Abstract
Microglia constitute a highly specialized network of tissue-resident immune cells that is important for the control of tissue homeostasis and the resolution of diseases of the CNS. Little is known about how their spatial distribution is established and maintained in vivo. Here we establish a new multicolor fluorescence fate mapping system to monitor microglial dynamics during steady state and disease. Our findings suggest that microglia establish a dense network with regional differences, and the high regional turnover rates found challenge the universal concept of microglial longevity. Microglial self-renewal under steady state conditions constitutes a stochastic process. During pathology this randomness shifts to selected clonal microglial expansion. In the resolution phase, excess disease-associated microglia are removed by a dual mechanism of cell egress and apoptosis to re-establish the stable microglial network. This study unravels the dynamic yet discrete self-organization of mature microglia in the healthy and diseased CNS.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Accession codes
References
Hanisch, U.K. & Kettenmann, H. Microglia: active sensor and versatile effector cells in the normal and pathologic brain. Nat. Neurosci. 10, 1387–1394 (2007).
Prinz, M. & Priller, J. Microglia and brain macrophages in the molecular age: from origin to neuropsychiatric disease. Nat. Rev. Neurosci. 15, 300–312 (2014).
Ransohoff, R.M. & Perry, V.H. Microglial physiology: unique stimuli, specialized responses. Annu. Rev. Immunol. 27, 119–145 (2009).
Ginhoux, F. et al. Fate mapping analysis reveals that adult microglia derive from primitive macrophages. Science 330, 841–845 (2010).
Gomez Perdiguero, E. et al. Tissue-resident macrophages originate from yolk-sac-derived erythro-myeloid progenitors. Nature 518, 547–551 (2015).
Hashimoto, D. et al. Tissue-resident macrophages self-maintain locally throughout adult life with minimal contribution from circulating monocytes. Immunity 38, 792–804 (2013).
Hagemeyer, N. et al. Transcriptome-based profiling of yolk sac-derived macrophages reveals a role for Irf8 in macrophage maturation. EMBO J. 35, 1730–1744 (2016).
Kierdorf, K. et al. Microglia emerge from erythromyeloid precursors via Pu.1- and Irf8-dependent pathways. Nat. Neurosci. 16, 273–280 (2013).
Schulz, C. et al. A lineage of myeloid cells independent of Myb and hematopoietic stem cells. Science 336, 86–90 (2012).
Sheng, J., Ruedl, C. & Karjalainen, K. Most tissue-resident macrophages except microglia are derived from fetal hematopoietic stem cells. Immunity 43, 382–393 (2015).
Goldmann, T. et al. Origin, fate and dynamics of macrophages at central nervous system interfaces. Nat. Immunol. 17, 797–805 (2016).
Grabert, K. et al. Microglial brain region-dependent diversity and selective regional sensitivities to aging. Nat. Neurosci. 19, 504–516 (2016).
Shemer, A., Erny, D., Jung, S. & Prinz, M. Microglia plasticity during health and disease: an immunological perspective. Trends Immunol. 36, 614–624 (2015).
Bruttger, J. et al. Genetic cell ablation reveals clusters of local self-renewing microglia in the mammalian central nervous system. Immunity 43, 92–106 (2015).
Elmore, M.R.P. et al. Colony-stimulating factor 1 receptor signaling is necessary for microglia viability, unmasking a microglia progenitor cell in the adult brain. Neuron 82, 380–397 (2014).
Sieweke, M.H. & Allen, J.E. Beyond stem cells: self-renewal of differentiated macrophages. Science 342, 1242974 (2013).
Ajami, B., Bennett, J.L., Krieger, C., Tetzlaff, W. & Rossi, F.M.V. Local self-renewal can sustain CNS microglia maintenance and function throughout adult life. Nat. Neurosci. 10, 1538–1543 (2007).
Mildner, A. et al. Microglia in the adult brain arise from Ly-6ChiCCR2+ monocytes only under defined host conditions. Nat. Neurosci. 10, 1544–1553 (2007).
Mildner, A. et al. Distinct and non-redundant roles of microglia and myeloid subsets in mouse models of Alzheimer's disease. J. Neurosci. 31, 11159–11171 (2011).
Davalos, D. et al. ATP mediates rapid microglial response to local brain injury in vivo. Nat. Neurosci. 8, 752–758 (2005).
Nimmerjahn, A., Kirchhoff, F. & Helmchen, F. Resting microglial cells are highly dynamic surveillants of brain parenchyma in vivo. Science 308, 1314–1318 (2005).
Lawson, L.J., Perry, V.H., Dri, P. & Gordon, S. Heterogeneity in the distribution and morphology of microglia in the normal adult mouse brain. Neuroscience 39, 151–170 (1990).
Jung, S. et al. Analysis of fractalkine receptor CX3CR1 function by targeted deletion and green fluorescent protein reporter gene insertion. Mol. Cell. Biol. 20, 4106–4114 (2000).
Parkhurst, C.N. et al. Microglia promote learning-dependent synapse formation through brain-derived neurotrophic factor. Cell 155, 1596–1609 (2013).
Goldmann, T. et al. A new type of microglia gene targeting shows TAK1 to be pivotal in CNS autoimmune inflammation. Nat. Neurosci. 16, 1618–1626 (2013).
Yona, S. et al. Fate mapping reveals origins and dynamics of monocytes and tissue macrophages under homeostasis. Immunity 38, 79–91 (2013).
Wieghofer, P., Knobeloch, K.P. & Prinz, M. Genetic targeting of microglia. Glia 63, 1–22 (2015).
Lawson, L.J., Perry, V.H. & Gordon, S. Turnover of resident microglia in the normal adult mouse brain. Neuroscience 48, 405–415 (1992).
Ajami, B., Bennett, J.L., Krieger, C., McNagny, K.M. & Rossi, F.M.V. Infiltrating monocytes trigger EAE progression, but do not contribute to the resident microglia pool. Nat. Neurosci. 14, 1142–1149 (2011).
Gómez-Nicola, D., Fransen, N.L., Suzzi, S. & Perry, V.H. Regulation of microglial proliferation during chronic neurodegeneration. J. Neurosci. 33, 2481–2493 (2013).
Olmos-Alonso, A. et al. Pharmacological targeting of CSF1R inhibits microglial proliferation and prevents the progression of Alzheimer's-like pathology. Brain 139, 891–907 (2016).
Snippert, H.J. et al. Intestinal crypt homeostasis results from neutral competition between symmetrically dividing Lgr5 stem cells. Cell 143, 134–144 (2010).
Wuidart, A. et al. Quantitative lineage tracing strategies to resolve multipotency in tissue-specific stem cells. Genes Dev. 30, 1261–1277 (2016).
Ghigo, C. et al. Multicolor fate mapping of Langerhans cell homeostasis. J. Exp. Med. 210, 1657–1664 (2013).
Alliot, F., Godin, I. & Pessac, B. Microglia derive from progenitors, originating from the yolk sac, and which proliferate in the brain. Brain Res. Dev. Brain Res. 117, 145–152 (1999).
Gosselin, D. et al. Environment drives selection and function of enhancers controlling tissue-specific macrophage identities. Cell 159, 1327–1340 (2014).
Lavin, Y. et al. Tissue-resident macrophage enhancer landscapes are shaped by the local microenvironment. Cell 159, 1312–1326 (2014).
Xu, J. et al. Temporal-spatial resolution fate mapping reveals distinct origins for embryonic and adult microglia in zebrafish. Dev. Cell 34, 632–641 (2015).
Gallego, B.I. et al. IOP induces upregulation of GFAP and MHC-II and microglia reactivity in mice retina contralateral to experimental glaucoma. J. Neuroinflammation 9, 92 (2012).
Cen, L.P. et al. Bilateral retinal microglial response to unilateral optic nerve transection in rats. Neuroscience 311, 56–66 (2015).
Bellingan, G.J., Caldwell, H., Howie, S.E., Dransfield, I. & Haslett, C. In vivo fate of the inflammatory macrophage during the resolution of inflammation: inflammatory macrophages do not die locally, but emigrate to the draining lymph nodes. J. Immunol. 157, 2577–2585 (1996).
Gautier, E.L., Ivanov, S., Lesnik, P. & Randolph, G.J. Local apoptosis mediates clearance of macrophages from resolving inflammation in mice. Blood 122, 2714–2722 (2013).
Schwab, J.M., Chiang, N., Arita, M. & Serhan, C.N. Resolvin E1 and protectin D1 activate inflammation-resolution programmes. Nature 447, 869–874 (2007).
Drew, P.J. et al. Chronic optical access through a polished and reinforced thinned skull. Nat. Methods 7, 981–984 (2010).
Preibisch, S., Saalfeld, S. & Tomancak, P. Globally optimal stitching of tiled 3D microscopic image acquisitions. Bioinformatics 25, 1463–1465 (2009).
Shankaran, M. et al. Measurement of brain microglial proliferation rates in vivo in response to neuroinflammatory stimuli: application to drug discovery. J. Neurosci. Res. 85, 2374–2384 (2007).
Salic, A. & Mitchison, T.J. A chemical method for fast and sensitive detection of DNA synthesis in vivo. Proc. Natl. Acad. Sci. USA 105, 2415–2420 (2008).
Emmenlauer, M. et al. XuvTools: free, fast and reliable stitching of large 3D datasets. J. Microsc. 233, 42–60 (2009).
Kim, D., Langmead, B. & Salzberg, S.L. HISAT: a fast spliced aligner with low memory requirements. Nat. Methods 12, 357–360 (2015).
Liao, Y., Smyth, G.K. & Shi, W. The Subread aligner: fast, accurate and scalable read mapping by seed-and-vote. Nucleic Acids Res. 41, e108 (2013).
Love, M.I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Alexa, A. & Rahnenfuhrer, J. TopGO: enrichment analysis for gene ontology. R package version 2.26.0 (2016).
Acknowledgements
The authors thank H. Snippert (University Medical Center Utrecht) and H. Clevers (Hubrecht Institute) for the R26RConfetti mouse line, M. Follo and team at Lighthouse Fluorescence Technologies Core Facility, T. Blank for critical feedback, J. Bührer for technical support, and CEMT, University of Freiburg for excellent animal care. T.L.T. is supported by the German Research Foundation (DFG, TA1029/1-1) and Ministry of Science, Research and the Arts, Baden-Wuerttemberg (Research Seed Capital). M.P. is supported by the BMBF Competence Network of Multiple Sclerosis (KKNMS), the Sobek-Stiftung and the DFG (SFB992, SFB1140, Reinhart-Koselleck grant). F.F.-K., J.A.P., S.J., J.P. and M.P. are supported by the SFB/TRR167 “NeuroMac”.
Author information
Authors and Affiliations
Contributions
T.L.T. and M.P. conceived the study. T.L.T. established the methods. D.M., O.R. and T.L.T. designed the software and validated imaging data. T.L.T., J.D., F.F.-K., G.L., S., D.G., M.D., A.D., T.S., A.A.-F., O.S., A.M., A.S. and S.J. performed experiments and/or analyses. T.L.T., L.M.S., J.A.P., D.G., J.P., O.R. and M.P. provided supervision. T.L.T., D.M. and M.P. wrote the manuscript. All authors contributed to the editing of the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Characterization of the Microfetti mouse model.
(a) Gating strategy for 8-color flow analysis of peripheral Ly-6C+ monocytes from Microfetti mice 5-7 days after TAM treatment. Living cells in the forward and side scatter were gated on CD45+CD11b+, followed by CD115+ to separate monocytes from granulocytes. Monocytes were distinguished by Ly-6C expression levels. Representative histograms of Confetti (GFP, YFP, RFP and CFP) expression in Microfetti Ly-6Clo monocytes (left). Confetti signals were gated on Confetti-negative WT counterparts. Expression of Confetti reporter in Ly-6Chi (open circle) and Ly-6Clo (colored) monocytes (right). Each symbol represents one mouse. Animals with Confetti-labeled Ly-6Clo monocytes were used for tracking long-term microglial self-renewal during homeostasis (n = 21 mice; data shown as total Confetti expression in Fig. 2b, 1 dead animal excluded).
(b) Detection of microglial division in vivo. Three Microfetti mice were examined by two-photon laser scanning microscopy via a cortical window six weeks after TAM treatment (see Supplemental Experimental Procedures). A representative image shows RFP+ microglial cells (arrowheads) emerged two days after targeted laser lesion (asterisk). One RFP+ microglial cell (arrow) was no longer detected in the same 200 μm depth volume on Day 2. Scale bar, 50 μm (same scale in both panels).
(c) Quantification of change in area of coverage by GFP+ and RFP+ microglia per minute of in vivo imaging over 30 minutes from Cx3cr1GFP/+ and Microfetti mice, respectively. Each symbol represents one microglial cell. Mann Whitney U-test, P = 0.64. ns, not significant. Data are represented as mean ± SEM. n = 3 mice per genotype.
(d) Representative maximum intensity projection images for c showing two consecutive time points, t1 (red) and t2 (green), with 1 minute interval. Static structures are revealed in the overlay (yellow). Scale bar, 30 μm (same scale in both panels). See also Supplementary Videos 1 and 2.
Supplementary Figure 2 Differential regional stability of the microglial network during steady state.
(a) Representative 2D renderings of Confetti+ (green, yellow, red, and cyan colored balls) and Iba-1 immunolabeled (magenta balls) microglia from 636 μm x 636 μm x 50 μm confocal volumes of the cortex, hippocampus and cerebellum over 1-36 weeks after TAM treatment of 8 weeks old female Microfetti mice.
(b) Confetti+ microglial densities in different brain regions over 36 weeks. Microglial density (y-axis) was obtained from dividing same color cell counts by ring volume of each central ring size (x-axis). Cell densities from recorded data (red line) were plotted against densities derived from 10,000 Monte Carlo (MC) simulation of random distribution of Confetti+ microglia within each dataset (blue box plots represent simulated density distributions: central mark is the median, box represents the 25th to 75th percentiles, whiskers extend up to the 98th percentile and data points above were regarded as outliers). Measured values (red line) that appear above the 98th percentile of MC simulations (blue whiskers), for example at 8 weeks after TAM treatment in the hippocampus and cerebellum where r = 40 μm, indicate the non-random appearance of same color Confetti+ microglial cells and imply that proliferation occurred. This peak represents a single cell replication event in an arbitrary volume of 0.001 mm3. Mean data are shown. n = 5 mice per time point.
(c) Quantitative analysis of randomness of microglial cell proliferation events detected in b. A pair of same color Confetti+ microglial cells with ≤50 μm spatial distance was considered a single cell replication event. Distribution of standard deviation (SD) of a MC simulation (10,000 iterations) of locations of proliferation events derived from a uniform distribution are represented as box plots (blue box plots: central mark is the median, box represents the 25th to 75th percentiles, whiskers represent the 2nd and 98th percentiles, and outliers are plotted in red + signs). Average SD of the distribution of observed replication events to a perfect uniform distribution (black cross) was compared to MC simulation. Where the observed SD falls within the MC range between the whiskers, the locations of proliferation events can be considered randomly distributed.
Supplementary Figure 3 Spatiotemporal analysis of microglial expansion and redistribution in the injured facial nucleus.
(a) Coronal brain section of a Cx3cr1GFP/+ mouse 7d post facial nerve lesion depicts areas analyzed for kinetics of microglial proliferation in the facial nucleus degeneration model. Microglial cells were marked by GFP (green). Neurons were immunolabeled with NeuN (blue) to locate the facial nucleus in the pons. The facial nucleus, a 200 μm broad perinuclear zone, and the parvicellular reticular nucleus, alpha part (PCRtA) were isolated in equal dimensions on lesion and contralateral sides for quantification of GFP+ microglia using the Imaris software.
(b) Quantification of GFP+ microglial cells in contralateral (black) and injured (red) facial nuclei from onset (1 to 2d) to peak (7 and 14d) to resolution of injury (60d) in 8-14 weeks old female Cx3cr1GFP/+ mice.
(c) Representative confocal images of microglia (GFP, green) with DAPI nuclear counterstain (blue) in matched facial nuclei during the time course of injury.
(d) Kinetics of microglial proliferation indicated by Ki67 immunohistochemistry.
(e) Representative confocal images for d. Microglia took on less ramified and more amoeboid morphology at 2 d after the lesion. GFP+ microglia (green), Ki67 (magenta) and DAPI (blue).
(f) Kinetics of microglial activation indicated by MHC class II immunohistochemistry.
(g) Representative confocal images for f. Microglia were highly activated and assumed a rod-like or amoeboid morphology 7d following nerve transection. GFP+ microglia (green), MHC class II (magenta) and DAPI (blue).
Data are represented as mean ± SEM in b, d and f. n = 4 (1 d), 5 (2 d), 6 (7 d), 5 (14 d) and 7 (60 d) mice pooled from five experiments, 3-6 sections each. Two-way ANOVA, * P = 0.0358 in b, ** P = 0.0036 in b, 0.00110 in f, *** P < 0.001 in b, d and f. Scale bars, 50 μm in c, 20 μm in e and g.
Supplementary Figure 4 Distribution of EdU+ GFP+ microglial cells outside of facial nuclei at 7 and 60 d after lesion.
(a) Quantification of EdU+ GFP+ microglial cells external of the facial nuclei (in female Cx3cr1GFP/+ mice injured at 8-14 weeks old) at peak of microgliosis (7 d) and recovery (60 d). Each black and red symbol represents one paired count. Data are pooled from two experiments and shown as mean ± SEM. n = 8 (7 d) and 9 (60 d) mice. Two-way ANOVA, time point *** P = 0.001, lesion *** P < 0.0001.
(b) Representative overview images for a showing lesion and contralateral pons. Dotted lines demarcate the facial nuclei. High magnification insets show EdU+ GFP+ microglial cells in the intermediate reticular nucleus. Scale bars, 500 μm, 50 μm in inset.
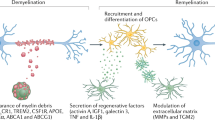
Supplementary Figure 5 Graphical abstract of context-dependent microglial renewal and clonal expansion.
Microglial renewal occurs randomly and steadily throughout the healthy CNS with regional differences in rate. The onset of neurodegeneration triggers rapid clonal expansion and activation of microglial cells. During recovery the altered microglial network gradually returns to homeostatic cell density and a non-activated state. Excess microglia associated with disease are eliminated via cell egress and local apoptotic cell death to regain steady state microglial interfaces in the CNS.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5 and Supplementary Tables 1–3. (PDF 1716 kb)
Supplementary Software
Supplementary Software. (ZIP 10418 kb)
Supplementary Video 1
Thirty-minute time lapse recording of steady state GFP+ adult microglial cells in a CX3CR1GFP/wt mouse via a cranial window. (MOV 562 kb)
Supplementary Video 2
Thirty-minute time lapse recording of a steady state RFP+ adult microglial cell in a Microfetti mouse via a cranial window. (MOV 338 kb)
Rights and permissions
About this article
Cite this article
Tay, T., Mai, D., Dautzenberg, J. et al. A new fate mapping system reveals context-dependent random or clonal expansion of microglia. Nat Neurosci 20, 793–803 (2017). https://doi.org/10.1038/nn.4547
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.4547
This article is cited by
-
Tmem119 expression is downregulated in a subset of brain metastasis-associated microglia
BMC Neuroscience (2024)
-
The aging mouse CNS is protected by an autophagy-dependent microglia population promoted by IL-34
Nature Communications (2024)
-
Multiomic spatial landscape of innate immune cells at human central nervous system borders
Nature Medicine (2024)
-
Prenatal Programming of Monocyte Chemotactic Protein-1 Signaling in Autism Susceptibility
Molecular Neurobiology (2024)
-
Spatio-temporal dynamics of microglia phenotype in human and murine cSVD: impact of acute and chronic hypertensive states
Acta Neuropathologica Communications (2023)