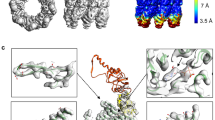
Abstract
The crystal structure of Escherichia coli GroEL shows a porous cylinder of 14 subunits made of two nearly 7-fold rotationally symmetrical rings stacked back-to-back with dyad symmetry. The subunits consist of three domains: a large equatorial domain that forms the foundation of the assembly at its waist and holds the rings together; a large loosely structured apical domain that forms the ends of the cylinder; and a small slender intermediate domain that connects the two, creating side windows. The three-dimensional structure places most of the mutation-ally defined functional sites on the channel walls and its outward imaginations, and at the ends of the cylinder.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Ellis, R. J. & van der Vies, S. M. A. Rev. Biochem. 60, 321–347 (1991).
Gething, M.-J. & Sambrook, J. Nature 355, 33–45 (1992).
Hendrick, J. P. & Hartl, F. U. A. Rev. Biochem. 62, 349–384 (1993).
Horwich, A. L. & Willison, K. R. Phil. Trans. R. Soc., Lond. B339, 313–326 (1993).
Hendrix, R. W. J. molec. Biol. 129, 375–392 (1979).
McMullen, T. W. & Hallberg, R. L. Molec. cell. Biol. 7, 4414–4423 (1987).
Pushkin, A. V. et al. Biochim. biophys. Acta 704, 379–384 (1982).
Trent, J. D., Nimmesgern, E., Wall, J. S., Hartl, F. U. & Horwich, A. L. Nature 354, 490–493 (1991).
Gao, Y., Thomas, J. O., Chow, R. L., Lee, G. H. & Cowan, N. J. Cell 69, 1043–1050 (1992).
Fayet, O., Ziegelhoffer, T. & Georgopoulos, C. J. Bact. 171, 1379–1385 (1989).
Cheng, M. Y. et al. Nature 337, 620–625 (1989).
Horwich, A. L., Low, K. B., Fenton, W. A., Hirshfield, I. N. & Furtak, K. Cell 74, 909–917 (1993).
Ursic, D. & Culbertson, M. R. Molec. cell. Biol. 11, 2629–2640 (1991).
Goloubinoff, P., Christeller, J. T., Gatenby, A. A. & Lorimer, G. H. Nature 342, 884–889 (1989).
Martin, J. et al. Nature 352, 36–42 (1991).
Mendoza, J. A., Lorimer, G. H. & Horowitz, P. M. J. biol. Chem. 266, 16973–16976 (1991).
Bochkareva, E. S., Lissin, N. M., Flynn, G. C., Rothman, J. E. & Girschovich, A. S. J. biol. Chem. 267, 6796–6800 (1992).
Landry, S. J., Jordan, R., McMacken, R. & Gierasch, L. M. Nature 355, 455–457 (1992).
Zahn, R., Spitzfaden, C., Ottiger, M., Wuthrich, K. & Pluckthun, A. Nature 368, 261–265 (1994).
Okazaki, A., Ikura, T., Nikaido, K. & Kuwajima, K. Nature struct. Biol. 1, 439–446 (1994).
Hayer-Hartl, M. K., Ewbank, J. J., Creighton, T. E. & Hartl, F. U. EMB0 J. 13, 3192–3202 (1994).
Langer, T., Pfeifer, G., Martin, J., Baumeister, W. & Hartl, F. U. EMB0 J. 11, 4757–4765 (1992).
Braig, K., Simon, M., Furuya, F., Hainfeld, J. F. & Horwich, A. L. Proc. natn. Acad. Sci. U.S.A. 90, 3978–3982 (1993).
Saibil, H. R. et al. Curr. Biol. 3, 265–273 (1993).
Ishii, N., Taguchi, H., Sasabe, H. & Yoshida, M. J. molec. Biol. 236, 691–696 (1994).
Gray, T. E. & Fersht, A. R. FEBS Lett. 292, 254–258 (1992).
Jackson, G. S. et al. Biochemistry 32, 2554–2563 (1993).
Todd, M., Viitanen, P. V. & Lorimer, G. H. Biochemistry 32, 8560–8567 (1993).
Martin, J., Mayhew, M., Langer, T. & Hartl, F. U. Nature 366, 228–233 (1993).
Schmidt, M., Buchner, J., Todd, M. J., Lorimer, G. H. & Viitanen, P. V. J. biol. Chem. 269, 10304–10311 (1994).
Azem, A., Kessel, M. & Goloubinoff, P. Science 265, 653–656 (1994).
Schmidt, M. et al. Science 265, 656–659 (1994).
Todd, M., Viitanen, P. V. & Lorimer, G. H. Science 265, 659–666 (1994).
Fitzgerald, P. M. D. J. appl. Crystallogr. 21, 273–278 (1988).
Otwinowski, Z. in Proc. CCP4 Study Weekend, 25–26 Jan 1991, Isomorphous Replacement and Anomalous Scattering (eds Wolf, W., Evans, P. R. & Leslie, A. G. W.) 80–86 (SERC Daresbury Laboratory, UK, 1991).
Jones, T. A. Proc. CCP4 Study Weekend, Molecular Replacement (eds Dodson, E. J., Glover, S. & Wolf, W. 91–105 (SERC Daresbury Laboratory, UK, 1992).
Jones, T. A. et al. Acta crystallogr. A47, 110–119 (1991).
Fenton, W. A., Kashi, Y., Furtak, K. & Horwich, A. L. Nature 371, 614–619 (1994).
Brunger, A. T. XPLOR Version 3.1 Manual (1993).
Read, R. Acta crystallogr. A42 140–149 (1986).
Luthy, R., Bowie, J. U. & Eisenberg, D. Nature 356, 83–85 (1992).
Lee, B. K. & Richards, F. M. J. molec. Biol. 55, 379–400 (1971).
Janin, J. & Chothia, C. J. biol. Chem. 265, 16027–16030 (1990).
Janin, J., Miller, S. & Chothia, C. J. molec. Biol. 204, 155–164 (1988).
Miller, S. Prot. Engng 3, 77–83 (1989).
Ellis, R. J. Nature 366, 213–214 (1993).
Weissman, J. S., Kashi, Y., Fenton, W. A., & Horwich, A. L. Cell 78, 693–702 (1994).
McLennan, N. F., Girshovich, A. S., Lissin, N. M., Charters, Y. & Masters, M. Molec. Microbiol. 7, 49–58 (1993).
Viitanen, P. V. et al. J. biol. Chem. 267, 695–698 (1992).
Otwinowski, Z. in Proc. CCP4 Study Weekend, 29–30 Jan 1991, Data Collection and Processing (eds Sawyer, L., Isaacs, N. & Burley, S.) 56–62 (SERC Daresbury Laboratory, UK, 1993).
Svensson, L. A., Surin, B. P., Dixon, N. E. & Spangfort, M. D. J. molec. Biol. 235, 47–52 (1994).
Burnett, B., Horwich, A. L. & Low, K. B. J. Bact. (in the press).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Braig, K., Otwinowski, Z., Hegde, R. et al. The crystal structure of the bacterial chaperonln GroEL at 2.8 Å. Nature 371, 578–586 (1994). https://doi.org/10.1038/371578a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/371578a0
This article is cited by
-
Facile hermetic TEM grid preparation for molecular imaging of hydrated biological samples at room temperature
Nature Communications (2023)
-
Three-dimensional structure determination of protein complexes using matrix-landing mass spectrometry
Nature Communications (2022)
-
Crystal structure of P. falciparum Cpn60 bound to ATP reveals an open dynamic conformation before substrate binding
Scientific Reports (2021)
-
A Review: Molecular Chaperone-mediated Folding, Unfolding and Disaggregation of Expressed Recombinant Proteins
Cell Biochemistry and Biophysics (2021)
-
Molecular chaperones and their denaturing effect on client proteins
Journal of Biomolecular NMR (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.