Abstract
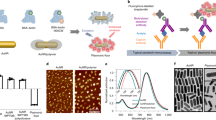
The current sensitivity of standard fluorescence-based protein detection limits the use of protein arrays in research and clinical diagnosis. Here, we use functionalized, macromolecular single-walled carbon nanotubes (SWNTs) as multicolor Raman labels for highly sensitive, multiplexed protein detection in an arrayed format. Unlike fluorescence methods, Raman detection benefits from the sharp scattering peaks of SWNTs with minimal background interference, affording a high signal-to-noise ratio needed for ultra-sensitive detection. When combined with surface-enhanced Raman scattering substrates, the strong Raman intensity of SWNT tags affords protein detection sensitivity in sandwich assays down to 1 fM—a three-order-of-magnitude improvement over most reports of fluorescence-based detection. We use SWNT Raman tags to detect human autoantibodies against proteinase 3, a biomarker for the autoimmune disease Wegener's granulomatosis, diluted up to 107-fold in 1% human serum. SWNT Raman tags are not subject to photobleaching or quenching. By conjugating different antibodies to pure 12C and 13C SWNT isotopes, we demonstrate multiplexed two-color SWNT Raman-based protein detection.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Bailey, R.C., Kwong, G.A., Radu, C.G., Witte, W.N. & Heath, J.R. DNA-encoded antibody libraries: a unified platform for multiplexed cell sorting and detection of genes and proteins. J. Am. Chem. Soc. 129, 1959–1967 (2007).
Hayes, F.J., Halsall, H.B. & Heineman, W.R. Simultaneous immunoassay using electrochemical detection of metal ion labels. Anal. Chem. 66, 1860–1865 (1994).
Landry, J.P., Zhu, X.D. & Gregg, J.P. Label-free detection of microarrays of biomolecules by oblique-incidence reflectivity difference microscopy. Opt. Lett. 29, 581–583 (2004).
Wegner, G.J., Lee, H.J. & Corn, R.M. Characterization and optimization of peptide arrays for the study of epitope-antibody interactions using surface plasmon resonance imaging. Anal. Chem. 74, 5161–5168 (2002).
Cao, Y.C., Jin, R., Nam, J.M., Thaxton, C.S. & Mirkin, C.A. Raman dye-labeled nanoparticle probes for proteins. J. Am. Chem. Soc. 125, 14676–14677 (2003).
Li, T., Guo, L. & Wang, Z. Microarray based Raman spectroscopic detection with gold nanoparticle probes. Biosens. Bioelectron. 23, 1125–1130 (2008).
Grubisha, D.S., Lipert, R.J., Park, H.Y., Driskell, J. & Porter, M.D. Femtomolar detection of prostate-specific antigen: an immunoassay based on surface-enhanced Raman scattering and immunogold labels. Anal. Chem. 75, 5936–5943 (2003).
Wu, G. et al. Bioassay of prostate-specific antigen (PSA) using microcantilevers. Nat. Biotechnol. 19, 856–860 (2001).
Goldman, E.R. et al. Conjugation of luminescent quantum dots with antibodies using an engineered adaptor protein to provide new reagents for fluoroimmunoassays. Anal. Chem. 74, 841–847 (2002).
Nam, J.M., Thaxton, C.S. & Mirkin, C.A. Nanoparticle-based bio-bar codes for the ultrasensitive detection of proteins. Science 301, 1884–1886 (2003).
Chen, R.J. et al. An investigation of the mechanisms of electronic sensing of protein adsorption on carbon nanotube devices. J. Am. Chem. Soc. 126, 1563–1568 (2004).
Kong, J. et al. Nanotube molecular wires as chemical sensors. Science 287, 622–625 (2000).
Zheng, G., Patolsky, F., Cui, Y., Wang, W.U. & Lieber, C.M. Multiplexed electrical detection of cancer markers with nanowire sensor arrays. Nat. Biotechnol. 23, 1294–1301 (2005).
MacBeath, G. & Schreiber, S.L. Printing proteins as microarrays for high-throughput function determination. Science 289, 1760–1763 (2000).
Hudson, M.E., Pozdnyakova, I., Haines, K., Mor, G. & Snyder, M. Identification of differentially expressed proteins in ovarian cancer using high-density protein microarrays. Proc. Natl. Acad. Sci. USA 104, 17494–17499 (2007).
Robinson, W.H. et al. Autoantigen microarrays for multiplex characterization of autoantibody responses. Nat. Med. 8, 295–301 (2002).
Song, S. et al. A cancer protein microarray platform using antibody fragments and its clinical applications. Mol. Biosyst. 3, 151–158 (2007).
Espina, V. et al. Protein microarray detection strategies: focus on direct detection technologies. J. Immunol. Methods 290, 121–133 (2004).
Hartwell, L., Mankoff, D., Paulovich, A., Ramsey, S. & Swisher, E. Cancer biomarkers: a systems approach. Nat. Biotechnol. 24, 905–908 (2006).
Jensen, L. & Schatz, G.C. Resonance Raman scattering of rhodamine 6G as calculated using time-dependent density functional theory. J. Phys. Chem. A 110, 5973–5977 (2006).
Baughman, R.H., Zakhidov, A.A. & de Heer, W.A. Carbon nanotubes–the route toward applications. Science 297, 787–792 (2002).
Heller, D.A., Baik, S., Eurell, T.E. & Strano, M.S. Single-walled carbon nanotube spectroscopy in live cells: towards long-term labels and optical sensors. Adv. Mater. 17, 2793–2799 (2005).
Sun, Y.P., Fu, K.F., Lin, Y. & Huang, W.J. Functionalized carbon nanotubes: properties and applications. Acc. Chem. Res. 35, 1096–1104 (2002).
Bahr, J.L. & Tour, J.M. Covalent chemistry of single-wall carbon nanotubes. J. Mater. Chem. 12, 1952–1958 (2002).
Kam, N.W.S., Liu, Z. & Dai, H.J. Functionalization of carbon nanotubes via cleavable disulfide bonds for efficient intracellular delivery of siRNA and potent gene silencing. J. Am. Chem. Soc. 127, 12492–12493 (2005).
Liu, Z. et al. In vivo biodistribution and highly efficient tumor targeting of carbon nanotubes in mice. Nat. Nanotechnol. 2, 47–52 (2007).
Chen, R.J. et al. Noncovalent functionalization of carbon nanotubes for highly specific electronic biosensors. Proc. Natl. Acad. Sci. USA 100, 4984–4989 (2003).
Schipper, M.L. et al. A pilot toxicology study of single-walled carbon nanotubes in a small sample of mice. Nat. Nanotechnol. 3, 216–221 (2008).
Welsher, K. & Liu, Z. Daranciang & Dai, H.J. Selective probing and imaging of cells with single walled carbon nanotubes as near-infrared fluorescence molecules. Nano Lett. 8, 586–590 (2008).
Nie, S. & Emory, S.R. Probing single molecules and single nanoparticles by surface-enhanced Raman scattering. Science 275, 1102–1106 (1997).
Kneipp, K., Kneipp, H. & Kneipp, J. Surface-enhanced Raman scattering in local optical fields of silver and gold nanoaggregates - from single-molecule Raman spectroscopy to ultrasensitive probing in live cells. Acc. Chem. Res. 39, 443–450 (2006).
Shim, M., Kam, N.W.S., Chen, R.J., Li, Y. & Dai, H.J. Functionalization of carbon nanotubes for biocompatibility and biomolecular recognition. Nano Lett. 2, 285–288 (2002).
Diamandis, E.P. & Christopoulos, T.K. Immunoassay (Academic Press, San Diego, 1996).
Fang, Y., Seong, N.H. & Dlott, D.D. Measurement of the distribution of site enhancement in surface-enhanced Raman scattering. Science 321, 388–392 (2008).
Jenne, D.E., Tschopp, J., Lüdemann, J., Utecht, B. & Gross, W.L. Wegener's autoantigen decoded. Nature 346, 520 (1990).
Seo, P. & Stone, J.H. The antineutrphil cytoplasmic antibody-associated vasculitides. Am. J. Med. 117, 39–50 (2004).
Li, X. et al. Selective synthesis combined with chemical separation of single-walled carbon nanotubes for chirality selection. J. Am. Chem. Soc. 129, 15770–15771 (2007).
Liu, L. & Fan, S.S. Isotope labeling of carbon nanotubes and formation of 12C-13C nanotube junctions. J. Am. Chem. Soc. 123, 11502–11503 (2001).
Ferraro, J.R., Nakamoto, K. & Brown, C.W . Introductory Raman Spectroscopy edn. 2 (Academic Press, San Diego, 2003).
Arnold, M.S., Green, A.A. & Hulvat, J.F. Stupp. S.I. & Hersam, M.C. Sorting carbon nanotubes by electronic structure using density differentiation. Nat. Nanotechnol. 1, 60–65 (2006).
Tu, X. & Zheng, M. A DNA-based approach to the carbon nanotube sorting problem. Nano. Res. 1, 185–194 (2008).
Liu, Z. et al. Multiplexed mutli-color Raman imaging of live cells with isotopically modified single walled carbon nanotubes. J. Am. Chem. Soc. doi:10.1021/ja806242t (20 September 2008).
Liu, Z. et al. Circulation and long-term fate of functionalized, biocompatible single-walled carbon nanotubes in mice probed by Raman spectroscopy. Proc. Natl. Acad. Sci. USA 105, 1410–1415 (2008).
Kam, N.W.S., Connell, M.O., Wisdom, J.A. & Dai, H.J. Carbon nanotubes as multifunctional biological transporters and near-infrared agents for selective cancer cell destruction. Proc. Natl. Acad. Sci. USA 102, 11600–11605 (2005).
Acknowledgements
This work was supported by the National Institutes of Health/National Cancer Institute–funded Center for Cancer Nanotechnology Excellence Focused on Therapeutic Response U54 CA119367 at Stanford University, and NIH-NCI R01 CA135109-01. The authors would like to thank Nozomi Nakayama-Ratchford and Sarunya Bangsaruntip for their assistance in developing carbon nanotube–protein conjugates.
Author information
Authors and Affiliations
Contributions
Z.C. and S.M.T. contributed equally to the work, developing the SWNT passivation and conjugation schemes presented, as well as the procedure for obtaining SERS-active surfaces and the related protein immobilization methodology. A.P.G. contributed synthesis of 6-arm branched carboxy-PEG. M.G.K. and P.J.U. contributed to fluorescence-based detection of anti-proteinase 3. D.D. and Z.L. contributed by assisting in preparing SWNT-antibody and SWNT-peptide conjugates. X.W. and G.Z. contributed by assisting in the preparation of SERS-active substrates. X.L., K.J. and S.F. contributed by providing isotopomer SWNTs. H.D. designed the research and contributed much direction and assistance to this project.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–15, Supplementary Table 1, Supplementary Methods (PDF 724 kb)
Rights and permissions
About this article
Cite this article
Chen, Z., Tabakman, S., Goodwin, A. et al. Protein microarrays with carbon nanotubes as multicolor Raman labels. Nat Biotechnol 26, 1285–1292 (2008). https://doi.org/10.1038/nbt.1501
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt.1501
This article is cited by
-
Nanomaterial-assisted microfluidics for multiplex assays
Microchimica Acta (2022)
-
Ultrabright fluorescent nanoscale labels for the femtomolar detection of analytes with standard bioassays
Nature Biomedical Engineering (2020)
-
Real-time Raman detection by the cavity mode enhanced Raman scattering
Nano Research (2019)
-
Biomimetic photonic structures with tunable structural colours: From natural to biomimetic to applications
Journal of Bionic Engineering (2018)
-
Light in diagnosis, therapy and surgery
Nature Biomedical Engineering (2017)