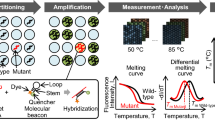
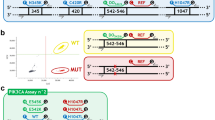
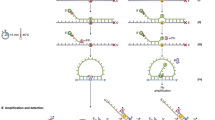
Abstract
Increased understanding of the molecular basis of colorectal cancer and recognition that extracellular DNA circulates in the plasma and serum of cancer patients enables new approaches to detection and monitoring. We used a polymerase chain reaction (PCR) assay to demonstrate mutant K-ras DNA in the plasma or serum of patients with colorectal cancer. Plasma or serum was fractionated from the blood of 31 patients with metastatic or unresected colorectal cancer and from 28 normal volunteers. DNA was extracted using either a sodium chloride or a gelatin precipitation method and then amplified in a two-stage PCR assay using selective restriction enzyme digestion to enrich for mutant K-ras DNA. Mutant K-ras DNA was detected in the plasma or serum of 12 (39%) patients, all confirmed by sequencing, but was not detected in any of the normal volunteers. K-ras mutations were detected in plasma or serum regardless of sex, primary tumour location, principal site of metastasis or proximity of chemotherapy and surgery to blood sampling. Tumour specimens available for 19 of the patients were additionally assayed for ras mutations and compared with blood specimens. Our results indicate mutant K-ras DNA is readily detectable by PCR in the plasma or serum of patients with advanced colorectal cancer. Thus, plasma- or serum-based nucleic acid amplification assays may provide a valuable method of monitoring and potentially detecting colorectal cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kopreski, M., Benko, F., Kwee, C. et al. Detection of mutant K-ras DNA in plasma or serum of patients with colorectal cancer. Br J Cancer 76, 1293–1299 (1997). https://doi.org/10.1038/bjc.1997.551
Issue Date:
DOI: https://doi.org/10.1038/bjc.1997.551
This article is cited by
-
Early detection: the impact of genomics
Virchows Archiv (2017)
-
Detection of MYCN amplification using blood plasma: noninvasive therapy evaluation and prediction of prognosis in neuroblastoma
Pediatric Surgery International (2013)
-
Comparative Screening of K-ras Mutations in Colorectal Cancer and Lung Cancer Patients Using a Novel Real-Time PCR with ADx-K-ras Kit and Sanger DNA Sequencing
Cell Biochemistry and Biophysics (2012)
-
Circulating tumour markers can define patients with normal colons, benign polyps, and cancers
British Journal of Cancer (2011)
-
Cell-free nucleic acids as biomarkers in cancer patients
Nature Reviews Cancer (2011)