic slices as a model to study real-time islet regeneration
Nature Communications (2020)
-
Modelling the endocrine pancreas in health and disease
Nature Reviews Endocrinology (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.