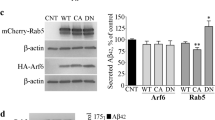
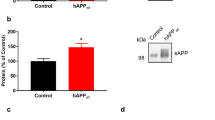
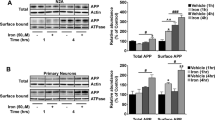
Abstract
Amyloid-β is a neurotoxic peptide which is implicated in the pathogenesis of Alzheimer's disease. It binds an intracellular polypeptide known as ERAB, thought to be a hydroxysteroid dehydrogenase enzyme, which is expressed in normal tissues, but is overexpressed in neurons affected in Alzheimer's disease. ERAB immunoprecipitates with amyloid-β, and when cell cultures are exposed to amyloid-β, ERAB inside the cell is rapidly redistributed to the plasma membrane. The toxic effect of amyloid-β on these cells is prevented by blocking ERAB and is enhanced by overexpression of ERAB. By interacting with intracellular amyloid-β, ERAB may therefore contribute to the neuronal dysfunction associated with Alzheimer's disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Haass, C. & Selkoe, D. Cellular procesisng of β-amyloid precursor protein and the genesis of amyloid β-peptide. Cell 75, 1039–1042 (1993).
Yankner, B. et al. Neurotoxicity of a fragment of the amyloid precursor associated with Alzheimer's disease. Science 245, 417–420 (1989).
Scheuner, D. et al. Secreted amyloid β-protein similar to that in the senile plaques of Alzheimer's disease is increased in vivo by the presenilin 1 and 2 and APP mutations linked to familial Alzheimer's disease. Nature Med. 2, 864–869 (1996).
Roses, A. From genes to mechanisms to therapies: lessons to be learned for neurological disorders. Nature Med. 2, 267–269 (1996).
Cai, X., Golde, T. & Younkin, S. Release of excess amyloid β rotein from a mutant amyloid β protein precursor. Science 259, 514–516 (1993).
Hensley, K. et al. Amodel for β-amyloid aggregation and neurotoxicity based on free radical generation by the peptide: relevance to Alzheimer disease. Proc. Natl Acad. Sci. USA 91, 3270–3274 (1994).
Yankner, B., Duffy, L. & Kirschner, D. Neurotrophic and neurotoxic effects of amyloid β protein: reversal by tachykinin neuropeptides. Science 250, 279–282 (1990).
Benzi, G. & Moretti, A. Are reactive oxygen species involved in Alzheimer's disease? Neurobiol. Aging 16, 661–674 (1995).
Cotman, C. & Anderson, A. Apotential role for apoptosis in neurodegeneration and Alzheimer's disease. Mol. Neurobiol. 10, 19–45 (1995).
Yan, S.-D. et al. RAGE in Alzheimer's disease: a receptor mediating amyloid-β peptide-induced oxidant stress and neurotoxicity, and microglial activation. Nature 382, 685–691 (1996).
Paresce, D., Ghosh, R. & Maxfirled, F. Microglial cells internalize aggregates of the Alzheimer's disease amyloid β-protein via a scavenger receptor. Neuron 17, 553–565 (1996).
El-Khoury, J. et al. Scavenger receptor-mediated adhesion of microglia to β-amyloid fibrils. Nature 382, 685–691 (1996).
Wild-Bode, C. et al. Intracellular generation and accumulation of Aβ terminating at amino acid 42. J. Biol. Chem. 272, 16085–16088 (1997).
Xia, W. et al. Enhanced production and oligomerization of the 42-residue amyloid β-protein by chinese hamster ovary cells stably expressing mutant presenilins. J. Biol. Chem. 272, 7977–7982 (1997).
Xu, H. et al. Generation of Alzheimer β-amyloid protein in the trans -Golgi network in the apparent absence of vesicle formation. Proc. Natl Acad. Sci. USA 94, 3748–3752 (1997).
Tiernari, P. et al. Intracellular and secreted Alzheimer β-amyloid species are generated by distinct mechanisms in cultured hippocampal neurons. Proc. Natl Acad. Sci. USA 94, 4125–4130 (1997).
Yoshikawa, K., Aizawa, T. & Hayashi, Y. Degeneration in vitro of post-mitotic neurons overexpressing the Alzheimer amyloid protein precursor. Nature 359, 64–67 (1992).
Fuller, S. et al. Intracellular produciton of βA4 amyloid of Alzheimer's disease: modulation by phosphoramidon and lack of coupling to the secretion of the amyloid precursor protein. Biochemistry 34, 8091–8098 (1995).
Frackowiak, J. et al. Secretion and accumulation of Alzheimer's β-protein by cultured vascular smooth muscle cells from old and young dogs. Brain Res. 676, 225–230 (1995).
Fields, S. & Song, O.-K. Anovel genetic system ot detect protein-protein interactions. Nature 340, 245–246 (1991).
Kozak, M. An analysis of 5′-noncoding sequences from 699 vertebrate messenger RNAs. Nucleic Acids Res. 15, 8125–8131 (1987).
Bernstein, F. et al. The Protein Data Bank: a computer-based archival file for macromolecular structures. J. Mol. Biol. 112, 535–542 (1977).
Devereux, J., Haeberli, P. & Smithies, O. Acomprehensive set of sequence analysis programs for the VAX. Nucleic Acids Res. 12, 387–395 (1984).
Ghosh, D. et al. Three-dimensional structure of holo 3α,20β-hydroxysteroid dehydrogenase: a member of a short-chain dehydrogenase family. Proc. Natl Acad. Sci. USA 88, 10064–10068 (1991).
Schembri, M., Bayly, R. & Davies, J. Phosphate concentration regulates transcription of acinetobacter polyhydroxyalkanoic acid biosynthetic genes. J. Bacteriol. 177, 4501–4507 (1995).
Behl, C., Davis, J., Lesley, R. & Schubert, D. Hydrogen peroxide mediates amyloid β protein toxicity. Cell 77, 817–827 (1994).
Busciglio, J. & Yankner, B. Apoptosis and increased generation of reactive oxygen species in Down's syndrome neurons in vitro . Nature 378, 776–779 (1995).
Vitek, M. et al. Advanced glycation endproducts contribute to amyloidosis in Alzheimer disease. Proc. Natl Acad. Sci. USA 91, 4766–4770 (1994).
Yan, S.-D. et al. Glycated tau protein in Alzheimer disease: a mechanism for induction of oxidant stress. Proc. Natl Acad. Sci. USA 91, 7787–7791 (1994).
Ledesma, M., Bonay, P., Colaco, C. & Avila, J. Analysis of microtubule-associated protein tau glycation in paired helical filaments. J. Biol. Chem. 269, 21614–2169 (1994).
Sherrington, R. et al. Cloning of a gene bearing missense mutations in early-onset familial Alzheimer's disease. Nature 375, 754–760 (1993).
Citron, M. et al. Mutation of the β-amyloid precursor protein in familial Alzheimer's disease increases β-protein production. Nature Med. 3, 67–72 (1997).
Levy-Lahad, E. et al. Afamilial Alzheimer's disease locus on chromosome 1. Science 269, 970–972 (1995).
Kovacs, D. et al. Alzheimer-associated presenilins 1 and 2: neuronal expression in brain and localization ot intracellular membranes in mammalian cells. Nature Med. 2, 224–229 (1996).
Duff, K. et al. Increased amyloid-β42(43) in brains of mice expressing mutant presenilin 1. Nature 383, 710–713 (1996).
Borchelt, D. et al. Familial Alzheimer's disease-linked presenilin 1 variants elevate Aβ1-42/1-40 ratio in vitro and in vivo . Neuron 17, 1005–1013 (1996).
Scheuner, D. et al. Secreted amyloid β-protein similar to that in the senile plaques of Alzheimer's disease is increased in vivo by the presenilin 1 and 2 and APP mutations linked to familial Alzheimer's disease. Nature Med. 2, 864–869 (1996).
Wolozin, B. et al. Participation of presenilin 2 in apoptosis: enhanced basal activity conferred by an Alzheimer mutaiton. Science 274, 1710–1713 (1996).
Wong, P. et al. Presenilin 1 required for Notch 1 and DII1 expression in the paraxial mesoderm. Nature 387, 288–292 (1997).
Shen, J. et al. Skeletal and CNS defects in presenilin-1-deficient mice. Cell 89, 629–639 (1997).
Knauer, M. et al. Intracellular accumulation and resistance to degradation of the Alzheimer amyloid A4/β protein. Proc. Natl Acad. Sci. USA 89, 7437–7441 (1992).
Turner, R. S. et al. Amyloid β40and β42are generated intracellularly in cultured human neurons and their secretion increases with maturation. J. Biol. Chem. 271, 8966–8970 (1996).
Fuller, S. et al. Intracellular production of βA4 amyloid of Alzheimer's disease: modulation by phosphoramidon and lack of coupling to the secretion of the amyloid precursor protein. Biochem. 34, 8091–8098 (1995).
Yang, A., Knauer, M., Burdick, D. & Glabe, C. Intracellular Aβ1–42 aggregates stimulate the accumulation of stable, insoluble amyloidogenic fragments of the amyloid precursor protein in transfected cells. J. Biol. Chem. 270, 14786–14792 (1995).
La Feria, F. et al. Neuronal cell death in Alzheimer's disease correlates with apoE uptake and intracellular Aβ stabilization. J. Clin. Invest. 100, 310–320 (1997).
Urmoneit, B. et al. Cerebrovascular smooth muscle cells internalize Alzheimer amyloid beta protein via a protein pathway: implications for cerebral amyloid angiopathy. Lab. Invest. 77, 157–166 (1997).
Matsubara, E., Soto, C., Governale, S., Frangione, B. & Ghiso, J. ApoJ and Alzheimer's amyloid-β solubility. Biochem. J. 316, 671–679 (1996).
Kuwabara, K. et al. Purification and characterization of a novel stress protein, ORP150, from cultured rat astrocytes and its expression in ischemic mouse brain. J. Biol. Chem. 271, 5025–5032 (1996).
Saido, T. et al. Amino- and carboxyl-terminal heterogeneity of β-amyloid peptides deposited in human brain. Neurosci. Lett. 215, 173–176 (1996).
Yan, S.-D. et al. Nonenzymatically glycated tau in Alzheimer's disease induces neuronal oxidant stress resulting in cytokine gene expression and release of amyloid-β peptide. Nature Med. 1, 693–699 (1995).
Acknowledgements
We thank G. C. Godman and G. Andres for help and advice during these studies and in preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Du Yan, S., Fu, J., Soto, C. et al. An intracellular protein that binds amyloid-β peptide and mediates neurotoxicity in Alzheimer's disease. Nature 389, 689–695 (1997). https://doi.org/10.1038/39522
Issue Date:
DOI: https://doi.org/10.1038/39522
This article is cited by
-
Role of SIRT3 in neurological diseases and rehabilitation training
Metabolic Brain Disease (2023)
-
Soluble and insoluble protein aggregates, endoplasmic reticulum stress, and vascular dysfunction in Alzheimer’s disease and cardiovascular diseases
GeroScience (2023)
-
Deacetylation of HSD17B10 by SIRT3 regulates cell growth and cell resistance under oxidative and starvation stresses
Cell Death & Disease (2020)
-
Interactions of 17β-Hydroxysteroid Dehydrogenase Type 10 and Cyclophilin D in Alzheimer's Disease
Neurochemical Research (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.