Abstract
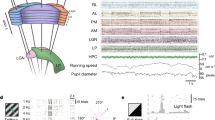
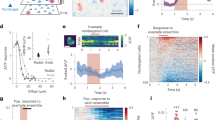
Neuronal responses during sensory processing are influenced by both the organization of intracortical connections and the statistical features of sensory stimuli. How these intrinsic and extrinsic factors govern the activity of excitatory and inhibitory populations is unclear. Using two-photon calcium imaging in vivo and intracellular recordings in vitro, we investigated the dependencies between synaptic connectivity, feature selectivity and network activity in pyramidal cells and fast-spiking parvalbumin-expressing (PV) interneurons in mouse visual cortex. In pyramidal cell populations, patterns of neuronal correlations were largely stimulus-dependent, indicating that their responses were not strongly dominated by functionally biased recurrent connectivity. By contrast, visual stimulation only weakly modified co-activation patterns of fast-spiking PV cells, consistent with the observation that these broadly tuned interneurons received very dense and strong synaptic input from nearby pyramidal cells with diverse feature selectivities. Therefore, feedforward and recurrent network influences determine the activity of excitatory and inhibitory ensembles in fundamentally different ways.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Markram, H. et al. Interneurons of the neocortical inhibitory system. Nat. Rev. Neurosci. 5, 793–807 (2004).
Gonchar, Y., Wang, Q. & Burkhalter, A. Multiple distinct subtypes of GABAergic neurons in mouse visual cortex identified by triple immunostaining. Front. Neuroanat. 1, 3 (2007).
Sillito, A.M. The contribution of inhibitory mechanisms to the receptive field properties of neurones in the striate cortex of the cat. J. Physiol. (Lond.) 250, 305–329 (1975).
Priebe, N.J. & Ferster, D. Inhibition, spike threshold, and stimulus selectivity in primary visual cortex. Neuron 57, 482–497 (2008).
Ozeki, H., Finn, I.M., Schaffer, E.S., Miller, K.D. & Ferster, D. Inhibitory stabilization of the cortical network underlies visual surround suppression. Neuron 62, 578–592 (2009).
Liu, B. et al. Intervening inhibition underlies simple-cell receptive field structure in visual cortex. Nat. Neurosci. 13, 89–96 (2010).
Holmgren, C., Harkany, T., Svennenfors, B. & Zilberter, Y. Pyramidal cell communication within local networks in layer 2/3 of rat neocortex. J. Physiol. (Lond.) 551, 139–153 (2003).
Thomson, A.M. & Lamy, C. Functional maps of neocortical local circuitry. Front. Neurosci. 1, 19–42 (2007).
Oswald, A.-M.M., Doiron, B., Rinzel, J. & Reyes, A.D. Spatial profile and differential recruitment of GABAB modulate oscillatory activity in auditory cortex. J. Neurosci. 29, 10321–10334 (2009).
Yoshimura, Y. & Callaway, E.M. Fine-scale specificity of cortical networks depends on inhibitory cell type and connectivity. Nat. Neurosci. 8, 1552–1559 (2005).
Ko, H. et al. Functional specificity of local synaptic connections in neocortical networks. Nature 473, 87–91 (2011).
Yoshimura, Y., Dantzker, J.L.M. & Callaway, E.M. Excitatory cortical neurons form fine-scale functional networks. Nature 433, 868–873 (2005).
Gilbert, C.D. & Wiesel, T.N. Columnar specificity of intrinsic horizontal and corticocortical connections in cat visual cortex. J. Neurosci. 9, 2432–2442 (1989).
Alonso, J.M., Usrey, W.M. & Reid, R.C. Rules of connectivity between geniculate cells and simple cells in cat primary visual cortex. J. Neurosci. 21, 4002–4015 (2001).
Alonso, J.M. & Martinez, L.M. Functional connectivity between simple cells and complex cells in cat striate cortex. Nat. Neurosci. 1, 395–403 (1998).
Hirsch, J.A. et al. Functionally distinct inhibitory neurons at the first stage of visual cortical processing. Nat. Neurosci. 6, 1300–1308 (2003).
Cardin, J.A., Palmer, L.A. & Contreras, D. Stimulus feature selectivity in excitatory and inhibitory neurons in primary visual cortex. J. Neurosci. 27, 10333–10344 (2007).
Nowak, L.G., Sanchez-Vives, M.V. & McCormick, D.A. Lack of orientation and direction selectivity in a subgroup of fast-spiking inhibitory interneurons: cellular and synaptic mechanisms and comparison with other electrophysiological cell types. Cereb. Cortex 18, 1058–1078 (2008).
Sohya, K., Kameyama, K., Yanagawa, Y., Obata, K. & Tsumoto, T. GABAergic neurons are less selective to stimulus orientation than excitatory neurons in layer II/III of visual cortex, as revealed by in vivo functional Ca2+ imaging in transgenic mice. J. Neurosci. 27, 2145–2149 (2007).
Kerlin, A.M., Andermann, M.L., Berezovskii, V.K. & Reid, R.C. Broadly tuned response properties of diverse inhibitory neuron subtypes in mouse visual cortex. Neuron 67, 858–871 (2010).
Ma, W. et al. Visual representations by cortical somatostatin inhibitory neurons—selective but with weak and delayed responses. J. Neurosci. 30, 14371–14379 (2010).
Runyan, C.A. et al. Response features of parvalbumin-expressing interneurons suggest precise roles for subtypes of inhibition in visual cortex. Neuron 67, 847–857 (2010).
Bock, D.D. et al. Network anatomy and in vivo physiology of visual cortical neurons. Nature 471, 177–182 (2011).
Tsodyks, M., Kenet, T., Grinvald, A. & Arieli, A. Linking spontaneous activity of single cortical neurons and the underlying functional architecture. Science 286, 1943–1946 (1999).
Ch'ng, Y.H. & Reid, R.C. Cellular imaging of visual cortex reveals the spatial and functional organization of spontaneous activity. Front. Integr. Neurosci. 4, 20 (2010).
Fiser, J., Chiu, C. & Weliky, M. Small modulation of ongoing cortical dynamics by sensory input during natural vision. Nature 431, 573–578 (2004).
MacLean, J.N., Watson, B.O., Aaron, G.B. & Yuste, R. Internal dynamics determine the cortical response to thalamic stimulation. Neuron 48, 811–823 (2005).
Luczak, A., Barthó, P. & Harris, K.D. Spontaneous events outline the realm of possible sensory responses in neocortical populations. Neuron 62, 413–425 (2009).
Jermakowicz, W.J., Chen, X., Khaytin, I., Bonds, A.B. & Casagrande, V.A. Relationship between spontaneous and evoked spike-time correlations in primate visual cortex. J. Neurophysiol. 101, 2279–2289 (2009).
Smith, M.A. & Kohn, A. Spatial and temporal scales of neuronal correlation in primary visual cortex. J. Neurosci. 28, 12591–12603 (2008).
Kohn, A. & Smith, M.A. Stimulus dependence of neuronal correlation in primary visual cortex of the macaque. J. Neurosci. 25, 3661–3673 (2005).
Nauhaus, I., Busse, L., Carandini, M. & Ringach, D.L. Stimulus contrast modulates functional connectivity in visual cortex. Nat. Neurosci. 12, 70–76 (2009).
Renart, A. et al. The asynchronous state in cortical circuits. Science 327, 587–590 (2010).
Ecker, A.S. et al. Decorrelated neuronal firing in cortical microcircuits. Science 327, 584–587 (2010).
Ohiorhenuan, I.E. et al. Sparse coding and high-order correlations in fine-scale cortical networks. Nature 466, 617–621 (2010).
Stosiek, C., Garaschuk, O., Holthoff, K. & Konnerth, A. In vivo two-photon calcium imaging of neuronal networks. Proc. Natl. Acad. Sci. USA 100, 7319–7324 (2003).
Ohki, K., Chung, S., Ch'ng, Y.H., Kara, P. & Reid, R.C. Functional imaging with cellular resolution reveals precise micro-architecture in visual cortex. Nature 433, 597–603 (2005).
Mrsic-Flogel, T.D. et al. Homeostatic regulation of eye-specific responses in visual cortex during ocular dominance plasticity. Neuron 54, 961–972 (2007).
Madisen, L. et al. A robust and high-throughput Cre reporting and characterization system for the whole mouse brain. Nat. Neurosci. 13, 133–140 (2010).
Vogelstein, J.T. et al. Fast nonnegative deconvolution for spike train inference from population calcium imaging. J. Neurophysiol. 104, 3691–3704 (2010).
Niell, C.M. & Stryker, M.P. Highly selective receptive fields in mouse visual cortex. J. Neurosci. 28, 7520–7536 (2008).
Galarreta, M. & Hestrin, S. A network of fast-spiking cells in the neocortex connected by electrical synapses. Nature 402, 72–75 (1999).
Gibson, J.R., Beierlein, M. & Connors, B.W. Two networks of electrically coupled inhibitory neurons in neocortex. Nature 402, 75–79 (1999).
Gentet, L.J., Avermann, M., Matyas, F., Staiger, J.F. & Petersen, C.C.H. Membrane potential dynamics of GABAergic neurons in the barrel cortex of behaving mice. Neuron 65, 422–435 (2010).
Zohary, E., Shadlen, M.N. & Newsome, W.T. Correlated neuronal discharge rate and its implications for psychophysical performance. Nature 370, 140–143 (1994).
Poulet, J.F.A. & Petersen, C.C.H. Internal brain state regulates membrane potential synchrony in barrel cortex of behaving mice. Nature 454, 881–885 (2008).
Chelaru, M.I. & Dragoi, V. Efficient coding in heterogeneous neuronal populations. Proc. Natl. Acad. Sci. USA 105, 16344–16349 (2008).
Nimmerjahn, A., Kirchhoff, F., Kerr, J.N.D. & Helmchen, F. Sulforhodamine 101 as a specific marker of astroglia in the neocortex in vivo. Nat. Methods 1, 31–37 (2004).
Brainard, D.H. The psychophysics toolbox. Spat. Vis. 10, 433–436 (1997).
Tanahira, C. et al. Parvalbumin neurons in the forebrain as revealed by PV-Cre transgenic mice. Neurosci. Res. 63, 213–223 (2009).
Acknowledgements
We thank T. Margrie, A. Arenz and E. Rancz for help with in vivo electrophysiology, J. Sjöström and K. Buchanan for help with in vitro electrophysiology, J. Rothman for NeuroMatic software, and M. Hübener and M. Sahani for comments on an earlier version of this manuscript. This work was supported by the Wellcome Trust (T.D.M.-F.), the European Research Council (T.D.M.-F.), the European Molecular Biology Organization (S.B.H.) and the Humboldt Foundation (S.B.H.). We also received funding from the European Community's Seventh Framework Programme (FP2007-2013) under grant agreement no. 223326 (T.D.M.-F.).
Author information
Authors and Affiliations
Contributions
S.B.H. and H.K. performed all in vivo and slice experiments. S.B.H., H.K., N.A.L. and T.D.M.-F. analyzed the data. H.R. carried out antibody labeling. B.P. developed software for visual stimulation, image acquisition and image analysis. J.V. developed spike inference algorithms. E.L. and H.Z. generated and supplied the mice. S.B.H., H.K., N.A.L. and T.D.M.-F. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–9 (PDF 1762 kb)
Rights and permissions
About this article
Cite this article
Hofer, S., Ko, H., Pichler, B. et al. Differential connectivity and response dynamics of excitatory and inhibitory neurons in visual cortex. Nat Neurosci 14, 1045–1052 (2011). https://doi.org/10.1038/nn.2876
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.2876
This article is cited by
-
Subfield-specific interneuron circuits govern the hippocampal response to novelty in male mice
Nature Communications (2024)
-
Local origin of excitatory–inhibitory tuning equivalence in a cortical network
Nature Neuroscience (2024)
-
The gephyrin scaffold modulates cortical layer 2/3 pyramidal neuron responsiveness to single whisker stimulation
Scientific Reports (2024)
-
Synaptic wiring motifs in posterior parietal cortex support decision-making
Nature (2024)
-
Multimodal Nature of the Single-cell Primate Brain Atlas: Morphology, Transcriptome, Electrophysiology, and Connectivity
Neuroscience Bulletin (2024)