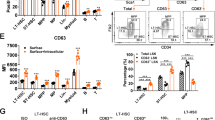
Abstract
Gain of Wnt signaling through β-catenin has been ascribed a critical function in the stimulation of hematopoietic stem cell self-renewal, whereas loss of β-catenin is reportedly dispensable for hematopoiesis. Here we have used conditional mouse genetics and transplantation assays to demonstrate that constitutive activation of β-catenin blocked multilineage differentiation, leading to the death of mice. Blood cell depletion was accompanied by failure of hematopoietic stem cells to repopulate irradiated hosts and to differentiate into mature cells. Activation of β-catenin enforced cell cycle entry of hematopoietic stem cells, thus leading to exhaustion of the long-term stem cell pool. Our data suggest that fine-tuned Wnt stimulation is essential for hematopoiesis and is thus critical for therapeutic hematopoietic stem cell population expansion.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Morrison, S.J. & Weissman, I.L. The long-term repopulating subset of hematopoietic stem cells is deterministic and isolatable by phenotype. Immunity 1, 661–673 (1994).
Reya, T. & Clevers, H. Wnt signalling in stem cells and cancer. Nature 434, 843–850 (2005).
Staal, F.J. & Clevers, H.C. WNT signalling and haematopoiesis: a WNT-WNT situation. Nat. Rev. Immunol. 5, 21–30 (2005).
Huelsken, J. & Birchmeier, W. New aspects of Wnt signaling pathways in higher vertebrates. Curr. Opin. Genet. Dev. 11, 547–553 (2001).
Willert, K. & Nusse, R. Beta-catenin: a key mediator of Wnt signaling. Curr. Opin. Genet. Dev. 8, 95–102 (1998).
Aberle, H., Bauer, A., Stappert, J., Kispert, A. & Kemler, R. β-catenin is a target for the ubiquitin-proteasome pathway. EMBO J. 16, 3797–3804 (1997).
Behrens, J. et al. Functional interaction of an axin homolog, conductin, with β-catenin, APC, and GSK3β. Science 280, 596–599 (1998).
Siegfried, E., Chou, T.B. & Perrimon, N. wingless signaling acts through zeste-white 3, the Drosophila homolog of glycogen synthase kinase-3, to regulate engrailed and establish cell fate. Cell 71, 1167–1179 (1992).
Wodarz, A. & Nusse, R. Mechanisms of Wnt signaling in development. Annu. Rev. Cell Dev. Biol. 14, 59–88 (1998).
Tamai, K. et al. LDL-receptor-related proteins in Wnt signal transduction. Nature 407, 530–535 (2000).
Behrens, J. et al. Functional interaction of β-catenin with the transcription factor LEF-1. Nature 382, 638–642 (1996).
Eastman, Q. & Grosschedl, R. Regulation of LEF-1/TCF transcription factors by Wnt and other signals. Curr. Opin. Cell Biol. 11, 233–240 (1999).
Morin, P.J. & Weeraratna, A.T. Wnt signaling in human cancer. Cancer Treat. Res. 115, 169–187 (2003).
Taketo, M.M. Shutting down Wnt signal-activated cancer. Nat. Genet. 36, 320–322 (2004).
Giles, R.H., van Es, J.H. & Clevers, H. Caught up in a Wnt storm: Wnt signaling in cancer. Biochim. Biophys. Acta 1653, 1–24 (2003).
Moon, R.T., Kohn, A.D., De Ferrari, G.V. & Kaykas, A. WNT and β-catenin signalling: diseases and therapies. Nat. Rev. Genet. 5, 691–701 (2004).
Rattis, F.M., Voermans, C. & Reya, T. Wnt signaling in the stem cell niche. Curr. Opin. Hematol. 11, 88–94 (2004).
Reya, T. et al. A role for Wnt signalling in self-renewal of haematopoietic stem cells. Nature 423, 409–414 (2003).
Austin, T.W., Solar, G.P., Ziegler, F.C., Liem, L. & Matthews, W. A role for the Wnt gene family in hematopoiesis: expansion of multilineage progenitor cells. Blood 89, 3624–3635 (1997).
Van Den Berg, D.J., Sharma, A.K., Bruno, E. & Hoffman, R. Role of members of the Wnt gene family in human hematopoiesis. Blood 92, 3189–3202 (1998).
Willert, K. et al. Wnt proteins are lipid-modified and can act as stem cell growth factors. Nature 423, 448–452 (2003).
Cobas, M. β-catenin is dispensable for hematopoiesis and lymphopoiesis. J. Exp. Med. 199, 221–229 (2004).
Xu, Y., Banerjee, D., Huelsken, J., Birchmeier, W. & Sen, J.M. Deletion of β-catenin impairs T cell development. Nat. Immunol. 4, 1177–1182 (2003).
Verbeek, S. et al. An HMG-box-containing T-cell factor required for thymocyte differentiation. Nature 374, 70–74 (1995).
Okamura, R.M. et al. Redundant regulation of T cell differentiation and TCRα gene expression by the transcription factors LEF-1 and TCF-1. Immunity 8, 11–20 (1998).
Reya, T. et al. Wnt signaling regulates B lymphocyte proliferation through a LEF-1 dependent mechanism. Immunity 13, 15–24 (2000).
Jamieson, C.H. et al. Granulocyte-macrophage progenitors as candidate leukemic stem cells in blast-crisis CML. N. Engl. J. Med. 351, 657–667 (2004).
Harada, N. et al. Intestinal polyposis in mice with a dominant stable mutation of the beta-catenin gene. EMBO J. 18, 5931–5942 (1999).
Kuhn, R., Schwenk, F., Aguet, M. & Rajewsky, K. Inducible gene targeting in mice. Science 269, 1427–1429 (1995).
Adolfsson, J. et al. Upregulation of Flt3 expression within the bone marrow Lin−Sca1+c-kit+ stem cell compartment is accompanied by loss of self-renewal capacity. Immunity 15, 659–669 (2001).
Christensen, J.L. & Weissman, I.L. Flk-2 is a marker in hematopoietic stem cell differentiation: a simple method to isolate long-term stem cells. Proc. Natl. Acad. Sci. USA 98, 14541–14546 (2001).
Almeida, M., Han, L., Bellido, T., Manolagas, S.C. & Kousteni, S. Wnt proteins prevent apoptosis of both uncommitted osteoblast progenitors and differentiated osteoblasts by β-catenin-dependent and -independent signaling cascades involving Src/ERK and phosphatidylinositol 3-kinase/AKT. J. Biol. Chem. 280, 41342–41351 (2005).
Kim, K., Pang, K.M., Evans, M. & Hay, E.D. Overexpression of β-catenin induces apoptosis independent of its transactivation function with LEF-1 or the involvement of major G1 cell cycle regulators. Mol. Biol. Cell 11, 3509–3523 (2000).
Cheng, T. et al. Hematopoietic stem cell quiescence maintained by 21cip1/waf1. Science 287, 1804–1808 (2000).
Fleming, W.H., Alpern, E.J., Uchida, N., Ikuta, K., Spangrude, G.J. & Weissman, I.L. Functional heterogeneity is associated with the cell cycle status of murine hematopoietic stem cells. J. Cell Biol. 122, 897–902 (1993).
Orschell-Traycoff, C.M. et al. Homing and engraftment potential of Sca-1+lin− cells fractionated on the basis of adhesion molecule expression and position in cell cycle. Blood 96, 1380–1387 (2000).
Cheshier, S.H., Morrison, S.J., Liao, X. & Weissman, I.L. In vivo proliferation and cell cycle kinetics of long-term self-renewing hematopoietic stem cells. Proc. Natl. Acad. Sci. USA 96, 3120–3125 (1999).
Lambert, J.F.S., Forget, B.G., Weissman, S.M. & Quesenberry, P.J. et al. Marrow stem cells shift gene expression and engraftment phenotype with cell cycle transit. J. Exp. Med. 197, 1563–1572 (2003).
Na Nakorn, T., Traver, D., Weissman, I.L. & Akashi, K. Myeloerythroid-restricted progenitors are sufficient to confer radioprotection and provide the majority of day 8 CFU-S. J. Clin. Invest. 109, 1579–1585 (2002).
Magli, M.C., Iscove, N.N. & Odartchenko, N. Transient nature of early haematopoietic spleen colonies. Nature 295, 527–529 (1982).
Socolovsky, M. et al. Ineffective erythropoiesis in Stat5a−/−5b−/− mice due to decreased survival of early erythroblasts. Blood 98, 3261–3273 (2001).
Akashi, K., Traver, D., Miyamoto, T. & Weissman, I.L. A clonogenic common myeloid progenitor that gives rise to all myeloid lineages. Nature 404, 193–197 (2000).
Hardy, R.R., Carmack, C.E., Shinton, S.A., Kemp, J.D. & Hayakawa, K. Resolution and characterization of pro-B and pre-pro-B cell stages in normal mouse bone marrow. J. Exp. Med. 173, 1213–1225 (1991).
Rolink, A., Streb, M. & Melchers, F. The κ/λ ratio in surface immunoglobulin molecules on B lymphocytes differentiating from DHJH-rearranged murine pre-B cell clones in vitro. Eur. J. Immunol. 21, 2895–2898 (1991).
Weissman, I., Spangrude, G., Heimfeld, S., Smith, L. & Uchida, N. Stem cells. Nature 353, 26 (1991).
Adolfsson, J. et al. Identification of Flt3+ lympho-myeloid stem cells lacking erythro-megakaryocytic potential a revised road map for adult blood lineage commitment. Cell 121, 295–306 (2005).
Zeng, H., Yucel, R., Kosan, C., Klein-Hitpass, L. & Moroy, T. Transcription factor Gfi1 regulates self-renewal and engraftment of hematopoietic stem cells. EMBO J. 23, 4116–4125 (2004).
Peled, A. et al. Dependence of human stem cell engraftment and repopulation of NOD/SCID mice on CXCR4. Science 283, 845–848 (1999).
Vermeulen, M. et al. Role of adhesion molecules in the homing and mobilization of murine hematopoietic stem and progenitor cells. Blood 92, 894–900 (1998).
Papayannopoulou, T., Craddock, C., Nakamoto, B., Priestley, G.V. & Wolf, N.S. The VLA4/VCAM-1 adhesion pathway defines contrasting mechanisms of lodgement of transplanted murine hemopoietic progenitors between bone marrow and spleen. Proc. Natl. Acad. Sci. USA 92, 9647–9651 (1995).
Feng, Z., Srivastava, A.S., Mishra, R. & Carrier, E. A regulatory role of Wnt signaling pathway in the hematopoietic differentiation of murine embryonic stem cells. Biochem. Biophys. Res. Commun. 324, 1333–1339 (2004).
Yoshida, T., Yao-Ming Ng, S., Zuniga-Pflucker, J.C. & Georgopoulos, K. Early hematopoietic lineage restrictions directed by Ikaros. Nat. Immunol. 7, 382–391 (2006).
Qiang, Y.W. & Rudikoff, S. Wnt signaling in B and T lymphocytes. Front. Biosci. 9, 1000–1010 (2004).
Domen, J., Cheshier, S.H. & Weissman, I.L. The role of apoptosis in the regulation of hematopoietic stem cells: Overexpression of Bcl-2 increases both their number and repopulation potential. J. Exp. Med. 191, 253–264 (2000).
Adams, J.M. et al. Control of apoptosis in hematopoietic cells by the Bcl-2 family of proteins. Cold Spring Harb. Symp. Quant. Biol. 64, 351–358 (1999).
Trowbridge, J.J., Xenocostas, A., Moon, R.T. & Bhatia, M. Glycogen synthase kinase-3 is an in vivo regulator of hematopoietic stem cell repopulation. Nat. Med. 12, 89–98 (2006).
Murdoch, B. et al. Wnt-5A augments repopulating capacity and primitive hematopoietic development of human blood stem cells in vivo. Proc. Natl. Acad. Sci. USA 100, 3422–3427 (2003).
Till, J.E. & Mc, C.E. A direct measurement of the radiation sensitivity of normal mouse bone marrow cells. Radiat. Res. 14, 213–222 (1961).
Passegue, E. Hematopoietic stem cells, leukemic stem cells and chronic myelogenous leukemia. Cell Cycle 4, 266–268 (2005).
Acknowledgements
We thank J. Loehler for histological consultations; J. Lausen, S. Eiglmeier and T. Dayaram for help with real-time RT-PCR; A. Wiener, C. Peter and U. Jahn (Department of Radiotherapy, Robert-Roessle Klinik, Berlin, Germany) for help with animal irradiation experiments; K. Raba and T. Kaiser (Flow cytometry and Cellsorting core facility of Deutsches Rheumaforschungszentrum, Berlin, Germany) for help with sorting of cells. Supported by the Lymphoma Research Foundation (F.R.), the Helmholtz Association (F.R.), the National Center of Competence in Research (J.H.) and the Fondation Leenaards (Lausanne, Switzerland; J.H.).
Author information
Authors and Affiliations
Contributions
All authors contributed to discussions, experimental design, data analysis and the preparation of the manuscript; M.S. designed and executed experiments and prepared the manuscript; J.H. examined the excision of exon 3 in MxCre+Ctnnb1(Ex3)fl/fl mice and the expression of genes downstream of β-catenin and helped to analyze data and to write the manuscript; J.H. and W.B. initiated the collaboration with M.M.T. to analyze the hematopoietic phenotype of Ctnnb1(Ex3)fl/fl mice; F.R. and D.G.T. helped with analyses of HSC gene expression; A.L. supervised the work; and A.L. and M.S. composed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Experimental schemes. (PDF 80 kb)
Supplementary Fig. 2
Apoptosis and adhesion marker expression in LSK cells. (PDF 81 kb)
Supplementary Table 1
Oligonucleotides used in real-time RT-PCR and genotyping PCR. (PDF 74 kb)
Rights and permissions
About this article
Cite this article
Scheller, M., Huelsken, J., Rosenbauer, F. et al. Hematopoietic stem cell and multilineage defects generated by constitutive β-catenin activation. Nat Immunol 7, 1037–1047 (2006). https://doi.org/10.1038/ni1387
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni1387
This article is cited by
-
DKK-1 and Its Influences on Bone Destruction: A Comparative Study in Collagen-Induced Arthritis Mice and Rheumatoid Arthritis Patients
Inflammation (2024)
-
WNT Signaling in Stem Cells: A Look into the Non-Canonical Pathway
Stem Cell Reviews and Reports (2024)
-
DNMT3A-coordinated splicing governs the stem state switch towards differentiation in embryonic and haematopoietic stem cells
Nature Cell Biology (2023)
-
Adhesion G protein-coupled receptor gluing action guides tissue development and disease
Journal of Molecular Medicine (2022)
-
Bmi1 Regulates Wnt Signaling in Hematopoietic Stem and Progenitor Cells
Stem Cell Reviews and Reports (2021)