Abstract
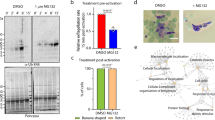
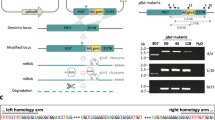
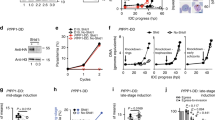
To enhance the repertoire of molecular tools for studying malaria parasite biology, we adapted a ligand-regulatable FKBP protein destabilization domain (ddFKBP) for use in P. falciparum. We destabilized the reporter yellow fluorescent protein (YFP) and the P. falciparum protease falcipain-2 in a ligand-reversible manner by tagging with ddFKBP. The swollen food vacuole phenotype of falcipain-2 knockout parasites could be rescued in a Shld1 ligand–dependent fashion by falcipain-2–ddFKBP expression.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Crabb, B.S. et al. Methods Mol. Biol. 270, 263–276 (2004).
Meissner, M. et al. Proc. Natl. Acad. Sci. USA 102, 2980–2985 (2005).
Ullu, E., Tschudi, C. & Chakraborty, T. Cell. Microbiol. 6, 509–519 (2004).
Banaszynski, L.A., Chen, L.C., Maynard-Smith, L.A., Ooi, A.G. & Wandless, T.J. Cell 126, 995–1004 (2006).
Bell, A., Wernli, B. & Franklin, R.M. Biochem. Pharmacol. 48, 495–503 (1994).
Liu, J., Istvan, E.S., Gluzman, I.Y., Gross, J. & Goldberg, D.E. Proc. Natl. Acad. Sci. USA 103, 8840–8845 (2006).
vanDooren, G.G. et al. Mol. Microbiol. 57, 405–419 (2005).
Herm-Goetz, A. et al. Nat. Methods advance online publication 11 November 2007 (doi:10.1038/nmeth1134).
Sijwali, P.S. & Rosenthal, P.J. Proc. Natl. Acad. Sci. USA 101, 4384–4389 (2004).
Klemba, M., Beatty, W., Gluzman, I. & Goldberg, D.E. J. Cell Biol. 164, 47–56 (2004).
Acknowledgements
We thank I. Gluzman (Washington University) for help with transfections, E. Istvan (Washington University) for falcipain-2 knockout parasites, T. Wandless (Stanford University) for FKBP plasmids pBMN L106P-YFP iHcRed-t and pBMN YFP-L106P iHcRed-t as well as for Shld1 compound, Jacobus Pharmaceuticals for WR99210, M. Drew (Washington University) for microscopy advice, G. McFadden (University of Melbourne) for plasmids pCHDR-3/4 and PfHSP86 5′-pENTR4/1, MR4 and J. Adams (University of South Florida) for anti-BiP, and M. Meissner (University of Heidelburg) for helpful comments. This work was supported by US National Institutes of Health grant AI47798.
Author information
Authors and Affiliations
Contributions
C.M.A. designed and executed experiments and wrote the manuscript. D.E.G. designed experiments and wrote the manuscript.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figure 1, Supplementary Methods (PDF 1149 kb)
Rights and permissions
About this article
Cite this article
Armstrong, C., Goldberg, D. An FKBP destabilization domain modulates protein levels in Plasmodium falciparum. Nat Methods 4, 1007–1009 (2007). https://doi.org/10.1038/nmeth1132
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmeth1132
This article is cited by
-
Distinct evolution of type I glutamine synthetase in Plasmodium and its species-specific requirement
Nature Communications (2023)
-
An intein-split transactivator for intersectional neural imaging and optogenetic manipulation
Nature Communications (2022)
-
An essential vesicular-trafficking phospholipase mediates neutral lipid synthesis and contributes to hemozoin formation in Plasmodium falciparum
BMC Biology (2021)
-
Improved Plasmodium falciparum dilution cloning through efficient quantification of parasite numbers and c-SNARF detection
Malaria Journal (2021)
-
The catalytic subunit of Plasmodium falciparum casein kinase 2 is essential for gametocytogenesis
Communications Biology (2021)