Abstract
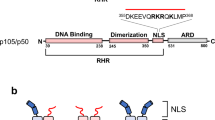
The 2.3-Â crystal structure of the transcription factor NF-κB p50 homodimer bound to a palindromic κB site reveals that the Rel homology region folds into two distinct domains, similar to those in the immunoglobulin superfamily. The p50 dimer envelopes an undistorted B-DNA helix, making specific contacts along the 10-base-pair κB recognition site mainly through loops connecting secondary structure elements in both domains. The carboxy-terminal domains form a dimerization interface between β-sheets using residues that are strongly conserved in the Rel family.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Sen, R. & Baltimore, D. Cell 47, 921–928 (1986).
Blank, V., Kourilsky, P. & Israel, A. Trends Biochem. Sci. 17, 135–140 (1990).
Baeuerle, P. & Henkel, T. A. Rev. Immun. 12, 141–179 (1994).
Grilli, M., Chiu, J.-S. & Lenardo, M. Int. Rev. Cytol. 143, 1–62 (1993).
Liou, H.-C. & Baltimore, D. Curr. Opin. Cell Biol. 5, 477–487 (1993).
Kopp, E. & Ghosh, S. Adv. Immun. 58, 1–27 (1994).
Ghosh, S. et al. Cell 62, 1019–1029 (1990).
Kieran, M. et al. Cell 62, 1007–1018 (1990).
Nolan, G., Ghosh, S., Liou, H.-C., Tempst, P. & Baltimore, D. Cell 64, 961–969 (1991).
Ruben, S. et al. Science 251, 1490–1493 (1991).
Schmid, R. M., Parkins, N. D., Duckett, C. S. & Nabel, G. J. Nature 352, 733–736 (1992).
Ryseck, R.-P. Molec. cell. Biol. 12, 674–684 (1992).
Tony, Y. et al. Cell 75, 753–763 (1993).
Baeuerle, P. A. & Baltimore, D. Science 242, 541–546 (1988).
Zabel, U. & Baeuerle, P. Cell 61, 255–256 (1990).
Palombella, V. J., Rando, O., Goldberg, A. & Maniatis, T. Cell 78, 773–785 (1994).
Ghosh, S. & Baltimore, D. Nature 344, 678–682 (1990).
Luthy, R., Bowie, J. & Einsenberg, D. Nature 356, 83–85 (1992).
Pabo, C. O. & Sauer, R. A. Rev. Biochem. 61, 1053–1095 (1992).
Yunje, C., Gorina, S., Jeffrey, P. & Pavletich, N. Science 265, 346–355 (1994).
Kunsch, C., Ruben, S. M. & Rosen, C. A. Molec. cell. Biol. 12, 4412–4421 (1992).
Winkler, F. K. et al. EMBO. J. 12, 1781–1795 (1993).
Bressler, P. et al. J. Virol. 67, 288–293 (1993).
Kumar, S., Rabson, A. & Gelinus, C. Molec. cell. Biol. 12, 3094–3106 (1992).
Liu, J., Fan, Q. R., Sodeoka, M., Lane, W. S. & Verdine, G. L. Curr. Biol. 1, 47–55 (1994).
Liu, J., Sodeoka, M., Lane, W. S. & Verdine, G. L. Proc. natn. Acad. Sci. U.S.A. 91, 908–912 (1994).
Tolenado, M. B., Ghosh, D., Trinh, F. & Leonard, W. J. Molec. cell. Biol. 13, 852–860 (1993).
Brownwell, E., Mittereder, N. & Rice, N. Oncogene 4, 935–942 (1989).
Northrop, J. P. et al. Nature 369, 497–502 (1994).
Kumar, S. & Gelinus, C. Proc. natn. Acad. Sci. U.S.A. 90, 8962–8966 (1993).
Dimitris, T. & Maniatis, T. Cell 71, 777–789 (1992).
Harpaz, Y. & Chothia, C. J. molec. Biol. 238, 528–539 (1994).
Van Roey, P. & Beerman, T. Proc. natn. Acad. Sci. U.S.A. 86, 6587–6591 (1989).
DeVos, A. M., Ultsch, M. & Kossiakoff, A. A. Science 255, 306–312 (1992).
Haltmark, D. Nature 367, 116–117 (1993).
Jones, T. A., Zou, J. Y., Cowan, S. W. & Kjeldgaard, M. Acta crystallogr. A47, 110–119 (1991).
Brünger, A. T. XPLOR version 3.1: a system for X-ray crystallography and NMR (Yale University Press, New Haven, 1993).
Read, R. J. Acta crystallogr. A42, 140–149 (1986).
Müller, C. W., Rey, F. A., Sodeoka, M., Verdine, G. L. & Harrison, S. C. Nature 373, 311–317 (1995).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ghosh, G., Duyne, G., Ghosh, S. et al. Structure of NF-κB p50 homodimer bound to a κB site. Nature 373, 303–310 (1995). https://doi.org/10.1038/373303a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/373303a0
This article is cited by
-
Exploration of the mechanism by which Huangqi Guizhi Wuwu decoction inhibits Lps-induced inflammation by regulating macrophage polarization based on network pharmacology
BMC Complementary Medicine and Therapies (2023)
-
NF-κB signaling in neoplastic transition from epithelial to mesenchymal phenotype
Cell Communication and Signaling (2023)
-
Backbone and side-chain chemical shift assignments of p50 subunit of NF-κB transcription factor
Biomolecular NMR Assignments (2021)
-
Backbone resonance assignments of the dimeric domain of the p50 NF-kappaB subunit
Biomolecular NMR Assignments (2020)
-
Insights into the assembly and architecture of a Staufen-mediated mRNA decay (SMD)-competent mRNP
Nature Communications (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.