Abstract
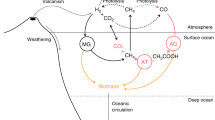
The advent of oxygenic photosynthesis on Earth may have increased global biological productivity by a factor of 100–1,000 (ref. 1), profoundly affecting both geochemical and biological evolution. Much of this new productivity probably occurred in microbial mats, which incorporate a range of photosynthetic and anaerobic microorganisms in extremely close physical proximity2,3. The potential contribution of these systems to global biogeochemical change would have depended on the nature of the interactions among these mat microorganisms. Here we report that in modern, cyanobacteria-dominated mats from hypersaline environments in Guerrero Negro, Mexico, photosynthetic microorganisms generate H2 and CO—gases that provide a basis for direct chemical interactions with neighbouring chemotrophic and heterotrophic microbes4. We also observe an unexpected flux of CH4, which is probably related to H2-based alteration of the redox potential within the mats. These fluxes would have been most important during the nearly 2-billion-year period during which photosynthetic mats contributed substantially to biological productivity5—and hence, to biogeochemistry—on Earth. In particular, the large fluxes of H2 that we observe could, with subsequent escape to space, represent a potentially important mechanism for oxidation of the primitive oceans and atmosphere.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
DesMarais, D. J. in Geomicrobiology (eds Banfield, J. & Nealson, K.) 429–445 (Mineralogical Society of America, Washington DC, 1997).
Cohen, Y. et al. Ancient Stromatolites and Microbial Mats (Liss, New York, 1984).
Stal, L. J. & Caumette, P. Microbial Mats: Structure, Development and Environmental Significance (Springer, Berlin, 1994).
Conrad, R. Biogeochemistry and ecophysiology of atmospheric CO and H2. Adv. Microb. Ecol. 10, 231–283 (1988).
Walter, M. R. (ed.) Stromatolites (Elsevier, Amsterdam, 1976).
DesMarais, D. J. in Advances in Microbial Ecology (ed. Jones, J. G.) 251–274 (Plenum, New York, 1995).
Canfield, D. E. & DesMarais, D. J. Biogeochemical cycles of carbon, sulfur, and free oxygen in a microbial mat. Geochim. Cosmochim. Acta 57, 3971–3984 (1993).
Bebout, B. M., Paerl, H. W., Bauer, J. M., Canfield, D. E. & DesMarais, D. J. in Microbial Mats: Structure, Development, and Environmental Significance (eds Stal, L. J. & Caumette, P.) 265–271 (Springer, Berlin, 1994).
Newton, W., Postgate, J. R. & Rodriguez-Barrueco, C. Recent Developments in Nitrogen Fixation (Academic, London, 1977).
Stal, L. J., Heyer, H., Bekker, S., Villbrandt, M. & Krumbein, W. E. in Microbial Mats: Physiological Ecology of Benthic Microbial Communities (eds Cohen, Y. & Rosenberg, E.) 255–276 (American Society for Microbiology, Washington DC, 1989).
Fay, P. Oxygen relations of nitrogen fixation in cyanobacteria. Microbiol. Rev. 56, 340–373 (1992).
Stal, L. J. & Moezelaar, R. Fermentation in cyanobacteria. FEMS Microbiol. Rev. 21, 179–211 (1997).
Lovley, D. R. & Klug, M. J. Sulfate reducers can outcompete methanogens at freshwater sulfate concentrations. Appl. Environ. Microbiol. 45, 187–192 (1983).
Iannotti, E. L., Kafkewitz, D., Wolin, M. J. & Bryant, M. P. Glucose fermentation products of Ruminococcus albus grown in continuous culture with Vibrio succinogenes: Changes caused by interspecies transfer of H2. J. Bacteriol. 114, 1231–1240 (1973).
Wolin, M. J. & Miller, T. L. Interspecies hydrogen transfer: 15 years later. ASM News 48, 561–565 (1982).
Dolfing, J. in Biology of Anaerobic Microorganisms (ed. Zehnder, A. J. B.) 417–468 (Wiley-Interscience, New York, 1988).
Schink, B. in Biology of Anaerobic Microorganisms (ed. Zehnder, A. J. B.) 771–846 (Wiley-Interscience, New York, 1988).
Skyring, G. W., Lynch, R. M. & Smith, G. D. in Microbial Mats: Physiological Ecology of Benthic Microbial Communities (eds Cohen, Y. & Rosenberg, E.) 170–179 (American Society for Microbiology, Washington DC, 1989).
Fenchel, T. & Finlay, B. J. Ecology and Evolution in Anoxic Worlds (Oxford Univ. Press, Oxford, 1995).
Nisbet, E. G. & Fowler, C. M. R. Archaean metabolic evolution of microbial mats. Proc. R. Soc. Lond. B 266, 2375–2382 (1999).
Holland, H. D. Chemical Evolution of the Atmosphere and Oceans (Princeton Univ. Press, Princeton, 1984).
Berner, R. A. & Canfield, D. E. A new model for atmospheric oxygen over Phanerozoic time. Am. J. Sci. 289, 333–361 (1989).
DesMarais, D. J., Strauss, H., Summons, R. E. & Hayes, J. M. Carbon isotope evidence for the stepwise oxidation of the Proterozoic environment. Nature 359, 605–609 (1992).
Walker, J. C. G. Evolution of the Atmosphere (Macmillan, New York, 1977).
Hoehler, T. M., Alperin, M. J., Albert, D. B. & Martens, C. S. Field and laboratory studies of methane oxidation in an anoxic marine sediment: Evidence for a methanogen-sulfate reducer consortium. Glob. Biogeochem. Cycles 8, 451–463 (1994).
Elderfield, H. & Schulz, A. Mid-ocean ridge hydrothermal fluxes and the chemical composition of the ocean. Annu. Rev. Earth Planet. Sci. 24, 191–224 (1996).
Field, C. B., Behrenfeld, M. J., Randerson, J. T. & Falkowski, P. Primary production of the biosphere: Integrating terrestrial and oceanic components. Science 281, 237–240 (1998).
Acknowledgements
We thank Exportadora de Sal, S.A. de C.V. for access to their salt ponds and for logistical support, and S. Miller, P. Visscher and L. Jahnke for discussions. This work was supported by NASA's Astrobiology Institute and Exobiology Program, and by Ames Research Center Director's Discretionary Funds. T.M.H. was supported by a National Research Council fellowship.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hoehler, T., Bebout, B. & Des Marais, D. The role of microbial mats in the production of reduced gases on the early Earth. Nature 412, 324–327 (2001). https://doi.org/10.1038/35085554
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/35085554
This article is cited by
-
Millimeter-scale vertical partitioning of nitrogen cycling in hypersaline mats reveals prominence of genes encoding multi-heme and prismane proteins
The ISME Journal (2022)
-
Gradients and consequences of heterogeneity in biofilms
Nature Reviews Microbiology (2022)
-
Bioremediation perspectives and progress in petroleum pollution in the marine environment: a review
Environmental Science and Pollution Research (2021)
-
Efficient recycling of nutrients in modern and past hypersaline environments
Scientific Reports (2019)
-
Mesophilic microorganisms build terrestrial mats analogous to Precambrian microbial jungles
Nature Communications (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.