Abstract
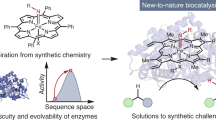
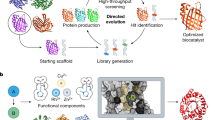
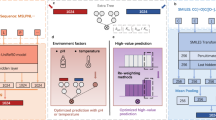
The rational design of artificial enzymes, either by applying physico–chemical intuition of protein structure and function or with the aid of computational methods, is a promising area of research with the potential to tremendously impact medicine, industrial chemistry and energy production. Designed proteins also provide a powerful platform for dissecting enzyme mechanisms of natural systems. Artificial enzymes have come a long way from simple α-helical peptide catalysts to proteins that facilitate multistep chemical reactions designed by state-of-the-art computational methods. Looking forward, we examine strategies employed by natural enzymes that could be used to improve the speed and selectivity of artificial catalysts.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Davie, E. A. C., Mennen, S. M., Xu, Y. & Miller, S. J. Asymmetric catalysis mediated by synthetic peptides. Chem. Rev. 107, 5759–5812 (2007).
List, B. Proline-catalyzed asymmetric reactions. Tetrahedron 58, 5573–5590 (2002).
Koder, R. L. & Dutton, P. L. Intelligent design: the de novo engineering of proteins with specified functions. Dalton Trans. 25, 3045–3051 (2006).
Lim, V. in Protein Folding, 28th Conference of the German Biochemical Society (ed. Jaenicke, R.) 149–166 (Elsevier, 1979).
Crick, F. H. C. The packing of alpha-helices: simple coiled coils. Acta Crystallogr. 6, 689–697 (1953).
Kamtekar, S., Schiffer, J. M., Xiong, H., Babik, J. M. & Hecht, M. H. Protein design by binary patterning of polar and nonpolar amino acids. Science 262, 1680–1685 (1993).
Lau, S. Y. M., Taneja, A. K. & Hodges, R. S. Synthesis of a model protein of defined secondary and quaternary structure. J. Biol. Chem. 259, 13253–13261 (1984).
DeGrado, W. F. & Lear, J. D. Induction of peptide conformation at apolar/water interfaces. 1. a study with model peptides of defined hydrophobic periodicity. J. Am. Chem. Soc. 107, 7684–7689 (1985).
Bryson, J. W. et al. Protein design: a hierarchic approach. Science 270, 935–941 (1995).
Handel, T. M., Williams, S. A. & DeGrado, W. F. Metal ion-dependent modulation of the dynamics of a designed protein. Science 261, 879–885 (1993).
Lovejoy, B. et al. Crystal structure of a synthetic triple-stranded alpha-helical bundle. Science 259, 1288–1293 (1993).
Harbury, P. B., Plecs, J. J., Tidor, B., Alber, T. & Kim, P. S. High-resolution protein design with backbone freedom. Science 282, 1462–1467 (1998).
Sasaki, T. & Kaiser, E. T. Helichrome: synthesis and enzymatic activity of a designed hemeprotein. J. Am. Chem. Soc. 111, 380–381 (1989).
Barbier, B. & Brack, A. Basic polypeptides accelerate the hydrolysis of ribonucleic acids. J. Am. Chem. Soc. 110, 6880–6882 (1988).
Barbier, B. & Brack, A. Conformation-controlled hydrolysis of polyribonucleotides by sequential basic polypeptides. J. Am. Chem. Soc. 114, 3511–3515 (1992).
Brack, A. & Spach, G. Multiconformational synthetic polypeptides. J. Am. Chem. Soc. 103, 6319–6323 (1981).
Johnsson, K., Allemann, R. K., Widmer, H. & Benner, S. A. Synthesis, structure and activity of artificial, rationally designed catalytic polypeptides. Nature 365, 530–532 (1993).
Taylor, S. E., Rutherford, T. J. & Allemann, R. K. Design, synthesis and characterisation of a peptide with oxaloacetate decarboxylase activity. Bioorg. Med. Chem. Lett. 11, 2631–2635 (2001).
Taylor, S. E., Rutherford, T. J. & Allemann, R. K. Design of a folded, conformationally stable oxaloacetate decarboxylase. J. Chem. Soc. Perkins Trans. 2, 751–755 (2002).
Nicoll, A. J. & Allemann, R. K. Nucleophilic and general acid catalysis at physiological pH by a designed miniature esterase. Org. Biomol. Chem. 2, 2175–2180 (2004).
Lee, D. H., Severin, K., Yokobayashi, Y. & Ghadiri, M. R. Emergence of symbiosis in peptide self-replication through a hypercyclic network. Nature 390, 591–594 (1997).
Saghatelian, A., Yokobayashi, Y., Soltani, K. & Ghadiri, M. R. A chiroselective peptide replicator. Nature 409, 797–801 (2001).
Butterfield, S. M., Cooper, W. J. & Waters, M. L. Minimalist protein design: a beta-hairpin peptide that binds ssDNA. J. Am. Chem. Soc. 127, 24–25 (2005).
Butterfield, S. M., Goodman, C. M., Rotello, V. M. & Waters, M. L. A peptide flavoprotein mimic: flavin recognition and redox potential modulation in water by a designed beta hairpin. Angew. Chem. Int. Ed. 43, 724–727 (2004).
Hughes, R. M. & Waters, M. L. Model systems for beta-hairpins and beta-sheets. Curr. Opin. Struct. Biol. 16, 514–524 (2006).
Olofsson, S. & Baltzer, L. Structure and dynamics of a designed helix-loop-helix dimer in dilute aqueous trifluoroethanol solution. A strategy for NMR spectroscopic structure determination of molten globules in the rational design of native-like proteins. Fold. Des. 1, 347–356 (1996).
Rossi, P., Tecilla, P., Baltzer, L. & Scrimin, P. De novo metallonucleases based on helix-loop-helix motifs. Chem. Eur. J. 10, 4163–4170 (2004).
Razkin, J., Lindgren, J., Nilsson, H. & Baltzer, L. Enhanced complexity and catalytic efficiency in the hydrolysis of phosphate diesters by rationally designed helix-loop-helix motifs. ChemBioChem 9, 1975–1984 (2008).
Razkin, J., Nilsson, H. & Baltzer, L. Catalysis of the cleavage of uridine 3′-2,2, 2-trichloroethylphosphate by a designed helix-loop-helix motif peptide. J. Am. Chem. Soc. 129, 14752–14758 (2007).
Tommos, C., Skalicky, J. J., Pilloud, D. L., Wand, A. J. & Dutton, P. L. De novo proteins as models of radical enzymes. Biochemistry 38, 9495–9507 (1999).
Dai, Q. H. et al. Structure of a de novo designed protein model of radical enzymes. J. Am. Chem. Soc. 124, 10952–19053 (2002).
Gibney, B. R., Rabanal, F., Skalicky, J. J., Wand, A. J. & Dutton, P. L. Design of a unique protein scaffold for maquettes. J. Am. Chem. Soc. 119, 2323–2324 (1997).
Gibney, B. R., Rabanal, F., Skalicky, J. J., Wand, A. J. & Dutton, P. L. Iterative protein redesign. J. Am. Chem. Soc. 121, 4952–4960 (1999).
Simons, K. T., Kooperberg, C., Huang, E. & Baker, D. Assembly of protein tertiary structures from fragments with similar local sequences using simulated annealing and bayesian scoring functions. J. Mol. Biol. 268, 209–225 (1997).
Koder, R. L. et al. Native-like structure in designed four helix bundles driven by buried polar interactions. J. Am. Chem. Soc. 128, 14450–14451 (2006).
Koder, R. L. et al. Design and engineering of an O2 transport protein. Nature 458, 305–309 (2009).
Kundu, S., Trent, J. T. & Hargrove, M. S. Plants, humans and hemoglobins. Trends Plant Sci. 8, 387–393 (2003).
Dahiyat, B. I. & Mayo, S. L. De novo protein design: fully automated sequence selection. Science 278, 82–87 (1997).
Kuhlman, B. et al. Design of a novel globular protein fold with atomic-level accuracy. Science 302, 1364–1368 (2003).
Wallar, B. J. & Lipscomb, J. D. Dioxygen activation by enzymes containing binuclear non-heme iron clusters. Chem. Rev. 96, 2625–2658 (1996).
Lombardi, A. et al. Retrostructural analysis of metalloproteins: application to the design of a minimal model for diiron proteins. Proc. Natl Acad. Sci. USA 97, 6298–305 (2000).
Summa, C. M., Lombardi, A., Lewis, M. & DeGrado, W. F. Tertiary templates for the design of diiron proteins. Curr. Opin. Struct. Biol. 9, 500–508 (1999).
Lazar, G. A., Desjarlais, J. R. & Handel, T. M. De novo design of the hydrophobic core of ubiquitin. Protein Sci. 6, 1167–1178 (1997).
Di Costanzo, L. et al. Toward the de novo design of a catalytically active helix bundle: a substrate-accessible carboxylate-bridged dinuclear metal center. J. Am. Chem. Soc. 123, 12749–57 (2001).
Kaplan, J. & DeGrado, W. F. De novo design of catalytic proteins. Proc. Natl Acad. Sci. USA 101, 11566–11570 (2004).
Maglio, O., Nastri, F., Pavone, V., Lombardi, A. & DeGrado, W. F. Preorganization of molecular binding sites in designed diiron proteins. Proc. Natl Acad. Sci. USA 100, 3772–3777 (2003).
Geremia, S. et al. Response of a designed metalloprotein to changes in metal ion coordination, exogenous ligands, and active site volume determined by X-ray crystallography. J. Am. Chem. Soc. 127, 17266–76 (2005).
DeGrado, W. F. et al. Sliding helix and change of coordination geometry in a model di-MnII protein. Angew. Chem. Int. Ed. 42, 417–420 (2003).
Summa, C. M., Rosenblatt, M. M., Hong, J. K., Lear, J. D. & DeGrado, W. F. Computational de novo design, and characterization of an A2B2 diiron protein. J. Mol. Biol. 321, 923–938 (2002).
Marsh, E. N. & DeGrado, W. F. Noncovalent self-assembly of a heterotetrameric diiron protein. Proc. Natl Acad. Sci. USA 99, 5150–5154 (2002).
Papoian, G. A., DeGrado, W. F. & Klein, M. L. Probing the configurational space of a metalloprotein core: an ab initio molecular dynamics study of Duo Ferro 1 binuclear Zn cofactor. J. Am. Chem. Soc. 125, 560–569 (2003).
Cochran, F. V. et al. Computational de novo design and characterization of a four-helix bundle protein that selectively binds a nonbiological cofactor. J. Am. Chem. Soc. 127, 1346–1347 (2005).
Nanda, V. et al. De novo design of a redox-active minimal rubredoxin mimic. J. Am. Chem. Soc. 127, 5804–5805 (2005).
Clarke, N. D. & Yuan, S. M. Metal search: a computer program that helps design tetrahedral metal-binding sites. Proteins 23, 256–263 (1995).
Klemba, M., Gardner, K. H., Marino, S., Clarke, N. D. & Regan, L. Novel metal-binding proteins by design. Nature Struct. Biol. 2, 368–373 (1995).
Regan, L. & Clarke, N. D. A tetrahedral zinc(II)-binding site introduced into a designed protein. Biochemistry 29, 10878–10883 (1990).
Hellinga, H. W. & Richards, F. M. Construction of new ligand binding sites in proteins of known structure. I. Computer-aided modeling of sites with pre-defined geometry. J. Mol. Biol. 222, 763–785 (1991).
Marvin, J. S. & Hellinga, H. W. Conversion of a maltose receptor into a zinc biosensor by computational design. Proc. Natl Acad. Sci. USA 98, 4955–4960 (2001).
Telmer, P. G. & Shilton, B. H. Structural studies of an engineered zinc biosensor reveal an unanticipated mode of zinc binding. J. Mol. Biol. 354, 829–840 (2005).
Benson, D. E., Wisz, M. S. & Hellinga, H. W. Rational design of nascent metalloenzymes. Proc. Natl Acad. Sci. USA 97, 6292–6297 (2000).
Pinto, A. L., Hellinga, H. W. & Caradonna, J. P. Construction of a catalytically active iron superoxide dismutase by rational protein design. Proc. Natl Acad. Sci. USA 94, 5562–5567 (1997).
Bolon, D. N. & Mayo, S. L. Enzyme-like proteins by computational design. Proc. Natl Acad. Sci. USA 98, 14274–14279 (2001).
Desmet, J., Demaeyer, M., Hazes, B. & Lasters, I. The dead-end elimination theorem and its use in protein side-chain positioning. Nature 356, 539–542 (1992).
Looger, L. L. & Hellinga, H. W. Generalized dead-end elimination algorithms make large-scale protein side-chain structure prediction tractable: implications for protein design and structural genomics. J. Mol. Biol. 307, 429–445 (2001).
Host, G. E., Razkin, J., Baltzer, L. & Jonsson, B. H. Combined enzyme and substrate design: grafting of a cooperative two-histidine catalytic motif into a protein targeted at the scissile bond in a designed ester substrate. ChemBioChem 8, 1570–1576 (2007).
Kuhlman, B. & Baker, D. Native protein sequences are close to optimal for their structures. Proc. Natl Acad. Sci. USA 97, 10383–10388 (2000).
Jiang, L. et al. De novo computational design of retro-aldol enzymes. Science 319, 1387–1391 (2008).
Rothlisberger, D. et al. Kemp elimination catalysts by computational enzyme design. Nature 453, 190–195 (2008).
Tanaka, F., Fuller, R., Shim, H., Lerner, R. A. & Barbas, C. F. Evolution of aldolase antibodies in vitro: correlation of catalytic activity and reaction-based selection. J. Mol. Biol. 335, 1007–1018 (2004).
Tantillo, D. J., Chen, J. & Houk, K. N. Theozymes and compuzymes: theoretical models for biological catalysis. Curr. Opin. Chem. Bio. 2, 743–750 (1998).
Zanghellini, A. et al. New algorithms and an in silico benchmark for computational enzyme design. Protein Sci. 15, 2785–2794 (2006).
Ladman, Y., Schwartz, J. T. & Wolfson, H. J. Affine invariant model-based object recognition. IEEE Trans. Robot. Automat. 6, 578–589 (1990).
Wolfson, H. J. & Rigoutsos, I. Geometric hashing: an overview. IEEE Comp. Sci. Eng. 4, 10–21 (1997).
Li, H., Helling, R., Tang, C. & Wingreen, N. Emergence of preferred structures in a simple model of protein folding. Science 273, 666–669 (1996).
Li, H., Tang, C. & Wingreen, N. S. Are protein folds atypical? Proc. Natl Acad. Sci. USA 95, 4987–4990 (1998).
Hilvert, D. Critical analysis of antibody catalysis. Annu. Rev. Biochem. 69, 751–792 (2000).
Thorn, S. N., Daniels, R. G., Auditor, M. T. & Hilvert, D. Large rate accelerations in antibody catalysis by strategic use of haptenic charge. Nature 373, 228–230 (1995).
Hollfelder, F., Kirby, A. J. & Tawfik, D. S. Off-the-shelf proteins that rival tailor-made antibodies as catalysts. Nature 383, 60–62 (1996).
Warshel, A. Electrostatic origin of the catalytic power of enzymes and the role of preorganized active sites. J. Biol. Chem. 273, 27035–27038 (1998).
Voigt, C. A., Mayo, S. L., Arnold, F. H. & Wang, Z. G. Computational method to reduce the search space for directed protein evolution. Proc. Natl Acad. Sci. USA 98, 3778–3783 (2001).
Voigt, C. A., Martinez, C., Wang, Z. G., Mayo, S. L. & Arnold, F. H. Protein building blocks preserved by recombination. Nature Struct. Biol. 9, 553–558 (2002).
Treynor, T. P., Vizcarra, C. L., Nedelcu, D. & Mayo, S. L. Computationally designed libraries of fluorescent proteins evaluated by preservation and diversity of function. Proc. Natl Acad. Sci. USA 104, 48–53 (2007).
Lippow, S. M., Wittrup, K. D. & Tidor, B. Computational design of antibody-affinity improvement beyond in vivo maturation. Nature Biotechnol. 25, 1171–1176 (2007).
Radzicka, A. & Wolfenden, R. A proficient enzyme. Science 267, 90–93 (1995).
Lienhard, G. E. Enzymatic catalysis and transition-state theory. Science 180, 149–154 (1973).
Kraut, D. A., Carroll, K. S. & Herschlag, D. Challenges in enzyme mechanism and energetics. Annu. Rev. Biochem. 72, 517–571 (2003).
Jencks, W. P. Catalysis in Chemistry and Enzymology (McGraw-Hill, 1969).
Noy, D., Moser, C. C. & Dutton, P. L. Design and engineering of photosynthetic light-harvesting and electron transfer using length, time, and energy scales. Biochim. Biophys. Acta 1757, 90–105 (2006).
Nagel, Z. D. & Klinman, J. P. Tunneling and dynamics in enzymatic hydride transfer. Chem. Rev. 106, 3095–3118 (2006).
Bruice, T. C. Computational approaches: Reaction trajectories, structures, and atomic motions. Enzyme reactions and proficiency. Chem. Rev. 106, 3119–3139 (2006).
Boehr, D. D., McElheny, D., Dyson, H. J. & Wright, P. E. The dynamic energy landscape of dihydrofolate reductase catalysis. Science 313, 1638–1642 (2006).
Gerstein, M., Lesk, A. M. & Chothia, C. Structural mechanisms for domain movements in proteins. Biochemistry 33, 6739–6749 (1994).
Henzler-Wildman, K. A. et al. A hierarchy of timescales in protein dynamics is linked to enzyme catalysis. Nature 450, 913–916 (2007).
Henzler-Wildman, K. A. et al. Intrinsic motions along an enzymatic reaction trajectory. Nature 450, 838–844 (2007).
Xiang, J. Y., Jung, J. Y. & Sampson, N. S. Entropy effects on protein hinges: The reaction catalyzed by triosephosphate isomerase. Biochemistry 43, 11436–11445 (2004).
Munro, A. W. et al. P450BM3: the very model of a modern flavocytochrome. Trends Biochem. Sci. 27, 250–257 (2002).
Wyman, J. & Gill, S. J. Binding and Linkage (University Science Books, 1990).
Anderson, J. L. R., Koder, R. L., Moser, C. C. & Dutton, P. L. Controlling complexity and water penetration in functional de novo protein design. Biochem. Soc. Trans. 36, 1106–1111 (2008).
Joyce, G. F. The antiquity of RNA-based evolution. Nature 418, 214–221 (2002).
Doudna, J. A. & Lorsch, J. R. Ribozyme catalysis: not different, just worse. Nature Struct. Mol. Biol. 12, 395–402 (2005).
Lee, S. & Jung, S. Cyclosophoraose as a catalytic carbohydrate for methanolysis. Carbohydr. Res. 339, 461–468 (2004).
Hill, D. J., Mio, M. J., Prince, R. B., Hughes, T. S. & Moore, J. S. A field guide to foldamers. Chem. Rev. 101, 3893–4011 (2001).
Goodman, C. M., Choi, S., Shandler, S. & DeGrado, W. F. Foldamers as versatile frameworks for the design and evolution of function. Nature Chem. Biol. 3, 252–262 (2007).
Nanda, V. & DeGrado, W. F. Computational design of heterochiral peptides against a helical target. J. Am. Chem. Soc. 128, 809–816 (2006).
Nanda, V. & Degrado, W. F. Simulated evolution of emergent chiral structures in polyalanine. J. Am. Chem. Soc. 126, 14459–14467 (2004).
Baldauf, C., Gunther, R. & Hoffmann, H.-J. Helix Formation in α, - and β, γ-hybrid peptides: Theoretical insights into mimicry of α- and β-peptides. J. Org. Chem. 71, 1200–1208 (2006).
Sandvoss, L. M. & Carlson, H. A. Conformational behavior of β-proline oligomers. J. Am. Chem. Soc. 125, 15855–15862 (2003).
Lee, O.-S. & Saven, J. G. Simulation studies of a helical m-phenylene ethylene foldamer. J. Phys. Chem. B 108, 11988–11994 (2004).
Smaldone, R. A. & Moore, J. S. Reactive sieving with foldamers: inspiration from nature and directions for the future. Chem. Eur. J. 14, 2650–2657 (2008).
Blake, C. C. et al. Structure of hen egg-white lysozyme. A three-dimensional Fourier synthesis at 2 Å resolution. Nature 206, 757–761 (1965).
Nanda, V. Do-it-yourself enzymes. Nature Chem. Biol. 4, 273–275 (2008).
Acknowledgements
VN acknowledges support from the NIH Director's New Innovator Award Program, 1-DP2-OD006478-01 and the NSF BMAT program DMR-0907273. RLK acknowledges supported by the following grants: MCB-0920448 from the NSF, MCB-5G12 RR03060 toward support for the NMR facilities at the City College of New York, P41 GM-66354 to the New York Structural Biology Center and infrastructure support from NIH 5G12 RR03060 from the National Center for Research Resources.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Nanda, V., Koder, R. Designing artificial enzymes by intuition and computation. Nature Chem 2, 15–24 (2010). https://doi.org/10.1038/nchem.473
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.473
This article is cited by
-
Introducing molecular imprinting onto nanozymes: toward selective catalytic analysis
Analytical and Bioanalytical Chemistry (2024)
-
Light-responsive organic artificial enzymes: Material designs and bio-applications
Nano Research (2023)
-
Single-atom nanozymes towards central nervous system diseases
Nano Research (2023)
-
Sb-doped FeOCl nanozyme-based biosensor for highly sensitive colorimetric detection of glutathione
Analytical and Bioanalytical Chemistry (2023)
-
A bound iron porphyrin is redox active in hybrid bacterial reaction centers modified to possess a four-helix bundle domain
Photochemical & Photobiological Sciences (2022)