Abstract
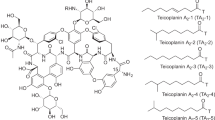
A series of lipophilic teicoplanin pseudoaglycon derivatives, including alkyl-, aryl-, calixarene- and protected sugar-containing conjugates, were prepared using azide–alkyne click chemistry. Out of the conditions applied, the CuSO4–ascorbate reagent system proved to be more efficient than the Cu(I)I–Et3N-mediated reaction. Some of the new compounds have high in vitro activity against glycopeptide-resistant Gram-positive bacteria, including vanA-positive Enterococcus faecalis. A few of them also display promising in vitro anti-influenza activity.
Similar content being viewed by others
Introduction
Glycopeptides are the drugs of first choice in cases of severe Gram-positive infections, which are mostly caused by pathogens, such as methicillin-resistant Staphylococcus aureus, coagulase-negative staphylococci and enterococci.1 The site of action of these agents is the bacterial peptidoglycan. They bind to the l-Lys-d-Ala-d-Ala termini of peptidoglycan precursors by five hydrogen bonds. As a result, the transglycosylation and transpeptidation steps in cell wall synthesis are inhibited. It is well known that this lack of structural integrity of the cell wall ultimately leads to osmotic lysis of bacterial cells.2
Resistance against glycopeptide antibiotics has been emerging for a few decades now, probably as a result of their extensive use in both clinical and agricultural settings.3, 4 For many years, vancomycin was the only glycopeptide used clinically. In the 1980s, vancomycin resistance started to develop. A new glycopeptide, teicoplanin, was approved in the late 1980s in Europe (and later in Japan) and became an alternative treatment option. Later, teicoplanin resistance among vancomycin-resistant enterococci (VRE) and coagulase-negative staphylococci became more and more frequent.5, 6 The newest semisynthetic glycopeptides—dalbavancin, telavancin and oritavancin—are several times more efficient than their predecessors, with oritavancin being the most potent and active even against VRE.7, 8, 9
The strong activity of these new antibiotics proves that carefully selected lipophilic substituents can enhance the activity of the parent compounds by different ways thereby overcoming the problem of glycopeptide resistance. Our group has already prepared a variety of such semisynthetic glycopeptides from vancomycin aglycon, ristocetin aglycon and teicoplanin pseudoaglycon, by using different chemical approaches. These include the conjugation with a squaric acid moiety, isoindole formation and the incorporation of variously substituted maleimides.10, 11, 12 Many of these compounds showed good in vitro activity against various bacteria, including VRE, and interestingly, some of them proved to be efficient against different strains of influenza virus.
Two teicoplanin pseudoaglycon derivatives that were prepared earlier by click chemistry showed outstanding antibacterial activity.13 One of them had a 4-(decyloxymethyl)-1,2,3-triazole side chain derivatized from the terminal amino group of the molecule by a diazotransfer and a subsequent azide–alkyne click reaction. The other derivative had a biphenyl group instead of the n-decyl group.
In the present work, our aim was to obtain structure–activity relationships in this group of derivatives by synthesizing compounds with a larger variety of side chains, as it is known from the literature that an optimal length of substituents is required for maximum activity.14 The preparation of new derivatives was accomplished by using the aforementioned CuAAC approach with varying alkyl and aryl side chain lengths and also with substituents that have more diverse chemical structures. Besides, we were interested in the potential anti-influenza activity of the new compounds.
Results and Discussion
To form the various side chains on the teicoplanin pseudoaglycon, we used different alkynyl ethers. These included a series of propargyl ethers such as n-hexadecyl (3a),15 n-dodecyl (3b),16 n-decyl (3c),13 n-octyl (3d),16 n-hexyl (3e), phenyl (3f) and α-naphthyl propargyl ether (3g).17 Compound 3c was prepared to resynthesize the corresponding teicoplanin pseudoaglycon n-decyl derivative 5c13 for the current biological evaluations. Two protected carbohydrate derivatives, 1,2:3,4-di-O-isopropylidene-6-O-(2-propargyl)-α-D-galactopyranose (3h)18 and a highly bulky 3′-O-propargyl lactose derivative (3i) were also utilized as lipophilic moieties. Compound 3i was prepared by stannylene acetal-mediated regioselective propargylation of the 3′-hydroxyl group of diol 1i19 using dibutyltin oxide and propargyl bromide followed by benzylation of the remaining free hydroxyl group of the resulting 2i (Scheme 1). The calix[4]arene derivative (3j)20 bearing a n-butynyl moiety was also incorporated within our studies.
Teicoplanin pseudoaglycon azide (4)13 was reacted with alkynyl ethers (3a–3j) in a Cu(I)-catalyzed azide–alkyne click reaction21 to afford final products 5a–5j in low-to-moderate yields (Scheme 2). Upon CuI-TEA catalysis in dimethylformamide (DMF), insufficient conversion of the reactants was observed and the yield of the corresponding triazole derivative after purification did not exceed 35%. We could achieve better conversions and significantly higher yields using the CuSO4-L-ascorbate-mediated reaction in tBuOH—water, 1:1 (v/v). (see Table 1). However, there is a limiting factor when using the latter method, which is the solubility of the chosen reactants in the tBuOH-water mixture. All of our teicoplanin pseudoaglycon derivatives have lipophilic side chains, therefore calculated logP values for the alkynyl ethers 3a–j can be informative for structure–biological activity studies (Table 1).
The antibacterial activity of the new derivatives was evaluated by the broth microdilution method10, 22 on a panel of Gram-positive bacteria (Table 2). Derivative 5a containing the n-hexadecyl group turned out to be multiple times less active than teicoplanin or showed no activity at all. Compound 5b, with the n-dodecyl side chain, was less active against both methicillin-sensitive Staphylococcus aureus and methicillin-resistant Staphylococcus aureus than teicoplanin, but it had good MIC/MBC values in the case of both S. epidermidis strains. It showed moderate activity against the enterococci. Compound 5c bearing the n-decyl side chain was more active against enterococci than the n-dodecyl derivative 5b. Compound 5d with the n-octyl side chain was highly active against all bacteria tested, showing excellent bactericidal effect. Although 5e had similar or better bacteriostatic activity, it was not exceptionally bactericidal against enterococci. 5f and 5g with the aromatic substituents were even more active against resistant enterococci. Both of them displayed not only marked bacteriostatic but also noticeable bactericidal effect against the three Enterococcus faecalis strains (vancomycin sensitive/resistant/teicoplanin resistant). Derivative 5h showed excellent activity against both S. epidermidis strains. Unfortunately, it was not particularly active against E. faecalis. 5i was totally inactive against the tested bacteria, possibly owing to a shielding effect of the bulky benzyl groups, which prevents the peptide core from binding to the molecular target. The same phenomenon was proposed in the case of compound 5j, which was not active against any of the bacteria tested.
As shown in Table 2, our recent measurement of MIC/MBC ratios are considerably different among derivatives with strikingly analogous structures. This fact may indicate the different activity of a compound against different bacterial strains or the different mode of action among derivatives against the same examined bacterial strain. The first case may result in the detectable different MIC/MBC ratios of one antibiotic examined in different strains from the same species.23 The practice of clinical microbiology laboratories to support the selection of effective therapy is the determination of MIC for different antibiotics of the pathogen. The determination of MBC, postantibiotic effect or postantibiotic sub-MIC effect is not usual routine diagnostics; however, these characteristics are different against the antibiotics and strongly influence the process of treatment. Odenholt et al.24 demonstrated high teicoplanin concentration-dependent killing of S. epidermidis and slight concentration-dependent killing of S. aureus, but for Enterococcus faecium only a bacteriostatic effect was noted. In that study, no or very short postantibiotic effects were noted for the investigated strains, but a lot of differences in regrowth were detected in the postantibiotic phase by sub-MICs against the different investigated species.24 The different MIC/MBC ratios of our study may be caused by similar specificity of the investigated derivatives.
The results indicate that modification of the teicoplanin pseudoaglycon with highly lipophilic (5a) and/or very bulky (5i, 5j) substituents eliminates the antibacterial effect. This clearly demonstrates that both the size/bulkiness and lipophilicity of the side chain have a strong influence on antibacterial activity, and the n-octyl, phenyl and α-naphthyl groups seem to be optimal. It is not clear whether aromaticity of the latter substituents has an impact on the activity, but the lipophilicity of the substituents in itself is clearly not the sole reason for biological properties of these compounds.
Oritavancin and other structurally related lipoglycopeptide derivatives are known to directly inhibit the transglycosylation step of peptidoglycan synthesis.25 Also, oritavancin bearing a chlorophenylbenzyl group was shown to cause disruption of the bacterial membrane.26 Moreover, semisynthetic lipoglycopeptides can form dimers more efficiently than vancomycin, and their enhanced antibacterial activity was attributed to their capability to dimerize, rather than their binding affinity to the cell-wall precursors. The membrane anchoring ability of such derivatives—which is due to their hydrophobic side chain—is also considered to be a relevant factor in the inhibition of VRE.27 As the mode of action of semisynthetic glycopeptide antibiotics seems rather complex, it is difficult to hypothesize which factors are responsible for the efficacy of the compounds presented in this paper, as no extensive mechanistic studies have been carried out thus far.
In addition, we evaluated the anti-influenza virus activity of the new compounds against different strains of influenza virus, using an established assay in Madin Darby canine kidney cell cultures28 (Table 3). Compound 5a (with n-hexadecyl group) had strong activity against all influenza strains tested, albeit at antiviral EC50 values that were only approximately threefold lower than the concentrations giving cytotoxicity. When comparing compounds 5a, 5b, 5c, 5d and 5e, it was intriguing that the analogs having an n-hexadecyl (5a) or n-octyl side chain (5d) displayed robust and comparable antiviral activity, whereas the analogs having an n-dodecyl (5b) or n-decyl side chain (5c) were inactive. This is even more remarkable in light of the finding that these four analogs had very similar cytotoxic activity. Compound 5e having an n-hexyl substituent displayed diminished activity against two of the four strains, while also being less cytotoxic than the other alkyl analogs, possibly owing to reduced lipophilicity of the substituent. This implies that the biochemical basis for the antiviral activity is critically dependent on the length of the aliphatic side chain and, second, that this antiviral activity is not strictly correlated to cytotoxicity. Although compound 5f (having a phenyl group) was only slightly active against one of the four strains, 5g (having a α-naphthyl group) exhibited more pronounced activity. These two compounds also had lower cytotoxicity than the alkyl substituted derivatives. On the other hand, the analogs 5i and 5j displayed not only reduced cytotoxicity but also lower antiviral activity that was less consistent for the four virus strains tested. Finally, among this compound series, 5h had the lowest antiviral activity and cytotoxicity.
In summary, we have prepared a small series of semisynthetic teicoplanin pseudoaglycon derivatives having different lipophilic moieties on the N-terminal by click chemistry. The new compounds possess significant in vitro biological activity. It is evident that the size and lipophilicity of the newly incorporated substituent are major determinants for activity against glycopeptide-resistant enterococci. In particular, the phenyl and α-naphthyl moieties were recognized as optimal substituents in this triazole functionalized group of derivatives. In the future, one or more of these candidates may be tested against a larger collection of VRE and in ex vivo or in vivo infection models.
Some of the new compounds show robust anti-influenza activity; however, they are also cytotoxic. Interestingly, while high hydrophobicity and bulkiness of side chains were detrimental for the antibacterial activity, these properties were favored to obtain anti-influenza effect. This anti-influenza activity critically depended on the length of the aliphatic side chain as n-octyl and n-hexadecyl provided equal activity but the analogs bearing n-dodecyl and n-decyl were inactive. We are currently investigating additional modifications with the hope to obtain compounds with improved antiviral activity and minimal cytotoxicity.
Experimental procedure
General information
Reagents were purchased from Sigma Aldrich Chemical Co., Budapest, Hungary, alkyne compounds 3a,15 3b,16 3c,13 3d,16 3e, 3g,17 3h,18 3j20 and teicoplanin pseudoaglycon azide (4)13 were prepared as described in other papers. Phenyl propargyl ether (3f) (technical, ⩾90%) was purchased from Sigma Aldrich Chemical Co. TLC analysis was performed on Kieselgel 60 F254 (Merck, Budapest, Hungary) silica gel plates with visualization by immersing in ammonium molybdate solution followed by heating or Pauly reagent in the case of teicoplanin derivatives. Column chromatography was performed on silica gel 60 (Merck; 0.063–0.200 mm). Organic solutions were dried over MgSO4 and concentrated under vacuum. The logP values of the alkynyl ethers 3a–j were calculated using the logP calculation plugin of ChemAxon’s Marvin Sketch (VG method, 0.1 m electrolyte concentration). The 1H (360, 400 and 500 MHz) and 13C NMR (90.54, 100.28, 125.76 MHz) spectra were recorded with Bruker DRX-360, Bruker DRX-400 and Bruker Avance II 500 spectrometers. Chemical shifts are referenced to Me4Si or dextran sodium sulfate (0.00 p.p.m. for 1H) and to solvent signals (CDCl3: 77.00 p.p.m., CD3OD: 49.15 p.p.m., DMSO-d6: 39.51 p.p.m. for 13C). MS (matrix assisted laser desorption/ionization time-of-flight MS (MALDI-TOF)) analysis was carried out in positive reflectron mode on a BIFLEX III mass spectrometer (Bruker, Bremen, Germany) with delayed-ion extraction. The matrix solution was a saturated solution of 2,4,6-trihydroxy-acetophenone in MeCN. ESI-TOF MS spectra were recorded by a microTOF-Q type QqTOFMS mass spectrometer (Bruker) in the positive ion mode using MeOH as the solvent. Elemental analysis (C, H, N, S) was performed on an Elementar Vario Micro Cube (Elementar, Hanau, Germany) instrument. The antibacterial activity of 5a–5j was tested against a panel of Gram-positive bacteria using broth microdilution method as described earlier.22
General method A for azide–alkyne click reaction
To a stirred solution of teicoplanin pseudoaglycon azide (4) in N,N-dimethylformamide, an alkyne compound (1.25 equiv.), Et3N (1.0 equiv.) and Cu(I)I (0.5 equiv.) were added under argon atmosphere and stirred for overnight at room temperature. The solvents were evaporated and the crude product was purified by flash chromatography in toluene:methanol 8:2→1:1. The product obtained was passed through a column containing Sephadex LH-20 gel in methanol.
General method B for azide–alkyne click reaction
To a stirred solution of teicoplanin pseudoaglycon azide (4) in t-butanol:water=1:1 (1 ml), an alkyne compound (1.25 equiv.), L-ascorbic acid (1.0 equiv.) and CuSO4 × 5 H2O (0.1 equiv.) were added and stirred for overnight at room temperature. The solvents were evaporated and the crude product was purified by flash chromatography in toluene:methanol 8:2→1:1. The product obtained was passed through a column containing Sephadex LH-20 gel in methanol.
Compound 2i
Compound 1i (1.148 g, 1.29 mmol) was dissolved in toluene (50 ml) and dibutyltin oxide (421 mg, 1.3 equiv.) was added. The reaction mixture was stirred for 3 h under reflux using a Dean–Stark apparatus. After evaporation, it was dissolved in DMF (20 ml), and then cesium fluoride (396 mg, 2 equiv.) and propargyl bromide (296 μl, 2 equiv.) were added. The reaction mixture was stirred overnight at room temperature. After evaporating the solvent, the residue was dissolved in CH2Cl2 (50 ml) and washed with distilled water three times (3 × 15 ml). The organic phase was dried, filtered and the solvents were evaporated. Then the crude product was purified by silica gel column chromatography in n-hexane–ethyl acetate, 1:1 (v/v) to give 2i (830 mg, 69%), [α]D −8.84 (c 0.18, CHCl3). 1H NMR (500 MHz, CDCl3) δ: 7.65 (2H, m), 7.60–7.46 (5H, m), 7.45–7.34 (17H, m), 7.33–7.28 (6H, m), 5.14 (1H, d, J=10.5 Hz), 4.91–4.80 (6H, m), 4.74 (1H, d, J=10 Hz), 4.65 (1H, d, J=12 Hz), 4.55 (3H, m), 4.50 (1H, d, J=4.5 Hz), 4.47 (1H, s), 4.42 (2H, m), 4.18 (1H, s), 4.10 (1H, t, J=9.5 Hz), 3.94–3.90 (1H, m), 3.84 (1H, d, J=10.5 Hz), 3.76–3.69 (2H, m), 3.66 (1H, t, J=8.5 Hz), 3.60–3.52 (3H, m), 3.48 (2H, t, J=6 Hz), (2.47 1H, s). 13C NMR (500 MHz, CDCl3) δ: 138.9, 138.5, 138.4, 138.2, 138.1, 133.7, 132.0,128.8, 128.3, 128.2, 128.2, 128.1, 128.1, 127.8, 127.7, 127.6, 127.5, 127.4, 127.3, 127.3, 102.5 (C-1′), 87.4 (C-1), 84.9, 80.9, 80.2, 79.9 (Cqpropargyl), 79.3 (2C), 76.3, 75.5, 75.4, 75.2, 74.7, 73.4, 73.1, 72.8, 68.3, 68.4, 66.7, 57.8. Anal. calcd. for C56H58O10S C 72.86, H 6.33, S 3.47. Found: C 72.82, H 6.40, S 3.40.
Compound 3i
Sodium hydride (54 mg, 1.5 equiv.) was suspended in DMF (10 ml) in a flask, and the solution of 2i (830 mg, 0.94 mmol) in DMF (20 ml) was added dropwise at 0 °C temperature. The mixture was stirred for 30 min, then benzyl bromide (145 μl, 1.3 equiv.) was added and the reaction mixture was stirred overnight at room temperature. Then a small amount of methanol was added and the solvents were evaporated. The residue was dissolved in CH2Cl2 (50 ml), washed with distilled water three times (3 × 15 ml). The organic phase was dried and filtered, the solvents were evaporated and the crude product was purified by silica gel column chromatography in n-hexane:ethyl acetate=1:1 to give 3i (500 mg, 58%), [α]D −18.91 (c 0.17, CHCl3). 1H NMR (500 MHz, CDCl3) δ: 7.58–7.53 (2H, m, arom.), 7.42–7.09 (33 H, m, arom.), 5.08 (1H, d, J=11 Hz), 4.96 (1H, d, J=11.5 Hz), 4.81–4.62 (6H, m), 4.54 (2H, t, J=12 Hz), 4.45–4.38 (2H, m), 4.35–4.20 (6H, m), 3.98–3.90 (2H, m), 3.83–3.66 (4H, m), 3.60 (1H, t, J=8,5 Hz), 3.52–3.32 (3H, m), 2.43 (1H, s, CHpropargyl).13C NMR (500 MHz, CDCl3) δ: 139.0, 138.9, 138.7, 138.4, 138.3, 138.1, 133.7, 132.0, 129.7, 128.8, 128.3, 128.2, 128.2, 128.1, 128.1, 128.0, 127.8, 127.7, 127.6, 127.5, 127.4, 127.4, 127.3, 127.3, 127.1, 102.7 (C-1′), 87.4 (C-1), 84.9, 82.0, 80.3, 80.1, 80.0, 79.8, 79.3, 76.4, 75.5, 75.4, 75.2, 74.7, 74.3, 74.2, 73.3, 68.3, 68.1, 58.4. Anal. calcd. for C63H64O10S C 74.68, H 6.37, S 3.16. Found: C 74.75, H 6.40, S 3.14.
Compound 5a
Teicoplanin pseudoaglycon azide (4) (100 mg, 0.07 mmol) and compound 3a (30 mg) were reacted according to general method A, yielding compound 5a (40 mg, 33%) as a pale yellow powder. NMR data can be found in Supplementary Tables S1 and S2 (Supplementary Information). MS (MALDI-TOF) m/z 1729.56 (M+Na+) (1729.57 calcd. for C85H92Cl2N10O24Na [M+Na+]). Anal. calcd. for C85H92Cl2N10O24 C 59.75, H 5.43, N 8.20. Found: C 59.72, H 5.40, N 8.14.
Compound 5b
Compound 4 (100 mg, 0.07 mmol) and compound 3b (24 mg) were reacted according to general method A, resulting in compound 5b (40 mg, 35%) as a white powder. NMR data can be found in Supplementary Tables S1 and S2. MS (MALDI-TOF) m/z 1673.48 (M+Na+) (1673.49 calcd. for C81H84Cl2N10O24Na [M+Na+]). Anal. calcd. for C81H84Cl2N10O24 C 58.87, H 5.12, N 8.48. Found: C 58.83, H 5.15, N 8.46.
Compound 5c
Compound 4 (100 mg, 0.07 mmol) and compound 3c (17 mg) were reacted according to general method B. The reaction resulted in compound 5c (70 mg, 61%) as a white powder. NMR, MS and elemental analysis properties corresponded to the data reported for the compound previously.13
Compound 5d
Compound 4 (100 mg, 0.07 mmol) was reacted with compound 3d (14 mg) according to general method A, resulting in compound 5d (20 mg, 18%) as a white powder. NMR data can be found in Supplementary Tables S1 and S2. MS (MALDI-TOF) m/z 1617.43 (M+Na+) (1617.43 calcd. for C77H76Cl2N10O24Na [M+Na+]). Anal. calcd. for C77H76Cl2N10O24 C 57.93, H 4.80, N 8.77 Found: C 57.79, H 4.69, N 8.67.
Compound 5e
Compound 4 (100 mg, 0.07 mmol) was reacted with compound 3e (12 mg) according to general method A, resulting in compound 5e (31 mg, 28%) as a white powder. NMR data can be found in Supplementary Tables S1 and S2. MS (MALDI-TOF) m/z1589.5 (M+Na+) (1589.41 calcd. for C75H72Cl2N10O24Na [M+Na+]) Anal. calcd. for C75H72Cl2N10O24 C 57.44, H 4.63, N 8.93 Found: C 57.40, H 4.68, N 8.87.
Compound 5f
Compound 4 (100 mg, 0.07 mmol) and compound 3f (12.5 μl) were reacted according to general method A, yielding compound 5f (35 mg, 32%) as a white powder. NMR data can be found in Supplementary Tables S1 and S2. MS (MALDI-TOF) m/z 1581.34 (M+Na+) (1581.34 calcd. for C75H64Cl2N10O24Na [M+Na+]). Anal. calcd. for C75H64Cl2N10O24 C 57.73, H 4.13, N 8.98. Found: C 57.69, H 4.16, N 8.95.
Compound 5g
Azide–alkyne click reaction performed according to general method B using compound 4 (100 mg, 0.07 mmol) and compound 3g (19 mg) resulted in compound 5g (54 mg, 48%) in the form of a white powder. NMR data can be found in Supplementary Tables S1 and S2. MS (MALDI-TOF) m/z 1631.35 (M+Na+) (1631.35 calcd. for C79H66Cl2N10O24Na [M+Na+]). Anal. calcd. for C79H66Cl2N10O24 C 58.92, H 4.13, N 8.70. Found: C 58.89, H 4.17, N 8.65.
Compound 5h
Compound 4 (100 mg, 0.07 mmol) and compound 3h (32 mg) were reacted according to general method B, yielding compound 5h (60 mg, 50%) as a white powder. NMR data can be found in Supplementary Tables S1 and S2. MS (MALDI-TOF) m/z 1747.42 (M+Na+) (1747.42 calcd. for C81H78Cl2N10O24Na [M+Na+]). Anal. calcd. for C81H78Cl2N10O24 C 56.35, H 4.55, N 8.11. Found: C 56.29, H 4.58, N 8.07.
Compound 5i
Compound 4 (100 mg, 0.07 mmol) and compound 3i (80 mg) were reacted according to general method A, resulting in compound 5i (49 mg, 28%) as a yellow powder. NMR data can be found in Supplementary Tables S1 and S2. MS (MALDI-TOF) m/z 2461.70 (M+Na+) (2461.70 calcd. for C129H120Cl2N10O33SNa [M+Na+]). Anal. calcd. for C129H120Cl2N10O33S C 63.46, H 4.95, N 5.74, S 1.31. Found: C 63.43, H 4.97, N 5.70, S 1.29.
Compound 5j
Compound 4 (107 mg, 0.075 mmol) and compound 3j (65 mg) were reacted according to general method A, yielding compound 5j (46 mg, 29%) as a yellow powder. NMR data can be found in Supplementary Tables S1 and S2. MS (ESI-TOF) m/z 2127.76 (M+H+) (2127.75 calcd. for C114H117Cl2N10O27 [M+H+]).

Preparation of the lipophilic disaccharide 3i.

Lipophilic derivatization of teicoplanin pseudoaglycon by azide–alkyne click reaction.
References
Binda, E., Marinelli, F. & Marcone, G. L. Old and new glycopeptide antibiotics: action and resistance. Antibiotics 3, 572–594 (2014).
Van Bambeke, F., Lambert, D. M., Mingeot-Leclercq, M. & Tulkens, P. M. in Infectious Diseases 2nd edn (eds Cohen, J. & Powderly, W. G.) 1717–1720 (Mosby Publishing, London, UK, 2004).
Bager, F., Madsen, M., Christensen, J. & Aarestrup, F. M. Avoparcin used as a growth promoter is associated with the occurrence of vancomycin-resistant Enterococcus faecium on Danish poultry and pig farms. Prev. Vet. Med. 31, 95–112 (1997).
Levine, D. P. Vancomycin: a history. Clin. Infect. Dis. 42, S5–S12 (2006).
Trueba, F., Garrabe, E, Hadef, R., Fabre, R. & Cavallo, J. High prevalence of teicoplanin resistance among Staphylococcus epidermidis strains in a 5-year retrospective study. J. Clin. Microbiol. 44, 1922–1923 (2006).
Sujatha, S. & Praharaj, I. Glycopeptide resistance in Gram-positive cocci: a review. Interdiscip. Perspect. Infect. Dis. 2012, 781679 (2012).
Chen, A. Y., Zervos, M. J. & Vazquez, J. A. Dalbavancin: a novel antimicrobial. Int. J. Clin. Pract. 61, 853–863 (2007).
Damodaran, S. E. & Madhan, S. Telavancin: a novel lipoglycopeptide antibiotic. J. Pharmacol. Pharmacother. 2, 135–137 (2011).
Van Bambeke, F. Renaissance of antibiotics against difficult infections: focus on oritavancin and new ketolides and quinolones. Ann. Med. 46, 512–529 (2014).
Sztaricskai, F. et al. A new series of glycopeptide antibiotics incorporating a squaric acid moiety. J. Antibiot. 59, 564–582 (2006).
Sipos, A. et al. Synthesis of isoindole and benzoisoindole derivatives of teicoplanin pseudoaglycon with remarkable antibacterial and antiviral activities. Bioorg. Med. Chem. Lett. 22, 7092–7096 (2012).
Csávás, M. et al. Synthesis and antibacterial evaluation of some teicoplanin pseudoaglycon derivatives containing alkyl- and arylthiosubstituted maleimides. J. Antibiot. 68, 579–585 (2015).
Pintér, G. et al. A diazo transfer—click reaction route to new, lipophilic teicoplanin and ristocetin aglycon derivatives with high antibacterial and anti-influenza virus activity: an aggregation and receptor binding study. J. Med. Chem. 52, 6053–6061 (2009).
Norris, E. A. & Thalia, I. N. Mechanism of action of oritavancin and related glycopeptide antibiotics. FEMS Microbiol. Rev. 26, 511–532 (2003).
Csávás, M. et al. Rapid synthesis of self-assembling 1,2-thiomannobioside glycoconjugates as potential multivalent ligands of mannose-binding lectins. Tetrahedron Lett. 55, 6983–6986 (2014).
Sahoo, L., Singhamahapatra, A. & Loganathan, D. Diversity oriented synthesis of novel haloglycolipids potentially useful for crystallization of integral membrane proteins. Org. Biomol. Chem. 12, 2615–2625 (2014).
Batool, T et al. A convenient method for the synthesis of (prop-2-ynyloxy)benzene derivatives via reaction with propargyl bromide. PLoS ONE 9, e115457 (2014).
Casaschi, A., Grigg, R. & Sansano, J. M. Palladium catalysed tandem cyclisation–anion capture. Part 6: Synthesis of sugar, nucleoside, purine, benzodiazepinone and β-lactam analogues via capture of in situ generated vinylstannanes. Tetrahedron 56, 7553–7560 (2000).
Tanaka, S., Goi, T., Tanaka, K. & Fukase, K. Highly efficient α-sialylation by virtue of fixed dipole effects of N-phthalyl group: Application to continuous flow synthesis of α(2-3)- and α(2-6)-Neu5Ac-Gal motifs by microreactor. J. Carbohydr. Chem. 26, 369–394 (2007).
Bois, J. et al. Easy and selective method for the synthesis of various mono-O-functionalized calix[4]arenes: de-O-functionalization using TiCl4 . J. Org. Chem. 75, 7550–7558 (2010).
Meldal, M. & Tornøe, V. Cu-catalyzed azide-alkyne cycloaddition. Chem. Rev. 108, 2952–3015 (2008).
Clinical and Laboratory Standards Institute. Performance standards for antimicrobial susceptibility testing: 22nd informational supplement. CLSI document M100-S22, Vol. 32, 1–184 (Clinical and Laboratory Standards Institute, Wayne, PA, 2012).
Gonzalez, N. et al. Influence of the MBC/MIC ratio on the antibacterial activity of vancomycin versus linezolid against methicillin-resistant Staphylococcus aureus isolates in a pharmacodynamic model simulating serum and soft tissue interstitial fluid concentrations reported in diabetic patients in a pharmacodynamic model. J. Antimicrob. Chemother. 68, 2291–2295 (2013).
Odenholt, I., Löwdin, E. & Cars, O. In vitro studies of the pharmacodynamics of teicoplanin against Staphylococcus aureus Staphylococcus epidermidis and Enterococcus faecium. Clin. Microbiol. Infect. 9, 930–970 (2003).
Chen, L. et al. Vancomycin analogues active against vanA-resistant strains inhibit bacterial transglycosylase without binding substrate. Proc. Natl Acad. Sci. USA 100, 5658–5663 (2003).
Belley, A. et al. Oritavancin disrupts membrane integrity of Staphylococcus aureus and vancomycin-resistant enterococci to effect rapid bacterial killing. Antimicrob. Agents Chemother. 54, 5369–5371 (2010).
Allen, N. E., LeTourneau, D. L. & Hobbs, J. N. The role of hydrophobic side chains as determinants of antibacterial activity of semisynthetic glycopeptide antibiotics. J. Antibiot. 50, 677–684 (1997).
Vanderlinden, E. et al. Novel inhibitors of influenza virus fusion: structure-activity relationship and interaction with the viral hemagglutinin. J.Virol. 84, 4277–4288 (2010).
Acknowledgements
We thank the financial support from the Hungarian Research Fund (OTKA K 109208). This research was also supported by the European Union and the State of Hungary, co-financed by the European Social Fund in the framework of TÁMOP-4.2.2B-15/1/KONV-2015-0001.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on The Journal of Antibiotics website
Supplementary information
Rights and permissions
About this article
Cite this article
Szűcs, Z., Csávás, M., Rőth, E. et al. Synthesis and biological evaluation of lipophilic teicoplanin pseudoaglycon derivatives containing a substituted triazole function. J Antibiot 70, 152–157 (2017). https://doi.org/10.1038/ja.2016.80
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ja.2016.80
This article is cited by
-
Synthesis of vancomycin fluorescent probes that retain antimicrobial activity, identify Gram-positive bacteria, and detect Gram-negative outer membrane damage
Communications Biology (2023)
-
N-Terminal guanidine derivatives of teicoplanin antibiotics strongly active against glycopeptide resistant Enterococcus faecium
The Journal of Antibiotics (2020)
-
New semisynthetic teicoplanin derivatives have comparable in vitro activity to that of oritavancin against clinical isolates of VRE
The Journal of Antibiotics (2019)
-
Fluorescence assay to predict activity of the glycopeptide antibiotics
The Journal of Antibiotics (2019)
-
Lipophilic teicoplanin pseudoaglycon derivatives are active against vancomycin- and teicoplanin-resistant enterococci
The Journal of Antibiotics (2017)