Abstract
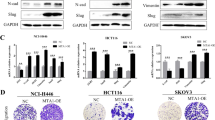
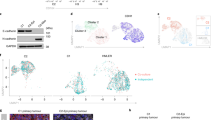
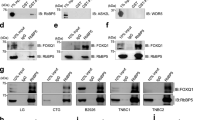
The epithelial–mesenchymal transition (EMT), one of the main mechanisms underlying development of cancer metastasis, induces stem-like properties in epithelial cells. Bmi1 is a polycomb-group protein that maintains self-renewal, and is frequently overexpressed in human cancers. Here, we show the direct regulation of BMI1 by the EMT regulator, Twist1. Furthermore, Twist1 and Bmi1 were mutually essential to promote EMT and tumour-initiating capability. Twist1 and Bmi1 act cooperatively to repress expression of both E-cadherin and p16INK4a. In patients with head and neck cancers, increased levels of both Twist1 and Bmi1 correlated with downregulation of E-cadherin and p16INK4a, and was associated with the worst prognosis. These results suggest that Twist1-induced EMT and tumour-initiating capability in cancer cells occurs through chromatin remodelling, which leads to unfavourable clinical outcomes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Accession codes
References
Thiery, J. P., Acloque, H., Huang, R. Y. & Nieto, M. A. Epithelial–mesenchymal transitions in development and disease. Cell 139, 871–890 (2009).
Mani, S. A. et al. The epithelial–mesenchymal transition generates cells with properties of stem cells. Cell 133, 704–715 (2008).
Morel, A. P. et al. Generation of breast cancer stem cells through epithelial–mesenchymal transition. PLoS ONE 3, e2888 (2008).
Valk-Lingbeek, M. E., Bruggeman, S. W. & van Lohuizen, M. Stem cells and cancer: the polycomb connection. Cell 118, 409–418 (2004).
Widschwendter, M. et al. Epigenetic stem cell signature in cancer. Nat. Genet. 39, 157–158 (2007).
Molofsky, A. V. et al. Bmi-1 dependence distinguishes neural stem cell self-renewal from progenitor proliferation. Nature 425, 962–967 (2003).
Iwama, A. et al. Enhanced self-renewal of haematopoietic stem cells mediated by the polycomb gene product Bmi-1. Immunity 21, 843–851 (2004).
Sangiorgi, E. & Capecchi, M. R. Bmi1 is expressed in vivo in intestinal stem cells. Nat. Genet. 40, 915–920 (2008).
Jacobs, J. J., Kieboom, K., Marino, S., Depinho, R. & van Lohuizen, M. The oncogene and polycomb-group gene Bmi1 regulates cell proliferation and senescence through the ink4a locus. Nature 397, 164–168 (1999).
Bruggeman, S. W. et al. Ink4a and Arf differentially affect cell proliferation and neural stem cell self-renewal in Bmi1-deficient mice. Genes & Dev. 19, 1438–1443 (2005).
Czermin, B. et al. Drosophila Enhancer of Zeste/ESC complexes have a histone H3 methyltransferase activity that marks chromosomal polycomb sites. Cell 111, 185–196 (2002).
Müller, J. et al. Histone methyltransferase activity of a Drosophila polycomb group repressor complex. Cell 111, 197–208 (2002).
Min, J., Zhang, Y. & Xu, R. M. Structural basis for specific binding of polycomb chromodomain to histone H3 methylated at Lys 27. Genes & Dev. 17, 1823–1828 (2003).
Bracken, A. P. et al. The polycomb group proteins bind throughout the INK4A–ARF locus and are disassociated in senescent cells. Genes & Dev. 21, 525–530 (2007).
Jacobs, J. J. et al. Bmi-1 collaborates with c-Myc in tumorigenesis by inhibiting c-Myc-induced apoptosis via INK4a/ARF. Genes Dev. 13, 2678–2690 (1999).
Leung, C. et al. Bmi1 is essential for cerebellar development and is overexpressed in human medulloblastomas. Nature 428, 337–341 (2004).
Prince, M. E. et al. Identification of a subpopulation of cells with cancer stem cell properties in head and neck squamous cell carcinoma. Proc. Natl Acad. Sci. USA. 104, 973–978 (2007).
Chiba, T. et al. The polycomb gene product BMI1 contributes to the maintenance of tumor-initiating side population cells in hepatocellular carcinoma. Cancer Res. 68, 7742–7749 (2008).
Seiwert, T. Y. & Cohen, E. E. State-of-the-art management of locally advanced head and neck cancer. Br. J. Cancer 92, 1341–1348 (2005).
Janssen, H. L., Haustermans, K. M., Balm, A. J. & Begg, A. C. Hypoxia in head and neck cancer: how much, how important? Head Neck 27, 622–638 (2005).
Hoogsteen, I. J., Marres, H. A., Bussink, J., van der Kogel, A. J. & Kaanders, J. H. Tumor microenvironment in head and neck squamous cell carcinomas: predictive value and clinical relevance of hypoxic markers. A review. Head Neck 29, 591–604 (2007).
Furlong, E. E., Andersen, E. C., Null, B., White, K. P. & Scott, M. P. Patterns of gene expression during Drosophila mesoderm development. Science 293, 1629–1633 (2001).
Yang, J. et al. Twist, a master regulator of morphogenesis, plays an essential role in tumor metastasis. Cell 117, 927–939 (2004).
Yang, M. H. et al. Direct regulation of TWIST by HIF-1α promotes metastasis. Nat. Cell Biol. 10, 295–305 (2008).
Keith, B. & Simon, M. C. Hypoxia-inducible factors, stem cells and cancer. Cell 129, 465–472 (2007).
Chen, Y. C. et al. Aldehyde dehydrogenase 1 is a putative marker for cancer stem cells in head and neck squamous cancer. Biochem. Biophys. Res. Commun. 385, 307–313 (2009).
Huang, L. E., Gu, J., Schau, M. & Bunn, H. F. Regulation of hypoxia-inducible factor 1alpha is mediated by an O2-dependent degradation domain via the ubiquitin–proteasome pathway. Proc. Natl Acad. Sci. USA. 95, 7987–7992 (1998).
Vermeulen, L. et al. Single-cell cloning of colon cancer stem cells reveals a multi-lineage differentiation capacity. Proc. Natl Acad. Sci. USA. 105, 13427–13432 (2008).
Cao, Q. et al. Repression of E-cadherin by the polycomb group protein EZH2 in cancer. Oncogene 27, 7274–7284 (2008).
Tamura, G. et al. E-Cadherin gene promoter hypermethylation in primary human gastric carcinomas. J. Natl Cancer Inst. 92, 569–573 (2000).
Peinado, H., Ballestar, E., Esteller, M. & Cano, A. Snail mediates E-cadherin repression by the recruitment of the Sin3A/histone deacetylase 1 (HDAC1)/HDAC2 complex. Mol. Cell Biol. 24, 306–319 (2004).
Hou, Z. et al. The LIM protein AJUBA recruits protein arginine methyltransferase 5 to mediate SNAIL-dependent transcriptional repression. Mol. Cell Biol. 28, 3198–3207 (2008).
Herranz, N. et al. Polycomb complex 2 is required for E-cadherin repression by the Snail1 transcription factor. Mol. Cell Biol. 28, 4772–4781 (2008).
Wellner, U. et al. The EMT-activator ZEB1 promotes tumorigenicity by repressing stemness-inhibiting microRNAs. Nat. Cell Biol. 11, 1487–95 (2009).
Yang, M. H. & Wu, K. J. TWIST activated by hypoxia inducible factor-1 (HIF-1): implication in metastasis and development. Cell Cycle 7, 2090–2096 (2008).
Li, J. et al. Oncoprotein Bmi-1 renders apoptotic resistance to glioma cells through activation of the IKK-nuclear factor-κB pathway. Am. J. Pathol. 176, 699–709 (2010).
Vormittag, L. et al. Co-expression of Bmi-1 and podoplanin predicts overall survival in patients with squamous cell carcinoma of the head and neck treated with radio(chemo)therapy. Int. J. Radiat. Oncol. Biol. Phys. 73, 913–918 (2009).
Gustafsson, M. V. et al. Hypoxia requires notch signaling to maintain the undifferentiated cell state. Dev. Cell 9, 617–628 (2005).
Timmerman, L. A. et al. Notch promotes epithelial–mesenchymal transition during cardiac development and oncogenic transformation. Genes Dev. 18, 99–115 (2004).
Androutsellis-Theotokis, A. et al. Notch signalling regulates stem cell numbers in vitro and in vivo. Nature 442, 823–826 (2006).
Huang, C. H. et al. Regulation of membrane-type 4 matrix metalloproteinase (MT4-MMP) by SLUG contributes to hypoxia-mediated metastasis. Neoplasia 11, 1371–1382 (2009).
Yang, M. H. et al. Increased NBS1 expression is a marker of aggressive head and neck cancer and overexpression of NBS1 contributes to transformation. Clin. Cancer Res. 12, 507–515 (2006).
Ginestier, C. et al. ALDH1 is a marker of normal and malignant human mammary stem cells and a predictor of poor clinical outcome. Cell Stem Cell 1, 555–567 (2007).
Huang, T. S. et al. Functional network reconstruction reveals somatic stemness genetic maps and dedifferentiation-like transcriptome reprogramming induced by GATA2. Stem Cells 26, 1186–1201 (2008).
Wang, H. W. et al. Kaposi sarcoma herpesvirus-induced cellular reprogramming contributes to the lymphatic endothelial gene expression in Kaposi sarcoma. Nat. Genet. 36, 687–693 (2004).
Birnie, R. et al. Gene expression profiling of human prostate cancer stem cells reveals a pro-inflammatory phenotype and the importance of extracellular matrix interactions. Genome Biol. 9, R83 (2008).
Yang, M. H. et al. Overexpression of NBS1 induces epithelial–mesenchymal transition and co-expression of NBS1 and Snail predicts metastasis of head and neck cancer. Oncogene 26, 1459–1467 (2007).
Ainbinder, E., Amir-Zilberstein, L., Yamaguchi, Y., Handa, H. & Dikstein, R. Elongation inhibition by DRB sensitivity-inducing factor is regulated by the A20 promoter via a novel negative element and NF-κB. Mol. Cell Biol. 24, 2444–2454 (2004).
Kang, M. K. et al. Elevated Bmi-1 expression is associated with dysplastic cell transformation during oral carcinogenesis and is required for cancer cell replication and survival. Br. J. Cancer 96, 126–133 (2007).
Foschini, M. P. et al. E-cadherin loss and Delta Np73L expression in oral squamous cell carcinomas showing aggressive behavior. Head Neck 30, 1475–1482 (2008).
Kumar, B. et al. EGFR, p16, HPV Titer, Bcl-xL and p53, sex, and smoking as indicators of response to therapy and survival in oropharyngeal cancer. J. Clin. Oncol. 26, 3128–3137 (2008).
Acknowledgements
We would like to thank M. C. Hung (M. D. Anderson Cancer Center) for critical comments on the manuscript. We thank L. E. Huang (University of Utah) for pHA-HIF1α(ΔODD) plasmid. We are grateful to T.Y. Chou and W.Y. Li of the Department of Pathology, Taipei Veterans General Hospital for providing advice on IHC analysis. This work was supported in part by National Science Council grants to M.H.Y. (96-2314-B-075-013 and 97-2314-B-010-003) and K.J.W.(97-2320-B-010-029), National Research Program for Genomic Medicine grants to K.J.W (DOH98-TD-G-111-027 and DOH99-TD-G-111-024); Taipei Veterans General Hospital grants to M.H.Y (VGH 98-C1-050; 98-ER2-008, 99-C1-077 and 99-ER2-007) and K.J.W (99-ER2-009), grants from Ministry of Education, Aim for the Top University Plan to M.H.Y (98A-C-T510 and 99A-C-T509) and to K.J.W (99A-C-T508), National Health Research Institutes grants to K.J.W (NHRI-EX-98-9611BI and NHRI-EX-99-9931BI) and a grant from Department of Health, Center of Excellence for Cancer Research at Taipei Veterans General Hospital (DOH99-TD-C-111-007) to M.H.Y.
Author information
Authors and Affiliations
Contributions
M.H.Y., D.S.S.H. and K.J.W. conceived and designed the experiments. M.H.Y., D.S.S.H., H.J.W., H.Y.L. and W.H.Y. performed the experiments with the assistance of C.H.H for in vivo work. H.W.W. performed the bioinformatics analysis. M.H.Y., D.S.S.H. and K.J.W. analysed the data. M.H.Y. and K.J.W. wrote the paper with the assistance of O.S.K.L. and H.W.W. The treatment and sample collection of head-and-neck cancer patients were performed by S.Y.K., S.K.T., S.Y.C., C.H.T. and M.H.Y.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 2858 kb)
Rights and permissions
About this article
Cite this article
Yang, MH., Hsu, DS., Wang, HW. et al. Bmi1 is essential in Twist1-induced epithelial–mesenchymal transition. Nat Cell Biol 12, 982–992 (2010). https://doi.org/10.1038/ncb2099
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb2099
This article is cited by
-
Harnessing function of EMT in cancer drug resistance: a metastasis regulator determines chemotherapy response
Cancer and Metastasis Reviews (2024)
-
Regulation and signaling pathways in cancer stem cells: implications for targeted therapy for cancer
Molecular Cancer (2023)
-
MicroRNAs as the pivotal regulators of cisplatin resistance in head and neck cancers
Cancer Cell International (2023)
-
TMPRSS4, a type II transmembrane serine protease, as a potential therapeutic target in cancer
Experimental & Molecular Medicine (2023)
-
Advancements in Understanding the Role of Circular RNA in Osteosarcoma
Molecular Biotechnology (2023)