Abstract
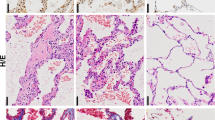
Accumulation of unwanted/misfolded proteins in aggregates has been observed in airways of patients with cystic fibrosis (CF), a life-threatening genetic disorder caused by mutations in the gene encoding the cystic fibrosis transmembrane conductance regulator (CFTR). Here we show how the defective CFTR results in defective autophagy and decreases the clearance of aggresomes. Defective CFTR-induced upregulation of reactive oxygen species (ROS) and tissue transglutaminase (TG2) drive the crosslinking of beclin 1, leading to sequestration of phosphatidylinositol-3-kinase (PI(3)K) complex III and accumulation of p62, which regulates aggresome formation. Both CFTR knockdown and the overexpression of green fluorescent protein (GFP)-tagged-CFTRF508del induce beclin 1 downregulation and defective autophagy in non-CF airway epithelia through the ROS–TG2 pathway. Restoration of beclin 1 and autophagy by either beclin 1 overexpression, cystamine or antioxidants rescues the localization of the beclin 1 interactome to the endoplasmic reticulum and reverts the CF airway phenotype in vitro, in vivo in Scnn1b-transgenic and CftrF508del homozygous mice, and in human CF nasal biopsies. Restoring beclin 1 or knocking down p62 rescued the trafficking of CFTRF508del to the cell surface. These data link the CFTR defect to autophagy deficiency, leading to the accumulation of protein aggregates and to lung inflammation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Mizushima, N., Levine, B., Cuervo, A. M. & Klionsky, D. J. Autophagy fights disease through cellular self-digestion. Nature 28, 1069–1075 (2008).
Moreau, K., Luo, S. & Rubinsztein, D. C. Cytoprotective roles for autophagy. Curr. Opin. Cell Biol. 22, 206–211 (2010).
Korolchuk, V. I., Mansilla, A., Menzies, F. M. & Rubinsztein, D. C. Autophagy inhibition compromises degradation of ubiquitin–proteasome pathway substrates. Mol. Cell 33, 517–527 (2009).
Kirkin, V., McEwan, D. G., Novak, I. & Dikic, I. A role for ubiquitin in selective autophagy. Mol. Cell 34, 259–269 (2009).
Bjørkøy, G. et al. p62/SQSTM1 forms protein aggregates degraded by autophagy and has a protective effect on huntingtin-induced cell death. J. Cell Biol. 171, 603–614 (2005).
Dohm, C. P., Kermer, P. & Bahr, M. Aggregopathy in neurodegenerative diseases: mechanisms and therapeutic implication. Neurodegen. Dis. 5, 321–338 (2008).
Williams, A. et al. Aggregate-Prone proteins are cleared from the cytosol by autophagy: Therapeutic Implications. Curr. Top. Dev. Biol. 76, 89–101 (2006).
Schessl, J., Zou, Y., McGrath, M. J., Cowling, B. S. & Maiti, B. Proteomic identification of FHL1 as the protein mutated in human reducing body myopathy. J. Clin. Invest. 118, 904–912 (2008).
Rodriguez-Gonzalez, A. et al. Role of the aggresome pathway in cancer: targeting histone deacetylase 6-dependent protein degradation. Cancer Res. 68, 2557–2560 (2008).
Maiuri, L. et al. Tissue transglutaminase activation modulates inflammation in cystic fibrosis via PPARγ down-regulation. J. Immunol. 180, 7697–7705 (2008).
Ratjen, F. & Doring, G. Cystic fibrosis. Lancet 361, 681–689 (2003).
Sha, Y., Pandit, L., Zeng, S. & Eissa, N. T. A critical role of CHIP in the aggresome pathway. Mol. Cell. Biol. 29, 116–128 (2009).
Bence, N. F., Sampat, R. M. & Kopito, R. R. Impairment of the ubiquitin–proteasome system by protein aggregation. Science 292, 1552–1555 (2001).
Fu, L. & Sztul, E. ER-associated complexes (ERACs) containing aggregated cystic fibrosis transmembrane conductance regulator (CFTR) are degraded by autophagy. Eur. J. Cell Biol. 88, 215–226 (2009).
Luciani, A. et al. SUMOylation of tissue transglutaminase as link between oxidative stress and inflammation. J. Immunol. 183, 2775–2784 (2009).
Trudel, S. et al. Peroxiredoxin 6 fails to limit phospholipid peroxidation in lung from Cftr-knockout mice subjected to oxidative challenge. PLoS One 4, e6075 (2009).
Sinha, S. & Levine, B. The autophagy effector Beclin 1: a novel BH3-only protein. Oncogene 27, S137–S148 (2008).
Maiuri, M. C., Criollo, A. & Kroemer, G. Crosstalk between apoptosis and autophagy within the Beclin 1 interactome. EMBO J. 29, 515–516 (2010).
He, C. & Levine, B. The Beclin 1 interactome. Curr. Opin. Cell Biol. 22, 140–149 (2010).
Klionsky, D. J., Abeliovich, H., Agostinis, P., Agrawal, D. K. & Aliev, G. Guidelines for the use and interpretation of assays for monitoring autophagy in higher eukaryotes. Autophagy 4, 151–175 (2008).
Mizushima, N., Yoshimori, T. & Levine, B. Methods in mammalian autophagy research. Cell 140, 313–326 (2010).
Raia, V. et al. Inhibition of p38 mitogen activated protein kinase controls airway inflammation in cystic fibrosis. Thorax 60, 773–780 (2005).
Legssyer, R. et al. Azithromycin reduces spontaneous and induced inflammation in DF508 cystic fibrosis mice. Respir. Res. 7, 134–136 (2006).
Matsunaga, K. et al. Two Beclin 1-binding proteins, Atg14L and Rubicon, reciprocally regulate autophagy at different stages. Nature Cell Biol. 11, 385–396 (2009).
Zhong, Y. et al. Distinct regulation of autophagic activity by Atg14L and Rubicon associated with Beclin 1-phosphatidylinositol-3-kinase complex. Nature Cell Biol. 11, 468–476 (2009).
Axe, E. L. et al. Autophagosome formation from membrane compartments enriched in phosphatidylinositol 3-phosphate and dynamically connected to the endoplasmic reticulum. J. Cell Biol. 182, 685–701 (2008).
Hayashi-Nishino, M. et al. A subdomain of the endoplasmic reticulum forms a cradle for autophagosome formation. Nature Cell Biol. 11, 1433–1437 (2009).
Maiuri, M. C. et al. Functional and physical interaction between Bcl-XL and a BH3-like domain in Beclin-1. EMBO J. 26, 2527–2539 (2007).
Pattingre, S. et al. Bcl-2 antiapoptotic proteins inhibit Beclin 1-dependent autophagy. Cell 23, 927–939 (2005).
Liang, C. et al. Beclin1-binding UVRAG targets the class C Vps complex to coordinate autophagosome maturation and endocytic trafficking. Nature Cell Biol. 10, 776–787 (2008).
Kawaguchi, Y. et al. The deacetylase HDAC6 regulates aggresome formation and cell viability in response to misfolded protein stress. Cell 115, 727–738 (2003).
Spencer, B. et al. Beclin 1 gene transfer activates autophagy and ameliorates the neurodegenerative pathology in α-synuclein models of Parkinson's and Lewy body diseases. J. Neurosci. 29, 13578–13588 (2009).
Stocker, A. G. et al. Single-dose lentiviral gene transfer for lifetime airway gene expression. J. Gene Med. 11, 861–867 (2009).
Lorand, L. & Graham, R. M. Transglutaminases: crosslinking enzymes with pleiotropic functions. Nature Rev. Mol. Cell Biol. 4, 140–156 (2003).
Akar, U. et al. Tissue transglutaminase inhibits autophagy in pancreatic cancer cells. Mol. Cancer Res. 5, 241–249 (2007).
Ron, D. & Walter, P. Signal integration in the endoplasmatic reticulum unfolded protein response. Nature Rev. Mol. Cell Biol. 8, 519–529 (2007).
Bartoszewski, R. et al. Activation of the unfolded protein response by ΔF508 CFTR. Am. J. Respir. Cell Mol. Biol. 39, 448–457 (2008).
Moscat, J. & Diaz-Meco, M. T. p62 at the crossroads of autophagy, apoptosis, and cancer. Cell 137, 1001–1004 (2009).
Mathew, R. et al. Autophagy suppresses tumorigenesis through elimination of p62. Cell 137, 1062–1075 (2009).
Komatsu, M. et al. Homeostatic levels of p62 control cytoplasmic inclusion body formation in autophagy-deficient mice. Cell 131, 1149–1163 (2007).
Riordan, J. R. CFTR function and prospects for therapy. Annu. Rev. Biochem. 77, 701–726 (2008).
Wang, X. et al. Hsp90 cochaperone Aha1 downregulation rescues misfolding of CFTR in cystic fibrosis. Cell 127, 803–815 (2006).
Wang, X., Koulov, A. V., Kellner, W. A., Riordan, J. R. & Balch, W. E. Chemical and biological folding contribute to temperature-sensitive ΔF508 CFTR trafficking. Traffic 11, 1878–1893 (2008).
Skach, W. R. CFTR: new members join the fold. Cell 127, 673–675 (2006).
Amaral, M. D. CFTR and chaperones: processing and degradation. J. Mol. Neurosci. 23, 41–48 (2004).
Pedemonte, N. et al. Small-molecule correctors of defective ΔF508-CFTR cellular processing identified by high-throughput screening. J. Clin. Invest. 115, 2564–2571 (2005).
Caohuy, H., Jozwik, C. & Pollard, H. B. Rescue of ΔF508-CFTR by the SGK1/Nedd4-2 signaling pathway. J. Biol. Chem. 284, 25241–25253 (2009).
Ghavami, S. et al. S100A8/A9 induces autophagy and apoptosis via ROS-mediated cross-talk between mitochondria and lysosomes that involves BNIP3. Cell Res. 20, 314–331 (2010).
Karpuj, M. V. et al. Prolonged survival and decreased abnormal movements in transgenic model of Huntington disease, with administration of the transglutaminase inhibitor cystamine. Nature Med. 8, 143–149 (2002).
Sablina, A. A., Budanov, A. V., Ilyinskaya, G. V., Agapova, L. S. & Kravchenko, J. E. The antioxidant function of the p53 tumor suppressor. Nature Med. 11, 1306–1313 (2005).
Mall, M., Grubb, B. R., Harkema, J. R., O'Neal, W. K. & Boucher, R. C. Increased airway epithelial Na+ absorption produces cystic fibrosis-like lung disease in mice. Nature Med. 10, 487–493 (2004).
Frizzell, R. A. & Pilewski, J. M. Finally, mice with CF lung disease. Nature Med. 10, 452–454 (2004).
Maiuri, C., Zalckvar, E., Kimchi, A. & Kroemer, G. Self-eating and self-killing: crosstalk between autophagy and apoptosis. Nature Rev Mol. Cell. Biol. 8, 741–752 (2007).
Hara, T. et al. Suppression of basal autophagy in neural cells causes neurodegenerative disease in mice. Nature 441, 885–889 (2006).
Takahashi, Y. et al. Bif-1 interacts with Beclin 1 through UVRAG and regulates autophagy and tumorigenesis. Nature Cell Biol. 9, 1142–1151 (2007).
Pickford, F. et al. The autophagy-related protein beclin 1 shows reduced expression in early Alzheimer disease and regulates amyloid beta accumulation in mice. J. Clin. Invest. 118, 2190–2199 (2008).
Tebbenkamp, A. T. & Borchel, D. R. Protein aggregate characterization in models of neurodegenerative disease. Methods Mol. Biol. 566, 85–91 (2009).
Martínez, A, Portero-Otin, M., Pamplona, R. & Ferrer, I. Protein targets of oxidative damage in human neurodegenerative diseases with abnormal protein aggregates. Brain Pathol. 20, 281–297 (2010).
Korolchuk, V. I., Menzies, F. M. & Rubinsztein, D. C. Mechanisms of cross-talk between the ubiquitin–proteasome and autophagy–lysosome systems. FEBS Lett. 584, 1393–1398 (2010).
Muma, N. A. Transglutaminase is linked to neurodegenerative disease. J. Neuropathol. Exp. Neurol. 66, 258–263 (2007).
Teichgräber, V. et al. Ceramide accumulation mediates inflammation, cell death and infection susceptibility in cystic fibrosis. Nature Med. 14, 382–391 (2008).
Vij, N., Mazur, S. & Zeitlin, P. L. CFTR is a negative regulator of NFκB mediated innate immune response. PLoS One 4, e4664 (2009).
Farinha, C. M. & Amaral, M. D. Most F508del-CFTR is targeted to degradation at an early folding checkpoint and independently of calnexin. Mol. Cell. Biol. 25, 5242–5252 (2005).
Scott-Ward, T. S. & Amaral, M. D. Deletion of Phe508 in the first nucleotide-binding domain of the cystic fibrosis transmembrane conductance regulator increases its affinity for the heat shock cognate 70 chaperone. FEBS J. 276, 7097–7109 (2009).
Rochat, T., Lacroix, J. S. & Jornot, L. N-acetylcysteine inhibits Na+ absorption across human nasal epithelial cells. J. Cell Physiol. 201, 106–116 (2004).
Luciani, A. et al. Lysosomal accumulation of gliadin p31-43 peptide induces oxidative stress and tissue transglutaminase-mediated PPARγ downregulation in intestinal epithelial cells and coeliac mucosa. Gut 59, 311–319 (2010).
Acknowledgements
We thank Noboru Mizushima for the gift of the pEGFP–LC3 and pcDNA3-HA–beclin 1 expression vectors; Ron Kopito for the gift of the pGFP–F508del-CFTR expression vector; Michael Bownlee for the gift of the adenoviral vectors; Gian Maria Fimia for the gift of the TG2 plasmid; Dieter C. Gruenert for the gift of CFBE41o− and 16HBE14o− cell lines; Maria Carla Panzeri for support in electron microscopy and in the analysis of the data; Rosarita Tatè for technical support in confocal microscopy; and Ilaria Russo for technical support in histology. Cftrtm1EUR (F508del (FVB/129) mice were obtained from Bob Scholte under European Economic Community European Coordination Action for Research in Cystic Fibrosis program EU FP6 LSHM-CT-2005-018932. This work was supported by the European Institute for Research in Cystic Fibrosis, Cancer Research UK, Rothschild Trust, Coeliac UK and Regione Campania (L. 229/99).
Author information
Authors and Affiliations
Contributions
A.L. co-designed the research concept, planned the overall experimental design, performed organ culture and confocal microscopy studies and wrote the manuscript. V.R.V. co-designed the research concept, planned the overall experimental design and performed immunoblot and immunoprecipitation experiments, cell cultures and transfections. S.E. contributed to the study design, interpretation and analysis of the data and performed immunoblot and immunoprecipitation experiments, cell cultures and transfections. N.B. contributed to the study design, provided scientific knowledge, contributed to the interpretation and analysis of the data, performed experiments on mice and wrote the manuscript. D.M. contributed to the study design, provided scientific knowledge, contributed to the interpretation and analysis of the data and performed the analysis of mitochondrial function. C.S. provided expression vectors and scientific knowledge and contributed to the analysis of the data. M.G. and L.P. performed experiments on mice and contributed to the interpretation and analysis of the data. I.G., M.P.M. and M.D. performed PCR and contributed to the interpretation and analysis of the data. S.G. contributed to the discussion of the data. E.M. and B.S. provided the lentiviral vectors and scientific knowledge. S.Q. contributed to the interpretation and analysis of the data and provided scientific knowledge. A.B. co-designed the research concept and co-supervised the project. V.R. and L.M. designed the research concept, planned the overall experimental design, supervised the study and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 3152 kb)
Rights and permissions
About this article
Cite this article
Luciani, A., Villella, V., Esposito, S. et al. Defective CFTR induces aggresome formation and lung inflammation in cystic fibrosis through ROS-mediated autophagy inhibition. Nat Cell Biol 12, 863–875 (2010). https://doi.org/10.1038/ncb2090
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb2090
This article is cited by
-
Cystic fibrosis transmembrane conductance regulator (CFTR): beyond cystic fibrosis
Egyptian Journal of Medical Human Genetics (2022)
-
Protein clearance strategies for disease intervention
Journal of Neural Transmission (2022)
-
Siah2–GRP78 interaction regulates ROS and provides a proliferative advantage to Helicobacter pylori-infected gastric epithelial cancer cells
Cellular and Molecular Life Sciences (2022)
-
Targeting CK2 mediated signaling to impair/tackle SARS-CoV-2 infection: a computational biology approach
Molecular Medicine (2021)
-
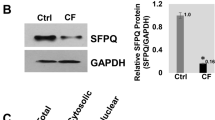
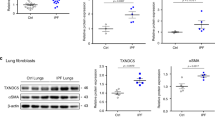
SFPQ rescues F508del-CFTR expression and function in cystic fibrosis bronchial epithelial cells
Scientific Reports (2021)