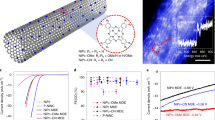
Abstract
A new concept for enzyme-catalyzed redox transformations features pairs of electron donor and acceptor enzymes attached to conducting particles. Electrons furnished by oxidation at one enzyme are used at the other. Graphite microparticles modified with hydrogenase and nitrate reductase or fumarate reductase catalyze reductions of nitrate or fumarate (1) by H2.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Vincent, K.A., Parkin, A. & Armstrong, F.A. Chem. Rev. 107, 4366–4413 (2007).
Léger, C. et al. Biochemistry 42, 8653–8662 (2003).
Vincent, K.A. et al. Chem. Commun. (Camb) 5033–5035 (2006).
Elliott, S.J. et al. Biochemistry 43, 799–807 (2004).
Hudson, J.M. et al. J. Am. Chem. Soc. 127, 6977–6989 (2005).
Blanford, C.F., Heath, R.S. & Armstrong, F.A. Chem. Commun. (Camb) 1710–1712 (2007).
Cammack, R., Frey, M. & Robson, R. Hydrogen as a Fuel: Learning from Nature 1–261 (Taylor and Francis, London and New York, 2001).
Bertero, M.G. et al. Nat. Struct. Biol. 10, 681–687 (2003).
Goldstein, J. et al. Scanning Electron Microscopy and X-ray Microanalysis: a Text for Biologists, Materials Scientists, and Geologists 79–147 (Plenum, New York and London, 1992).
Blanford, C.F. & Armstrong, F.A. J. Solid State Electrochem. 10, 826–832 (2006).
Rajarathnam, K., Hochmans, J., Schindler, M. & Ferguson-Miller, S. Biochemistry 28, 3168–3176 (1989).
Trieber, C.A., Rothery, R.A. & Weiner, J.H. J. Biol. Chem. 269, 7103–7109 (1994).
Lide, D.R. & Frederikse, H.P.R. CRC Handbook of Chemistry and Physics 12–40–12–42 (CRC Press, Boca Raton, Florida, USA, 1996).
Yajima, S. & Hirai, T. J. Mater. Sci. 4, 692–698 (1969).
Acknowledgements
This research was supported by grants from the UK Biotechnology and Biological Sciences Research Council (BB/D52222X/1), the UK Engineering and Physical Sciences Research Council (Supergen V) and the Leverhulme Trust (F/08699/C). J.H.W. is a Canada Research Chair and thanks the Alberta Heritage Foundation for Medical Research for a travel award. The authors thank N. Boroumand (University of Alberta), G. Cecchini (NCIRE Veterans Health Research Institute, San Francisco) and B. Friedrich (Humboldt-Universität zu Berlin), respectively, for samples of nitrate reductase, fumarate reductase and Ralstonia metallidurans hydrogenase (CH34).
Author information
Authors and Affiliations
Contributions
K.A.V. jointly designed the coupling experiments, jointly performed the nitrate reductase/hydrogenase experiments and jointly wrote the paper; X.L. jointly performed the nitrate reductase/hydrogenase experiments; C.F.B. jointly designed and conducted the fumarate reductase/hydrogenase experiments, carried out the SEM and N2 porosimetry and jointly wrote the paper; N.A.B. conducted the fumarate reductase/hydrogenase experiments and purified A. vinosum hydrogenase; J.H.W. jointly designed the coupling experiments and jointly wrote the paper; F.A.A. jointly designed the experiments and jointly wrote the paper.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–3 and Supplementary Methods (PDF 323 kb)
Rights and permissions
About this article
Cite this article
Vincent, K., Li, X., Blanford, C. et al. Enzymatic catalysis on conducting graphite particles. Nat Chem Biol 3, 761–762 (2007). https://doi.org/10.1038/nchembio.2007.47
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.2007.47
This article is cited by
-
Non-contact biomimetic mechanism for selective hydrogenation of nitroaromatics on heterogeneous metal nanocatalysts
Science China Chemistry (2022)
-
Relating diffusion along the substrate tunnel and oxygen sensitivity in hydrogenase
Nature Chemical Biology (2010)