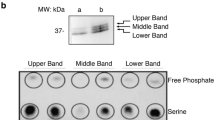
Abstract
ACTIVATION of receptor-linked and cytoplasmic protein tyrosine kinases is crucial in the control of normal and abnormal cell growth and differentiation1,2. Some substrates of protein tyrosine kinases such as phospholipase Cγ and ras GTPase-activating protein (GAP) contain sequences homologous to the src protein domains SH2 and SH3 (refs 3–9). The proto-oncogene vav is expressed in haematopoietic cells and its product Vav contains sequence motifs commonly found in transcription factors, such as helix–loop–helix, leucine-zipper and zinc-finger motifs and nuclear localization signals10–12, as well as a single SH2 and two SH3 domains. Here we show that stimulation of T-cell antigen receptor on normal human peripheral blood lymphocytes or on human leukaemic T cells, and the crosslinking of IgE receptors on rat basophilic leukaemia cells, both promote the phosphorylation of tyrosine residues in Vav. Moreover, activation of the receptor for epidermal growth factor leads to marked tyrosine phosphorylation of Vav in cells transiently expressing vav, and Vav associates with the receptor through its SH2 domain. We propose that vav encodes a new class of substrates whose tyrosine phosphorylation may provide a mechanism for direct signal transduction linking receptors at the cell surface to transcriptional control.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Ullrich, A. & Schlessinger, J. Cell 61, 203–211 (1990).
Cantley, L. C. et al. Cell 64, 281–302 (1991).
Ellis, C., Moran, M., McCormick, F. & Pawson, T. Nature 343, 377–381 (1990).
Kaplan, D. R., Morrison, D. K., Wong, G., McCormick, F. & Williams, L. T. Cell 61, 121–133 (1990).
Margolis, B. et al. Cell 57, 1101–1107 (1989).
Meisenhelder, J., Suh, P.-G., Rhee, S. G. & Hunter, T. Cell 57, 1109–1122 (1989).
Molloy, C. J. et al. Nature 342, 711–714 (1989).
Wahl, M. I., Nishibe, S., Suh, P.-G., Rhee, S. G. & Carpenter, G. Proc. natn. Acad. Sci. U.S.A. 86, 1568–1572 (1989).
Koch, C. A., Anderson, D., Moran, M. F., Ellis, C. & Pawson, T. Science 252, 668–674 (1991).
Katzav, S., Martin-Zanca, D. & Barbacid, M. EMBO J. 8, 2283–2290 (1989).
Katzav, S., Cleveland, J. L., Heslop, H. E. & Pulido, D. Molec. cell. Biol. 11, 1912–1920 (1991).
Coppola, J., Bryant, S., Koda, T., Conway, D. & Barbacid, M. Cell Growth Differ. 2, 95–105 (1991).
Morgan, C., Pollard, J. W. & Stanley, E. R. J. Cell Physiol. 130, 420–427 (1987).
Klausner, R. D. & Samelson, L. E. Cell 64, 875–878 (1991).
Park, D. J., Rho, H. W. & Rhee, S. G. Proc. natn. Acad. Sci. U.S.A. 88, 5453–5456 (1991).
Weiss, A., Koretzky, G., Schatzman, R. & Kadlecek, T. Proc. natn. Acad. Sci. U.S.A. 88, 5484–5488 (1991).
Metzger, H. et al. A. Rev. Immun. 4, 419–470 (1986).
Oliver, J. M., Seagrave, J. C., Stump, R. F., Pfeiffer, J. R. & Geanin, G. Prog. Allergy 42, 195–245 (1988).
Benhamou, M., Gutkind, J. S., Robbins, K. C. & Siraganian, R. P. Proc. natn. Acad. Sci. U.S.A. 87, 5327–5330 (1990).
Connelly, P. A., Farrell, C. A., Merenda, J. M., Conklyn, M. J. & Showell, H. J. Biochem. biophys. Res. Commun. 177, 192–201 (1991).
Bolen, J. Cell Growth Differ. 2, 365–414 (1991).
Anderson, D. et al. Science 250, 979–982 (1990).
Moran, M. F. et al. Proc. natn. Acad. Sci. U.S.A. 87, 8622–8626 (1990).
Margolis, B. et al. EMBO J. 9, 4375–4380 (1990).
Bustelo, X. R., Ledbetter, J. A. & Barbacid, M. Nature 356, 68–71 (1992).
Pain, B. et al. Cell 65, 37–46 (1991).
Blackwood, E. M. & Eisenman, R. N. Science 251, 1211–1217 (1991).
Prendergast, G. C., Lawe, D. & Ziff, E. Cell 65, 395–407 (1991).
Honegger, A. M. et al. Cell 51, 199–209 (1987).
Matsushime, H., Roussel, M. F., Ashmun, R. A. & Sherr, C. J. Cell 65, 701–713 (1981).
Cooper, J. A., Sefton, B. M. & Hunter, T. Meth. Enzym. 99, 387–402 (1983).
Hara, T. & Fu, S. M. J. exp. Med. 161, 641–656 (1985).
Suh, P.-G., Ryu, S. H., Choi, W. C., Lee, K. Y. & Rhee, S. G. J. biol. Chem. 263, 14497–14504 (1988).
Drucker, B., Mamon, T. & Roberts, T. New Engl. J. Med. 321, 1383–1391 (1989).
Pfeiffer, J. R., Seagrave, J. C., Davis, B. H., Deanin, G. G. & Oliver, J. M. J. Cell Biol. 101, 2145–2155 (1985).
Chen, C. & Okayama, H. Molec. cell. Biol. 7, 2745–2752 (1987).
Smith, D. B. & Johnson, K. S. Gene 67, 31–40 (1988).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Margolis, B., Hu, P., Katzav, S. et al. Tyrosine phosphorylation of vav proto-oncogene product containing SH2 domain and transcription factor motifs. Nature 356, 71–74 (1992). https://doi.org/10.1038/356071a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/356071a0
This article is cited by
-
TGFβ promotes mesenchymal phenotype of pancreatic cancer cells, in part, through epigenetic activation of VAV1
Oncogene (2017)
-
VAV2, a guanine nucleotide exchange factor for Rac1, regulates glucose-stimulated insulin secretion in pancreatic beta cells
Diabetologia (2015)
-
Vav1
AfCS-Nature Molecule Pages (2010)
-
Rho family GTPases and their regulators in lymphocytes
Nature Reviews Immunology (2009)
-
Characterization of VIK-1: a new Vav-interacting Kruppel-like protein
Oncogene (2005)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.